Preclinical CRO Market Size 2026-2030
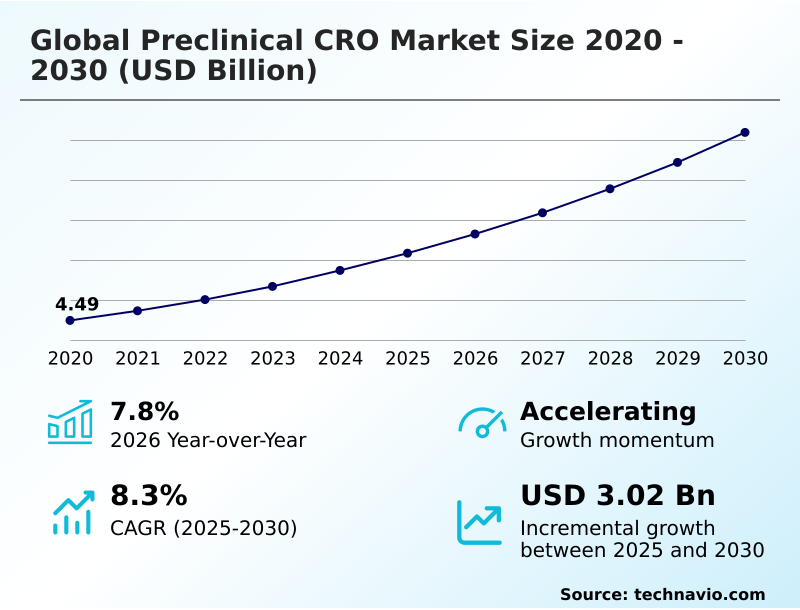
The preclinical cro market size is valued to increase by USD 3.02 billion, at a CAGR of 8.3% from 2025 to 2030. Regulatory modernization and industrialization of non-animal testing methodologies will drive the preclinical cro market.
Major Market Trends & Insights
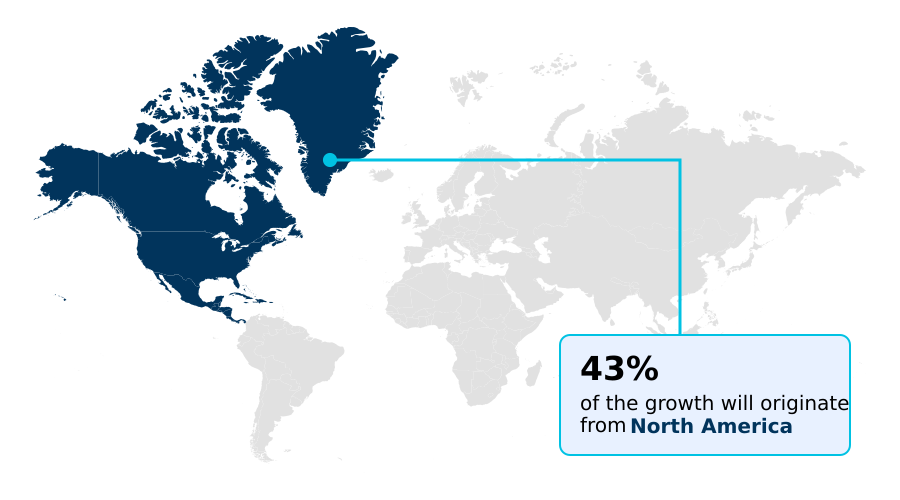
- North America dominated the market and accounted for a 42.8% growth during the forecast period.
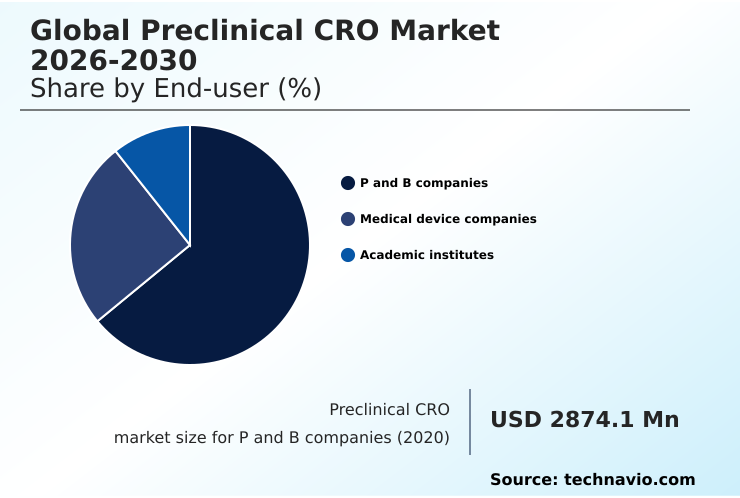
- By End-user - P and B companies segment was valued at USD 3.71 billion in 2024
- By Service - Toxicology testing segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 4.70 billion
- Market Future Opportunities: USD 3.02 billion
- CAGR from 2025 to 2030 : 8.3%
Market Summary
- The Preclinical CRO market is undergoing significant technical maturation, defined by a strategic shift from providing supplemental capacity to delivering high-fidelity scientific expertise. This evolution is driven by the increasing complexity of drug candidates, including cell and gene therapies, and the biopharmaceutical industry's imperative to mitigate early-stage clinical failures.
- Key services like preclinical safety assessment, drug metabolism and pharmacokinetics (dmpk), and integrated drug discovery are now table stakes. A pivotal business scenario involves leveraging predictive toxicology platforms early in the discovery phase.
- By using computational toxicology and in vitro toxicology assays, sponsors can de-risk compound libraries with greater precision, preventing the allocation of substantial R&D capital to candidates destined to fail in later IND-enabling studies.
- This data-driven approach, which integrates everything from high-throughput screening (hts) to advanced patient-derived xenograft (pdx) models, transforms the Preclinical CRO from a service vendor into a strategic partner. These organizations provide the essential validation framework required to transition promising molecules from laboratory research to clinical investigation, ensuring adherence to stringent good laboratory practice (glp) standards.
What will be the Size of the Preclinical CRO Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Preclinical CRO Market Segmented?
The preclinical cro industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
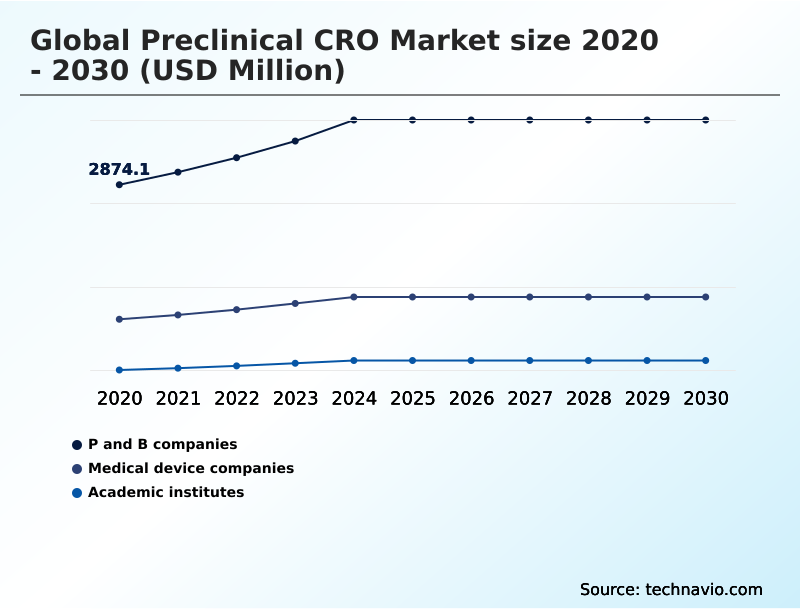
- End-user
- P and B companies
- Medical device companies
- Academic institutes
- Service
- Toxicology testing
- Bioanalysis and DMPK studies
- Compound management
- Others
- Application
- Oncology
- Neurology
- Cardiology
- Infectious diseases
- Others
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By End-user Insights
The p and b companies segment is estimated to witness significant growth during the forecast period.
The P and B companies segment represents the primary consumer of outsourced preclinical services, driven by the need to de-risk assets and accelerate development timelines for advanced therapy medicinal products (atmp).
These organizations increasingly partner with CROs for specialized expertise in drug metabolism and pharmacokinetics (dmpk), high-throughput screening (hts), and complex bioanalytical testing services. This strategic outsourcing allows internal teams to focus on core discovery and commercial strategy.
The reliance on external partners for absorption, distribution, metabolism, and excretion (adme) studies and the use of patient-derived xenograft (pdx) models is standard practice.
This collaborative model, which supports early-stage drug development support from large molecule bioanalysis to good laboratory practice (glp) compliance and regulatory submission support, underpins a significant portion of the market, with this segment constituting nearly 65% of activity.
The P and B companies segment was valued at USD 3.71 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 42.8% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Preclinical CRO Market Demand is Rising in North America Request Free Sample
The global geographic landscape is led by North America, which is projected to contribute over 42% of the market's incremental growth, fueled by a robust biotechnology ecosystem and high R&D spending.
This region shows strong demand for in vivo pharmacology and complex biologics testing, particularly for cell and gene therapy solutions and specialized oncology models.
Meanwhile, Asia is forecast to exhibit the highest regional growth rate at 8.7%, driven by expanding infrastructure and cost efficiencies in countries like China and India.
European markets remain critical for their expertise in immunogenicity testing and adherence to stringent regulatory standards.
Service providers are expanding their global footprint to offer harmonized preclinical cmc services and ex vivo analysis, leveraging preclinical imaging modalities to ensure data consistency across multi-regional trials, which is crucial for pharmacokinetic (pk) analysis and assessing pharmacodynamic (pd) biomarkers.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the biopharmaceutical sector is increasingly focused on optimizing the preclinical pathway. Selecting a preclinical CRO partner is no longer a simple procurement decision but a crucial strategic choice impacting long-term viability.
- Companies evaluate partners based on their capabilities in specific areas, such as providing a preclinical CRO for cell and gene therapy or offering robust in vivo efficacy models for oncology. The demand for GLP toxicology testing for small molecules remains high, while non-GLP screening and discovery services offer flexibility for early-stage research.
- A key consideration is the trade-off between traditional and new approach methodologies, where organ-on-a-chip vs animal models presents a compelling case for improved human relevance and cost savings. For instance, shifting from non-human primate model alternatives to patient-derived organoids in drug screening can reduce study costs by over 30% while mitigating ethical concerns.
- This shift is particularly impactful for ADME profiling for biologic drug candidates and the safety assessment of novel biologics. Effective PK/PD modeling and simulation services and specialized bioanalytical services for large molecules are critical for achieving ind-enabling studies timeline reduction.
- Firms must also address challenges in preclinical data reproducibility by investing in high-throughput toxicology screening methods and integrated preclinical to clinical services, ensuring a seamless transition for programs including preclinical research for orphan drugs.
What are the key market drivers leading to the rise in the adoption of Preclinical CRO Industry?
- Regulatory modernization and the resulting industrialization of non-animal testing methodologies serve as a primary driver propelling market growth and technological innovation.
- Regulatory modernization is a primary market driver, compelling the adoption of non-animal testing methodologies (nams). Legislative momentum is pushing drug developers to integrate sophisticated alternatives like organ-on-a-chip technology, human-relevant models, and bio-printed human tissues for preclinical safety assessment.
- This transition toward human-centric microphysiological systems is supported by regulatory roadmaps for phasing out mandatory animal testing, which enhances the value of in vitro toxicology assays. Domestic investment in such non-invasive evaluation technologies is growing by nearly 8% annually.
- This shift, which requires rigorous analytical method validation at GLP-certified laboratories, is instrumental in reducing the duration and ethical burden of investigational new drug (ind) enabling studies.
- By focusing on target identification and validation with higher human relevance, these new approach methodologies serve as a cornerstone for technological leadership.
What are the market trends shaping the Preclinical CRO Industry?
- The institutionalization of artificial intelligence and reinforcement learning is fundamentally reshaping early discovery workflows, compressing drug discovery timelines and enhancing predictive accuracy.
- A fundamental market trend is the institutionalization of artificial intelligence (AI) to compress drug discovery timelines. The industry is moving toward closed-loop discovery systems where predictive toxicology platforms analyze massive datasets to forecast molecular success. This data-driven pharmacology, leveraging computational toxicology and high-fidelity computational simulations, enables the de novo molecule design of compounds with optimized profiles.
- This shift has been shown to reduce the preclinical candidate development phase by more than 50% compared to traditional three-to-four-year cycles. Leading CROs are integrating generative AI for integrated drug discovery, transforming their role from service providers to strategic partners.
- Through customized assay development and multi-omics data integration, these firms offer lead optimization services that maximize the return on R&D investment by enhancing the predictability of preclinical outcomes.
What challenges does the Preclinical CRO Industry face during its growth?
- Geopolitical trade friction and the corresponding fragmentation of global bio-service supply chains represent a key challenge, introducing operational risks and cost inflation.
- The market is navigating significant disruption from geopolitical trade tensions, which are fragmenting global supply chains for non-clinical development. This has created operational risks for firms reliant on overseas partners for services such as GLP-compliant toxicology studies and access to non-human primate (nhp) models.
- The decoupling of research ecosystems has inflated cross-border collaboration costs by 15-20%, increasing competition for available laboratory capacity and contract vivarium services in Western markets. This environment complicates preclinical data management and forces sponsors to reconsider their functional service provider (fsp) models.
- The administrative and capital-intensive task of reshoring activities like safety pharmacology and accessing rodent research models for translational research poses a primary hurdle, threatening to delay IND-enabling toxicology package timelines.
Exclusive Technavio Analysis on Customer Landscape
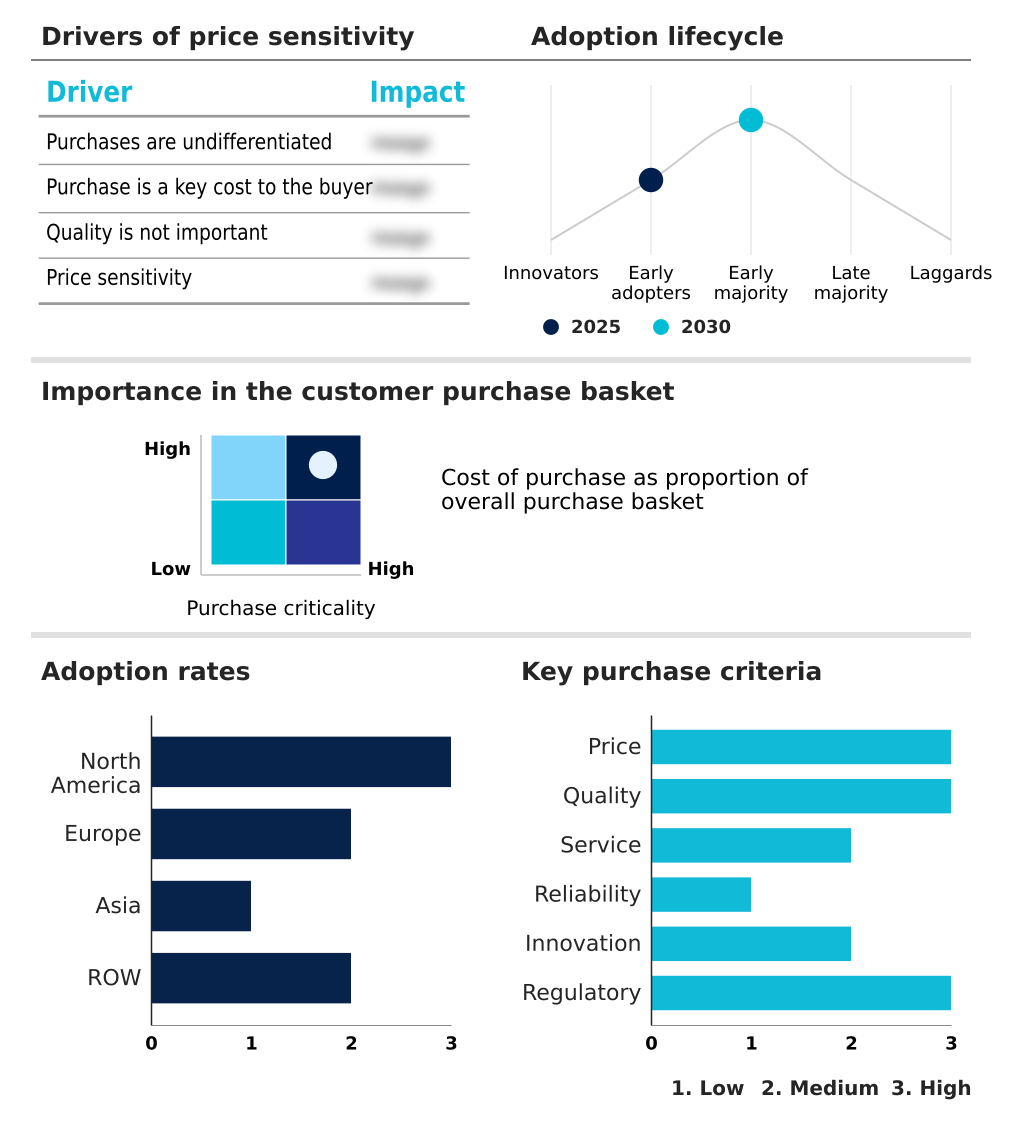
The preclinical cro market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the preclinical cro market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Preclinical CRO Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, preclinical cro market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Altasciences - Offerings are centered on delivering in vivo pharmacology, comprehensive toxicology studies, and advanced therapy development, with a specific focus on cell and gene therapy applications.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Altasciences
- AmplifyBio
- BioEmission Technology Solutions
- Charles River Laboratories
- CYNBIOSE
- Eurofins Scientific SE
- Gempharmatech Co. Ltd.
- Global Center for Medical Innovation
- ICON plc
- Imavita S.A.S.
- IQVIA Holdings Inc.
- JSR Corp.
- Kunming Biomed International Ltd.
- Laboratory Corp.
- Medpace Holdings Inc.
- Parexel International Corp.
- Thermo Fisher Scientific Inc.
- Veeda Clinical Research Ltd.
- Vivotecnia
- WuXi AppTec Co. Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Preclinical cro market
- In April 2025, the US Food and Drug Administration released its finalized roadmap outlining the comprehensive phasing out of mandatory animal testing for monoclonal antibodies, signaling a major shift in nonclinical safety assessment protocols.
- In January 2025, a leading contract research organization announced the launch of a new generative AI platform designed to accelerate de novo molecule design, reportedly reducing initial screening timelines by up to 40% for certain compound classes.
- In November 2024, a specialized toxicology CRO announced the successful validation of its new organ-on-a-chip platform for cardiotoxicity screening, offering a qualified, human-relevant alternative to traditional animal models for regulatory submissions.
- In September 2024, a major biopharma services company completed its acquisition of a niche provider of patient-derived xenograft (PDX) models, significantly expanding its specialized oncology research capabilities for immuno-oncology studies.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Preclinical CRO Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 293 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 8.3% |
| Market growth 2026-2030 | USD 3023.4 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 7.8% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, South Korea, Indonesia, Thailand, Brazil, Saudi Arabia, UAE, Turkey, South Africa, Israel, Argentina and Colombia |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The market is defined by a technology-driven evolution where high-fidelity computational simulations and closed-loop discovery systems are becoming standard. Boardroom-level strategy now directly connects R&D investment to the adoption of artificial intelligence in drug discovery for lead optimization services and de novo molecule design.
- This integration facilitates a shift towards predictive, human-relevant models, utilizing technologies like organ-on-a-chip and bio-printed human tissues. The core service portfolio, encompassing preclinical safety assessment, drug metabolism and pharmacokinetics (dmpk), and investigational new drug (ind) enabling studies, is being revolutionized.
- A focus on data from in vivo pharmacology, bioanalytical testing services, and high-throughput screening (hts) allows for a more robust non-clinical development process. Firms are leveraging translational research models, including patient-derived xenograft (pdx) and rodent research models, alongside advanced in vitro toxicology assays.
- This data-centric approach has demonstrated the ability to shorten preclinical timelines to as little as thirteen months by optimizing studies like chronic toxicology testing, safety pharmacology, and immunogenicity testing, all conducted under stringent good laboratory practice (glp).
What are the Key Data Covered in this Preclinical CRO Market Research and Growth Report?
-
What is the expected growth of the Preclinical CRO Market between 2026 and 2030?
-
USD 3.02 billion, at a CAGR of 8.3%
-
-
What segmentation does the market report cover?
-
The report is segmented by End-user (P and B companies, Medical device companies, and Academic institutes), Service (Toxicology testing, Bioanalysis and DMPK studies, Compound management, and Others), Application (Oncology, Neurology, Cardiology, Infectious diseases, and Others) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Regulatory modernization and industrialization of non-animal testing methodologies, Geopolitical trade friction and fragmentation of global bio-service supply chains
-
-
Who are the major players in the Preclinical CRO Market?
-
Altasciences, AmplifyBio, BioEmission Technology Solutions, Charles River Laboratories, CYNBIOSE, Eurofins Scientific SE, Gempharmatech Co. Ltd., Global Center for Medical Innovation, ICON plc, Imavita S.A.S., IQVIA Holdings Inc., JSR Corp., Kunming Biomed International Ltd., Laboratory Corp., Medpace Holdings Inc., Parexel International Corp., Thermo Fisher Scientific Inc., Veeda Clinical Research Ltd., Vivotecnia and WuXi AppTec Co. Ltd.
-
Market Research Insights
- Market dynamics are increasingly shaped by the dual pressures of innovation and efficiency. The adoption of predictive toxicology platforms and data-driven pharmacology enables a reduction in development timelines by over 50% compared to traditional methods. This acceleration is critical for sponsors seeking early-stage drug development support.
- However, this progress is tempered by geopolitical headwinds that have inflated cross-border research collaboration costs by approximately 15-20%. As a result, firms are re-evaluating their reliance on certain functional service provider (fsp) models and seeking partners that offer robust scientific and regulatory consulting to navigate supply chain complexities.
- The ability to provide customized assay development and integrated discovery and development services in a cost-effective, decentralized model is becoming a key differentiator for success.
We can help! Our analysts can customize this preclinical cro market research report to meet your requirements.


 RIA -
RIA - 
