Overview of the drug development pipeline for uveal melanoma
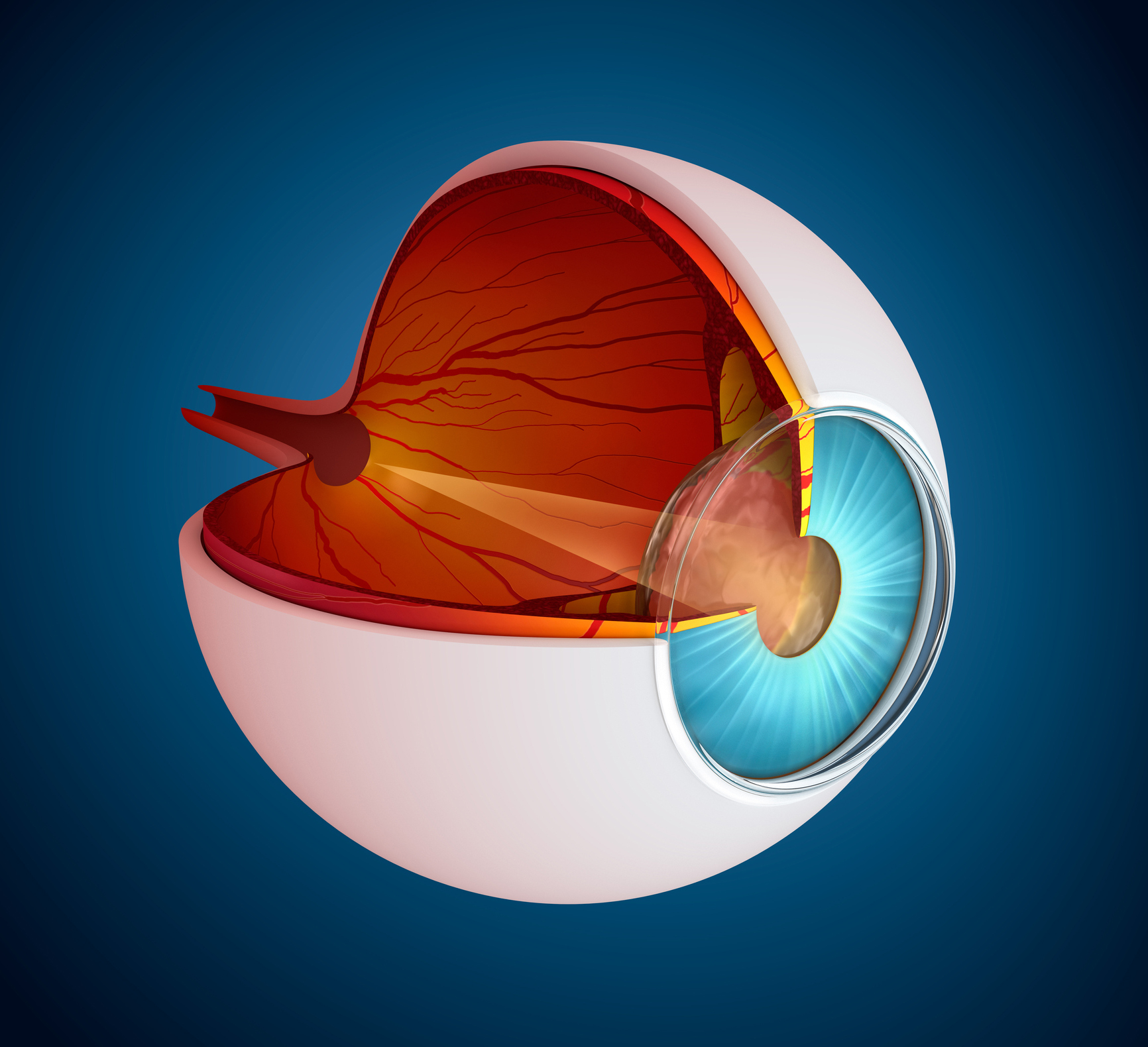
Uveal melanoma is one of the most common ocular melanomas. It arises from melanocytes in the iris, ciliary body, or choroid. Early diagnosis and local treatment are crucial, as the survival rate depends on them. However, approximately 50% of patients will develop metastatic disease with 6-12 months survival from metastatic diagnosis. The genomic analysis led to the development of gene-expression profiles, which predict metastatic progression effectively. The incidence of uveral melanoma varies by gender, race, and country. According to the National Cancer Institute, males have a higher incidence rate than females in the US with a ratio of 4.9 vs 3.7 per million of population. The incidence rate in the US remains lower than the rest of the world where it ranges between 5.3 and 10.9 cases per million of population. As a result, with increasing incidences of the disease, the drug development for uveal melanoma is expected to increase considerably in the forthcoming years.
According to this pipeline analysis report, most of the drug molecules in the pipeline are being developed for uveal melanoma. Our market research analysts have also identified that most of these molecules are in the pre-clinical development stage and a considerable number of molecules have been discontinued from development.
Companies covered
This pipeline analysis report provides a detailed analysis of the companies that are involved in the development of drug molecules for the treatment of uveal melanoma. In addition to providing information on the various stages of molecules developed by companies for different indications, this pipeline analysis report also provides information about the drug molecules discontinued by companies.
Some of the companies covered in this pipeline analysis report are –
- A6 Pharmaceuticals
- Aura Biosciences
- Bellicum Pharmaceuticals
- Delcath Systems
- Immunocore
Therapeutic assessment of the drug development pipeline for uveal melanoma by route of administration
- Oral
- Intravenous
- Intravitreal
- Intraarterial
The oral route of administration (ROA) involves the application of the drug directly into the mouth cavity, which will have a more direct effect on the target cells.
Therapeutic assessment of the drug development pipeline for uveal melanoma by therapy
- Monotherapy
- Combination therapy
- Monotherapy/Combination therapy
According to this pipeline analysis report, most of the molecules that are currently in the drug development pipeline for uveal melanoma are being developed as monotherapy drugs and most of these molecules are in the pre-clinical stage of development.
Key questions answered in the report include
- What are the drug molecules in the various development stages for uveal melanoma?
- What are the companies that are currently involved in the development of drug molecules for uveal melanoma?
- Insight into discontinued/inactive molecules with appropriate reasoning?
- What are the major regulatory authorities approving drugs in various regions?
- Detailed profiling of each active molecule
Technavio also offers customization on reports based on specific client requirement.
 RIA -
RIA - 
