
Enjoy complimentary customisation on priority with our Enterprise License!
The global electrophysiology catheters market size is estimated to grow by USD 2,249.07 million at a CAGR of 8.73% between 2022 and 2027.
MI procedures are used to treat conditions such as cancer, cardiac arrhythmias, dermatological conditions, and selective solid tumors of the kidney, lung, liver, and musculoskeletal system. Many providers focus on conducting training programs on the use of electrophysiological catheters for healthcare providers. The MI procedure has become the standard technique in most surgeries through the use of improved electrophysiological devices. Hence, the increasing demand for MI procedures will drive the growth of the global electrophysiological catheter market during the forecast period.
Technavio has segmented the market into End-user, Product and Geography
It also includes an in-depth analysis of drivers, trends, and challenges. Our report examines historic data from 2017 to 2021, besides analyzing the current market scenario.
To learn more about this report, Request Free Sample
The market share growth by the hospitals and cardiac segment will be significant during the forecast period. Owing to the increasing incidence of heart disease and the rising trend of digital catheterization laboratories in hospitals and cardiovascular centers, hospitals are expected to hold the largest market share. The growth of the hospital market segment is driven by factors such as the increasing incidence of coronary heart disease and the concentration of key players.
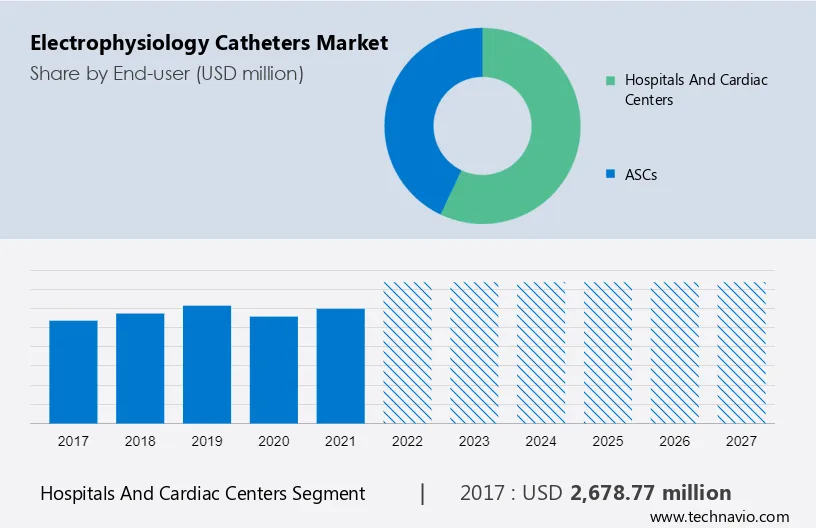
Get a glance at the market contribution of various segments View a PDF Sample
The hospitals and cardiac segment was valued at USD 2,678.77 million in 2017. The number of catheterization laboratories has risen significantly across the globe, which has driven the expansion of the global electrophysiological catheter market. Owing to the high quality of patient care, hospitals, and cardiology centers will have a high demand for electrophysiological catheters, which will drive the growth of the hospital and cardio center segment in the market during the forecast period.
The growing prevalence of chronic disorders and diseases, such as cardiovascular diseases, especially arrhythmias, is increasing the need for ablation catheters. The trend of patients seeking minimally invasive surgical methods has increased in the past few years. The advent of minimally invasive technologies, such as cryoablation, laser ablation, ultrasound ablation, and advanced mapping technologies, is also expected to fuel the growth of the market. Thus, such factors will boost the growth of the ablation catheter segment of the global electrophysiology catheters market during the forecast period.
The cardiac diagnostic catheter segment is experiencing moderate growth, driven by the increasing number of people performing cardiac ablation procedures. The cost of cardiac diagnostic catheters, used for electrophysiological mapping, is high because of the introduction of advanced technology. Companies like Abbott are focused on developing sensor-based cardiac diagnostic catheters that can be used to visualize the structure of the heart to treat various heart conditions. The main suppliers in the cardiac diagnostic catheter segment are Boston Scientific, Medtronic, Abbott, and BD. Thus, such factors will drive the growth of the segment during the forecast period.
For more insights about the market share of various regions View PDF Sample now!
North America is estimated to contribute 38% to the growth of the global market during the forecast period. Technavio’s analysts have elaborately explained the regional trends and drivers will shape the market during the forecast period. Developed countries in the region such as the US and Canada have well-established healthcare facilities, which increases the demand for balloon catheters and cardiac ablation catheters.
Moreover, market players use advanced technology to develop innovative products that outperform traditional devices in design and functionality. They also invest heavily in R&D to develop advanced balloon angioplasty. Companies operating in the US electrophysiological devices market are using these technologies to expand their product portfolio and increase market share. Hence, such factors are expected to drive market growth in this region during the forecast period.
The outbreak of COVID-19 in 2020, negatively affected the overall economy of North America, especially in the US. However, because all pharmaceuticals as well as the entire supply chain are grouped into essential services, the demand and supply of electrophysiological devices have not been significantly impacted in North America. In addition, in 2021, the implementation of large-scale vaccination campaigns will lift the lockdown restrictions, leading to the resumption of production and supply chain activities, thereby increasing demand for electrophysiological catheters in the region. Hence, such factors are expected to drive market growth in this region during the forecast period.
Buy Now Full Report and Discover More
Companies are implementing various strategies, such as strategic alliances, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the market. The report also includes detailed analyses of the competitive landscape of the market and information about 20 market companies, including:
Abbott Laboratories: The company offers electrophysiology catheters such as Advisor HD grid sensor-enabled mapping catheter.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key market players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
There are multiple factors influencing market growth. Our researchers analyzed the data with 2022 as the base year, along with the key drivers, trends, and challenges.
The rising incidence of cardiac diseases and the growth of insurance providers is the key factor driving the growth of the market. The prevalence of heart disease is increasing in both developed and developing countries. The incidence of valvular heart disease is also increasing due to increasing life expectancy worldwide. Valve disease is one of the most serious and common heart valve diseases and tends to increase with age. Likewise, uncontrolled or high blood pressure is one of the main causes of atrial fibrillation, which can lead to stroke or heart attack.
Furthermore, the EU offers the European Health Insurance Card (EHIC) allows people to choose the health services provided by each country. On the other hand, the UK's National Health Service (NHS) offers screening for medical conditions such as AF, heart attack, and peripheral artery disease. Thus, such factors will drive the growth of the global electrophysiology catheter market during the forecast period.
The use of remote navigation in cardiac mapping and imaging is the key trend in the market. Conventional cardiac mapping requires some experience to develop the necessary technical skills, as it involves the use of a combination of a deflectable catheter and a deflectable sheath. Thus, the development of a remote desktop positioning system with a software interface to facilitate catheter navigation has gained momentum. Over the past decade, remote-controlled catheter ablation has emerged as a new concept to improve catheter maneuverability and stability.
Furthermore, integrating remote navigation with a 3D mapping system can provide highly accurate 3D mapping through catheter-programmed mapping for the ablation of complex arrhythmias. This technology systematically reduces clinician fatigue and exposure to fluoroscopy, eliminates potential catheter manipulation errors, and reduces the risk of cardiac perforation. Hence, such factors will drive the growth of the global electrophysiology catheter market during the forecast period.
A stringent regulatory framework is a challenge that affects the growth of the market. Most electrophysiological devices such as catheters, are classified by the US FDA as Class II devices, subject to strict regulations before they are approved. Failure to provide data on the positive effects of clinical trials will affect US FDA approval of Class II and III devices. Electrophysiological catheters must comply with the respective regional/national regulations.
Furthermore, manufacturers of these devices must obtain regulatory approval before launching such products in the market, which is a lengthy and expensive process. Some advanced electrophysiological catheter manufacturers are in the process of having their products approved by various regulatory agencies. Therefore, the stringent regulatory approval process will impede the growth of the market during the forecast period.
Buy Now Full Report and Discover More
The market report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their growth strategies.
Global Electrophysiology Catheters Market Customer Landscape
The electrophysiology catheters market report forecasts market growth by revenue at global, regional & country levels and provides an analysis of the latest trends and growth opportunities from 2017 to 2027.
|
Electrophysiology Catheters Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
163 |
|
Base year |
2022 |
|
Historic period |
2017-2021 |
|
Forecast period |
2023-2027 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 8.73% |
|
Market growth 2023-2027 |
USD 2,249.07 million |
|
Market structure |
Fragmented |
|
YoY growth 2022-2023(%) |
7.97 |
|
Regional analysis |
North America, Europe, Asia, and Rest of World (ROW) |
|
Performing market contribution |
North America at 38% |
|
Key countries |
US, Germany, UK, China, and Japan |
|
Competitive landscape |
Leading Vendors, Market Positioning of Vendors, Competitive Strategies, and Industry Risks |
|
Key companies profiled |
Abbott Laboratories, Acutus Medical Inc., AngioDynamics Inc., APT Medical, AtriCure Inc., BIOTRONIK SE and Co. KG, Boston Scientific Corp., CardioFocus Inc., CathRx Pty Ltd., Japan Lifeline Co. Ltd., Johnson and Johnson, Koninklijke Philips N.V., Medtronic Plc, Merit Medical Systems Inc., MicroPort Scientific Corp., Millar Inc., Nihon Kohden Corp., OSYPKA AG, Siemens Healthineers AG, Stereotaxis Inc., and Stryker Corp. |
|
Market dynamics |
Parent market analysis, Market growth inducers and obstacles, Fast-growing and slow-growing segment analysis, COVID-19 impact and recovery analysis and future consumer dynamics, and Market condition analysis for the forecast period. |
|
Customization purview |
If our report has not included the data that you are looking for, you can reach out to our analysts and get segments customized. |
View Sample PDF at your Fingertips
We can help! Our analysts can customize this market research report to meet your requirements. Get in touch
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation by End-user
7 Market Segmentation by Product
8 Customer Landscape
9 Geographic Landscape
10 Drivers, Challenges, and Trends
11 Vendor Landscape
12 Vendor Analysis
13 Appendix
Get lifetime access to our
Technavio Insights
Cookie Policy
The Site uses cookies to record users' preferences in relation to the functionality of accessibility. We, our Affiliates, and our Vendors may store and access cookies on a device, and process personal data including unique identifiers sent by a device, to personalise content, tailor, and report on advertising and to analyse our traffic. By clicking “I’m fine with this”, you are allowing the use of these cookies. Please refer to the help guide of your browser for further information on cookies, including how to disable them. Review our Privacy & Cookie Notice.
