
Enjoy complimentary customisation on priority with our Enterprise License!
The US Bilevel Positive Airway Pressure (BiPAP) Market size is estimated to grow by USD 154.51 million at a CAGR of 5.16% between 2023 and 2028.
Over the years, awareness and understanding of respiratory diseases have increased. The incidence of respiratory disorders has increased significantly; respiratory disease is expected to be the third leading cause of death in the future. The early detection of respiratory disease with counseling for treatment and smoking cessation can control respiratory disorders. Community leaders, policymakers, and government agencies are working in collaboration to create patient awareness about respiratory diseases. The Global Operations Leadership Development (GOLD) Program and the US chronic obstructive pulmonary disease (COPD) Coalition have made efforts to increase awareness about COPD and other respiratory diseases. Such efforts to increase the awareness of respiratory diseases and the available treatment and diagnostic options will drive the growth of the market during the forecast period.
To learn more about this report, Download Report Sample
The market share growth by the offline segment will be significant during the forecast period. Offline stores include the retail and specialty stores segment, such as supermarkets, hypermarkets, and other retailers. End-users, when purchasing BiPAP products from supermarkets and chemists in the US, can take advice from professional sales personnel before making the purchase. This will increase their awareness about the use of BiPAP products in the U.S.
Get a glance at the market contribution of various segments Download PDF Sample
The offline segment was the largest and was valued at USD 350.54 million in 2018. Further, drugstores and health professionals have helped consumers in accepting the use of BiPAP devices in the US. Despite the increasing sales in the past two years, most companies in the US are still challenged in terms of the display of their products in drugstores for promotion through advertisements. Moreover, the concept of open shelves in drugstores has enabled consumers to buy products without a prescription. Convenience stores and other general merchandise stores also contribute to the growth of the retail segment. The overall retail and specialty store is expected to witness steady growth during the forecast period as retailers have started dedicated additional shelves for BiPAP devices to ensure a larger footfall, thereby driving the demand in the bilevel positive airway pressure (BiPAP) market in US. Thus, the offline segment is expected to fuel the growth of the market significantly during the forecast period.
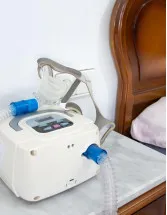
The devices segment is expected to grow during the forecast period, owing to factors such as the increasing number of product launches, rising prevalence of sleep apnea, business strategies adopted by key vendors, and favorable reimbursement policies for BiPAP devices in the US. The adoption of mobile health and mHealth technology by key vendors to make BiPAP therapy more efficient will increase the adoption of BiPAP devices, as they help monitor patients' conditions remotely. mHealth is a promising way of providing remote and easy clinical control for follow-ups and support to BiPAP devices in the US. Key vendors are developing mobile health platforms that connect wirelessly to BiPAP devices and care providers, which enables patients to use these machines at home. Such favorable insurance coverage for BiPAP devices, coupled with technological innovations, such as sleep tracking apps and the integration of software into BiPAP devices, will boost the adoption of BiPAP devices in the US in-home care settings, which in turn will fuel the growth of the devices segment, thereby driving the market during the forecast period.
Companies are implementing various strategies, such as strategic alliances, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the market. The report also includes detailed analyses of the competitive landscape of the market and information about 20 market companies, including:
3B Medical Inc. - The company offers bilevel positive airway pressure products such as the 3B Luna G3 30VT BiPAP machine, which has a variety of features designed to make BiPAP therapy more comfortable for users. These include five therapy modes, a humidifier, and an option for heated tubing.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key market players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
There are multiple factors influencing market growth. Our researchers analyzed the data with 2023 as the base year, along with the key drivers, market trends and analysis, and challenges.
The increasing prevalence of chronic disorders is notably driving market growth. Cardiovascular diseases (CVDs) are a group of illnesses that impact the heart and blood vessels, including coronary heart disease, peripheral arterial disease, aortic disease, and stroke. They are usually caused due to the high risk of blood clots and fatty deposition in arteries. An unhealthy and sedentary lifestyle, increasing geriatric population, consumption of excessive saturated trans-fat food, tobacco abuse, high blood pressure, smoking, high cholesterol, diabetes, obesity, physical inactivity, and family history of CVD are risk factors for CVDs.
Furthermore, angioplasty, atherectomy, bypass surgery, cardiomyoplasty, heart transplant, radiofrequency ablation, and surgical ventricular restoration are some of the cardiovascular surgeries. According to Eurostat, in Europe, Germany led the cardiovascular surgeries market in 2020, with approximately 51,000 heart bypass surgeries performed in the country. Hence, the demand for BiPAP devices is expected to increase as patients require surgeries to treat damaged and diseased heart valves, which, in turn, will drive the market growth during the forecast period.
An increase in breathing-related diseases is an emerging trend shaping market growth. The market witnessed a surge in the adoption of and demand for BiPAP devices in 2020. The mass assembly of employees at workplaces is strictly discouraged by governments and manufacturing associations to reduce the risk of disease transmission. Hence, vendors are implementing strict personal hygiene measures to ensure employee safety. For instance, Philips has implemented personal hygiene measures throughout the organization to maintain a safe working environment for employees performing business-critical activities such as R&D, production, and supply chain. In addition, new workspace guidelines, taking into consideration adequate safety measures, have led to additional expenses on safety gear, including face masks as well as hand sanitizers.
Moreover, the dependence of companies on single suppliers in their existing supply chain models is increasing the need for the timely procurement of the components and parts used in manufacturing BiPAP devices. As a result, bilevel positive airway pressure (BiPAP) market in US companies are constantly evaluating their options for working with multiple suppliers and have already started increasing their production capacity. Moreover, other health-related issues, such as asthma, are also increasing at a rapid rate in the US, which is further expected to fuel the market growth and trends during the forecast period.
The presence of stringent regulations is a significant challenge hindering market growth. Regulations are governed by state and foreign government agencies, such as the Food and Drug Administration (FDA) and the Center for Medicare Services (CMS) of the US Department of Health and Human Services (HHS). These regulatory agencies govern the launch of medical devices in terms of design, testing, manufacture, labeling, promotion, and marketing, which is tedious and costly for the manufacturers. Hence, the manufacturers of medical devices, such as BiPAP devices, should adhere to regulations and ensure that the distributed products are safe and effective for their intended use. These regulations differ from country to country.
Moreover, the FDAs Center for Devices and Radiological Health (CDRH) regulates the development, testing, manufacturing, labeling, and marketing of medical devices. Under the Federal Food, Drug, and Cosmetic Act (FFDCA) of 1938, medical devices are classified into Class I, Class II, and Class III, depending on the degree of risk associated with the use of the devices. BiPAP devices are classified under Class II devices with low-to-moderate risks. Hence, vendors must obtain premarket notification 510(k) before marketing such devices. This will hinder the growth of the market during the forecast period.
The market forecasting report includes the adoption lifecycle of the market research and growth, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the market analysis and report also include key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth and forecasting strategies.
Customer Landscape
The Global Bilevel Positive Airway Pressure (BiPAP) Devices Market is significantly influenced by various factors, including the prevalence of chronic respiratory conditions and an aging population seeking effective healthcare solutions. The market's growth is further propelled by continuous technological progress in respiratory devices, facilitating the expansion of healthcare-at-home initiatives. Education and awareness initiatives play a crucial role in driving the adoption of BiPAP devices, especially as respiratory disorders diagnoses rise globally. Despite these positive trends, challenges persist, such as the increasing costs of healthcare and concerns about hospital readmission rates.
Notably, patients' preference for non-invasive ventilation methods contributes to the market's expansion, although industry limitations such as BiPAP device costs, device compatibility issues, and the appeal of alternative medicines and equipment can impede growth. The comfort of the device and access to medical experts are critical factors influencing patient choices. Moreover, regulatory obstacles and uncertainties in the global economy may impact market dynamics. Infection risk and the necessary device maintenance requirements further shape the landscape of the BiPAP devices market, highlighting the need for a comprehensive approach to address both opportunities and challenges.
The market report forecasts market growth by revenue and provides an analysis of the latest trends and growth opportunities from 2018 to 2028.
|
US Bilevel Positive Airway Pressure (BiPAP) Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
149 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 5.16% |
|
Market growth 2024-2028 |
USD 154.51 million |
|
Market structure |
Fragmented |
|
YoY growth 2023-2024(%) |
4.54 |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
|
Key companies profiled |
3B Medical Inc., Beijing Aeonmed Co. Ltd., BMC MEDICAL CO. LTD., BPL MEDICAL TECHNOLOGIES Pvt. Ltd., Breas Medical AB, Deck Mount Electronics Pvt. Ltd., Eakin Healthcare Group, Fisher and Paykel Healthcare Corp. Ltd., Hunan Beyond Medical Technology Co. Ltd., Koninklijke Philips N.V., Lowenstein Medical Technology GmbH and Co. KG, Medical Depot Inc., Medtronic Plc, MOBIAK S.A, Norco Inc., Oxymed, ResMed Inc., Smiths Group Plc, Transcend Inc., and Vyaire Medical Inc. |
|
Market dynamics |
Parent market growth analysis, Market growth inducers and obstacles, Fast-growing and slow-growing segment analysis, COVID-19 impact and recovery analysis and future consumer dynamics, and Market condition analysis for the forecast period. |
|
Customization purview |
If our report has not included the data that you are looking for, you can reach out to our analysts and get segments customized. |
Download Sample PDF at your Fingertips
We can help! Our analysts can customize this market research report to meet your requirements. Get in touch
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation by Distribution Channel
7 Market Segmentation by Product
8 Market Segmentation by End-user
9 Customer Landscape
10 Drivers, Challenges, and Trends
11 Vendor Landscape
12 Vendor Analysis
13 Appendix
Get lifetime access to our
Technavio Insights
Cookie Policy
The Site uses cookies to record users' preferences in relation to the functionality of accessibility. We, our Affiliates, and our Vendors may store and access cookies on a device, and process personal data including unique identifiers sent by a device, to personalise content, tailor, and report on advertising and to analyse our traffic. By clicking “I’m fine with this”, you are allowing the use of these cookies. Please refer to the help guide of your browser for further information on cookies, including how to disable them. Review our Privacy & Cookie Notice.
