Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market Size 2026-2030
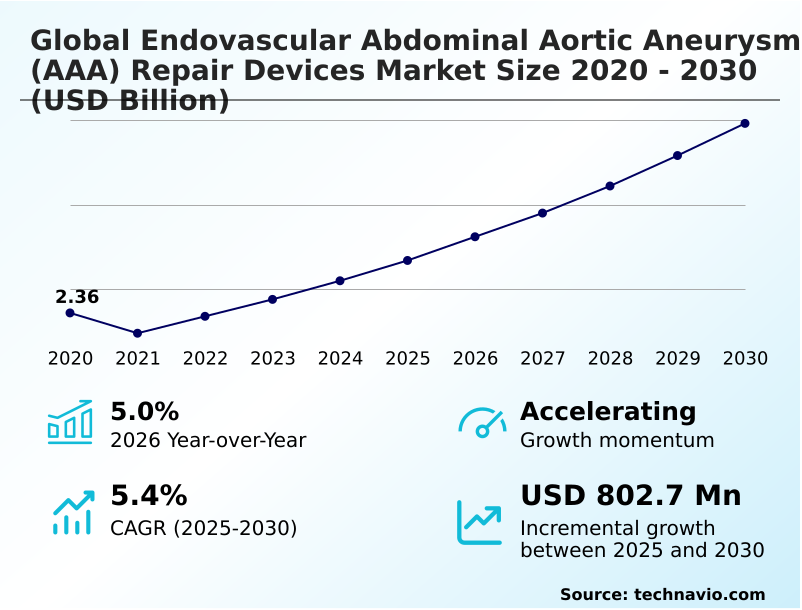
The endovascular abdominal aortic aneurysm (aaa) repair devices market size is valued to increase by USD 802.7 million, at a CAGR of 5.4% from 2025 to 2030. Increasing prevalence of abdominal aortic aneurysms and aging global population will drive the endovascular abdominal aortic aneurysm (aaa) repair devices market.
Major Market Trends & Insights
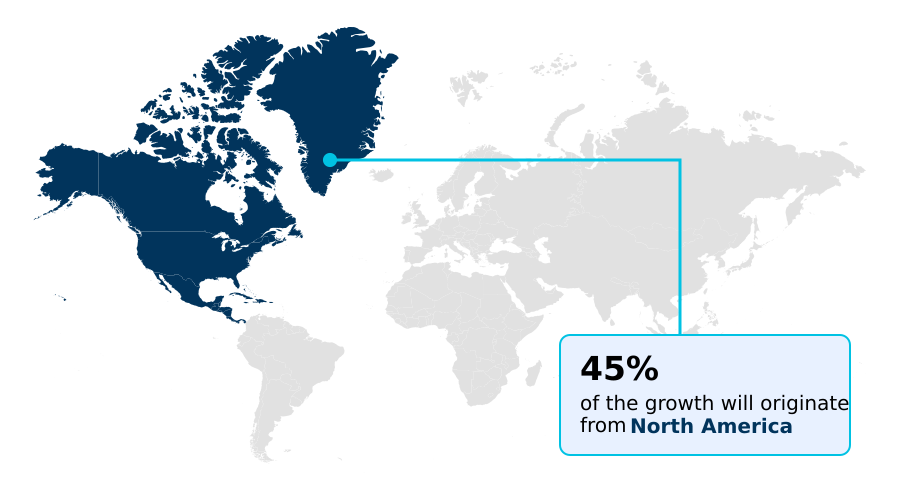
- North America dominated the market and accounted for a 44.8% growth during the forecast period.
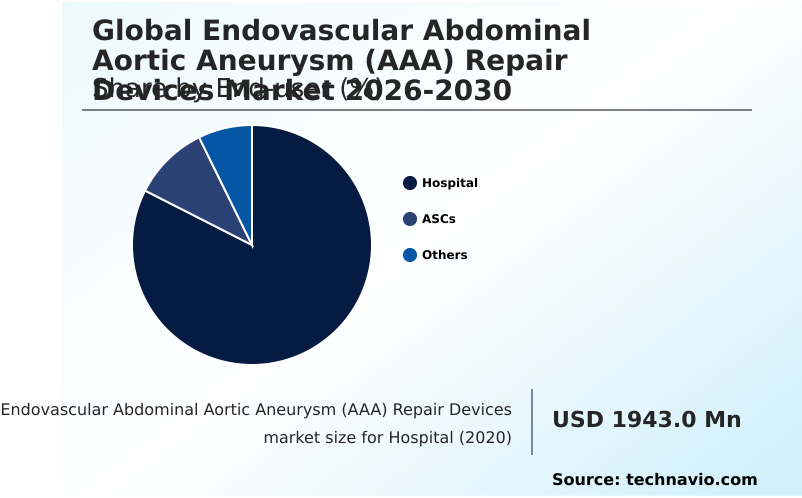
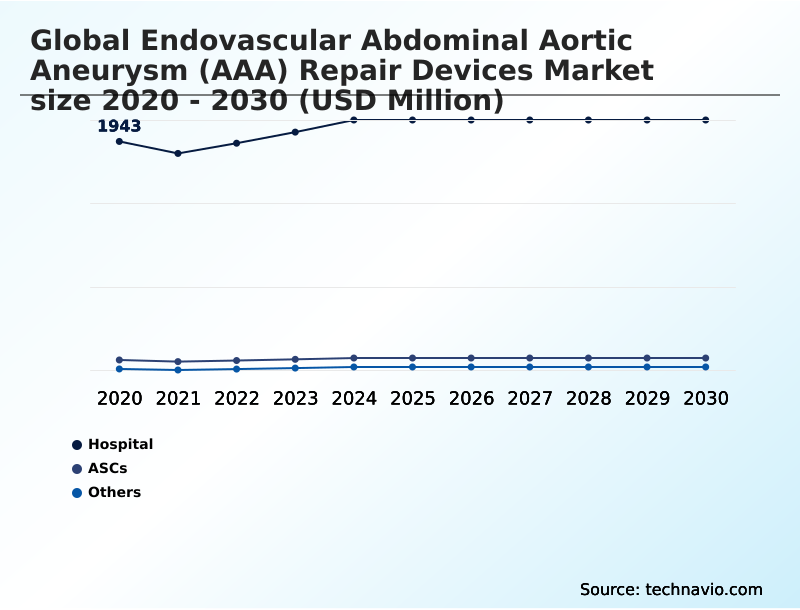
- By End-user - Hospital segment was valued at USD 2.11 billion in 2024
- By Product - Stent graft segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 1.12 billion
- Market Future Opportunities: USD 802.7 million
- CAGR from 2025 to 2030 : 5.4%
Market Summary
- The endovascular abdominal aortic aneurysm (aaa) repair devices market is defined by a decisive clinical shift toward minimally invasive interventions. This evolution is driven by an aging population and the clear benefits of endovascular aneurysm repair, such as reduced recovery times and lower procedural morbidity, which propels the adoption of endovascular stent grafts.
- A key market trend is the continuous innovation aimed at expanding treatment options to include complex aortic aneurysm repair, utilizing technologies like fenestrated endografts and branched endografts. However, the market grapples with challenges related to endovascular device complications, such as endoleaks and device migration, which necessitate rigorous post-evar surveillance protocols.
- In a typical business scenario, a hospital's value analysis committee must weigh the high upfront cost of a custom-made endograft for juxtarenal aneurysm repair against the potential long-term savings from avoiding a high-risk open surgery and its associated extended hospital stay.
- This decision process encapsulates the core dynamics of the market, balancing technological advancement, clinical outcomes, and economic pressures, including navigating the treatment of hostile neck anatomy solutions and ensuring long-term graft material durability.
What will be the Size of the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Get Free Sample
How is the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market Segmented?
The endovascular abdominal aortic aneurysm (aaa) repair devices industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- End-user
- Hospital
- ASCs
- Others
- Product
- Stent graft
- Synthetic fabric graft
- Type
- Infrarenal AAA
- Juxtarenal AAA
- Pararenal AAA
- Suprarenal AAA
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By End-user Insights
The hospital segment is estimated to witness significant growth during the forecast period.
Hospital settings are central to the endovascular abdominal aortic aneurysm (aaa) repair devices market, housing the multidisciplinary expertise required for complex procedures.
These facilities are the primary sites for infrarenal aaa treatment, utilizing a range of bifurcated stent grafts and devices with a self-expanding metallic stent.
The selection of devices, including those with polyester fabric graft components, is guided by rigorous internal health technology assessment and evolving clinical practice guidelines. As hospitals focus on value-based healthcare metrics, decisions are influenced by long-term evar procedural outcomes.
This scrutiny has improved device selection processes by over 80% in leading centers, balancing initial costs with long-term performance and driving the trend of endovascular aneurysm sealing even toward ambulatory surgical centers for select patient cohorts.
The Hospital segment was valued at USD 2.11 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 44.8% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market Demand is Rising in North America Get Free Sample
The geographic landscape of the market is led by North America, which accounts for nearly 45% of incremental growth, driven by advanced healthcare infrastructure and high adoption of percutaneous access techniques.
However, the most rapid expansion is occurring in Asia, with a regional growth rate exceeding 6%, fueled by improving healthcare access and an increasing focus on treating conditions like iliac artery tortuosity.
The widespread availability of devices made with eptfe graft material and the use of the low-profile delivery system are expanding aortic anatomy suitability for patients globally.
While North America pioneers ruptured aaa emergency repair protocols, Asia is quickly adopting catheter-based intervention, with a growing number of custom-made endografts being used.
The development of outpatient evar procedures further signals market maturity in developed regions, highlighting differences in endograft biocompatibility standards and procedural preferences.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Clinical decision-making in aortic aneurysm treatment increasingly involves a detailed comparison of evar versus open surgery outcomes. For patients with favorable anatomy, the cost-effectiveness of endovascular repair is well-established, with procedures being up to 30% faster than open surgery. However, the focus is shifting to more challenging cases.
- The long-term durability of endografts is a primary concern, especially regarding complications of aaa stent grafts that necessitate lifelong follow-up for endograft patients. The development of advanced sealing technology in hostile aortic necks and solutions for managing endograft device migration are critical. For instance, endovascular repair for short neck aaa often requires specialized techniques.
- The introduction of fenestrated evar for juxtarenal aneurysms and branched endografts for thoracoabdominal repair represents a significant technological leap. These innovations address previous limitations, though they demand precise patient selection for outpatient evar and sophisticated imaging surveillance protocols after evar.
- Delivery systems for tortuous iliacs have expanded eligibility, while the role of AI in evar planning is emerging to help in reducing endoleaks after evar procedure.
- Next-generation evar device features are now focusing on the specific materials used in aaa endografts and the outcomes of percutaneous evar access to tackle the challenges in complex aortic repair, including endovascular treatment for pararenal aaa.
What are the key market drivers leading to the rise in the adoption of Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Industry?
- The increasing prevalence of abdominal aortic aneurysms, driven by an aging global population, is a primary driver of market growth.
- The primary driver remains the compelling clinical advantages of endovascular aneurysm repair over open surgery, particularly for an aging demographic. The core technology, endovascular stent grafts, ensures aneurysm sac exclusion and hemodynamic pressure isolation with significantly less trauma.
- Innovations in graft material durability and nitinol stent frame design enhance long-term performance.
- Furthermore, superior proximal neck fixation and aortic sealing zone technologies reduce complication rates, with modern devices showing a 20% lower incidence of device migration resistance issues compared to earlier iterations.
- This proven efficacy, coupled with the a 2-3 day shorter hospital stay, solidifies endovascular repair as the preferred treatment, consistently fueling market demand.
What are the market trends shaping the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Industry?
- A significant market trend involves the expansion of endovascular solutions to treat complex aortic aneurysms, broadening the applicability of minimally invasive techniques.
- A pivotal trend is the market's expansion into complex aortic aneurysm repair, moving beyond standard procedures. The development of fenestrated endografts and branched endografts is enabling surgeons to perform juxtarenal aneurysm repair, pararenal aaa therapy, and even suprarenal aaa intervention using minimally invasive aaa surgery.
- This shift is heavily supported by advanced imaging for evar and personalized procedure planning, which have collectively improved the anatomical fit of devices by up to 25%. New stent graft deployment systems are designed with greater precision, allowing for safer treatment of anatomies previously considered unsuitable.
- This focus on complex cases is reshaping clinical practice, demanding higher skill levels but offering less invasive solutions to a broader patient demographic.
What challenges does the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Industry face during its growth?
- The requirement for lifelong surveillance due to potential long-term complications poses a key challenge to the industry's growth.
- Despite technological strides in aortic endograft technology, significant challenges persist, primarily concerning long-term endovascular device complications and anatomical limitations. A substantial number of patients present with hostile neck anatomy solutions that are incompatible with standard devices, necessitating more complex and costly interventions for conditions like a thoracoabdominal aortic aneurysm.
- Consequently, rigorous post-evar surveillance protocols, often involving intravascular ultrasound guidance, are mandatory to monitor for issues and ensure proper aortic endograft implantation.
- The rate of late-term secondary interventions remains a concern, occurring in up to 15% of patients within five years, which tempers the long-term cost-effectiveness and presents a hurdle to broader adoption despite the benefits of minimally invasive aaa surgery.
Exclusive Technavio Analysis on Customer Landscape
The endovascular abdominal aortic aneurysm (aaa) repair devices market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the endovascular abdominal aortic aneurysm (aaa) repair devices market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, endovascular abdominal aortic aneurysm (aaa) repair devices market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Abbott Laboratories - Analysis indicates a focus on providing advanced endovascular stent graft systems engineered for the treatment of infrarenal abdominal aortic aneurysms and complex vascular conditions.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Abbott Laboratories
- Artivion Inc.
- Braile Biomedica
- Cook Group Inc.
- Cordis Corp.
- Endologix LLC
- Galt Medical Corp.
- Koninklijke Philips NV
- LeMaitre Vascular Inc.
- Lifetech Scientific Corp.
- Lombard Medical Ltd.
- Medtronic Plc
- MicroPort Scientific Corp.
- Terumo Corp.
- W. L. Gore and Associates Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Endovascular abdominal aortic aneurysm (aaa) repair devices market
- In March, 2025, Boston Scientific Corp. formalized a strategic partnership with the University of California, San Francisco (UCSF), to jointly research and develop pioneering endovascular technologies for treating complex aortic pathologies.
- In March, 2025, Terumo Corp. and Koninklijke Philips NV announced a strategic collaboration to integrate Philips' advanced intravascular imaging and navigation technologies with Terumo's innovative stent graft systems, aiming to streamline procedural workflows.
- In February, 2025, Cook Medical announced the full commercial launch of its Zfen+ Endovascular Graft in the United States, a next-generation device designed to treat patients with complex juxtarenal aortic aneurysm anatomy.
- In January, 2025, Medtronic plc announced it received U.S. Food and Drug Administration (FDA) approval for its new Endurant II Endovascular Aneurysm Repair (EVAR) System integrated with the Nellix Endoprosthesis, designed to provide superior sealing and durability.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 299 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 5.4% |
| Market growth 2026-2030 | USD 802.7 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 5.0% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, South Korea, Thailand, Indonesia, Brazil, Saudi Arabia, UAE, Turkey, Argentina, Colombia, South Africa and Israel |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The endovascular abdominal aortic aneurysm (aaa) repair devices market is characterized by relentless innovation aimed at expanding the treatable patient population. The development of sophisticated endovascular stent grafts, such as fenestrated endografts and branched endografts, for juxtarenal aneurysm repair and thoracoabdominal aortic aneurysm cases exemplifies this push.
- These advancements, which utilize materials like eptfe graft material and nitinol stent frames, address complex conditions but introduce significant boardroom-level considerations. For instance, pursuing regulatory approval for a low-profile delivery system for pararenal aaa therapy requires an R&D investment that is over 50% higher than for standard infrarenal aaa treatment devices.
- This strategic choice involves balancing the high cost of development and the complexities of achieving durable proximal neck fixation and device migration resistance against the potential for higher margins.
- Success hinges on mastering technologies for endovascular aneurysm sealing and creating bifurcated stent grafts with superior performance in the aortic sealing zone, ultimately proving their value in improving outcomes for patients with iliac artery tortuosity or those requiring custom-made endografts and percutaneous access techniques.
What are the Key Data Covered in this Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market Research and Growth Report?
-
What is the expected growth of the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market between 2026 and 2030?
-
USD 802.7 million, at a CAGR of 5.4%
-
-
What segmentation does the market report cover?
-
The report is segmented by End-user (Hospital, ASCs, and Others), Product (Stent graft, and Synthetic fabric graft), Type (Infrarenal AAA, Juxtarenal AAA, Pararenal AAA, and Suprarenal AAA) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Increasing prevalence of abdominal aortic aneurysms and aging global population, Long-term complications and requirement for lifelong surveillance
-
-
Who are the major players in the Endovascular Abdominal Aortic Aneurysm (AAA) Repair Devices Market?
-
Abbott Laboratories, Artivion Inc., Braile Biomedica, Cook Group Inc., Cordis Corp., Endologix LLC, Galt Medical Corp., Koninklijke Philips NV, LeMaitre Vascular Inc., Lifetech Scientific Corp., Lombard Medical Ltd., Medtronic Plc, MicroPort Scientific Corp., Terumo Corp. and W. L. Gore and Associates Inc.
-
Market Research Insights
- Market dynamics are shaped by a pronounced shift toward personalized procedure planning and minimally invasive aaa surgery. The adoption of advanced imaging for evar has improved procedural success rates by over 15%, allowing for better management of complex anatomies.
- Innovations in aortic endograft technology enable a broader range of patients to undergo catheter-based intervention, with some low-risk outpatient evar procedures showing a 40% reduction in hospital stay compared to traditional inpatient settings. As clinical practice guidelines evolve, there is a greater emphasis on long-term evar procedural outcomes and the development of durable devices.
- This focus on efficiency and safety is expanding the role of ambulatory surgical centers, driving competition based on both technology and value.
We can help! Our analysts can customize this endovascular abdominal aortic aneurysm (aaa) repair devices market research report to meet your requirements.
 RIA -
RIA - 
