APAC - Epilepsy Therapeutic Market 2024-2028
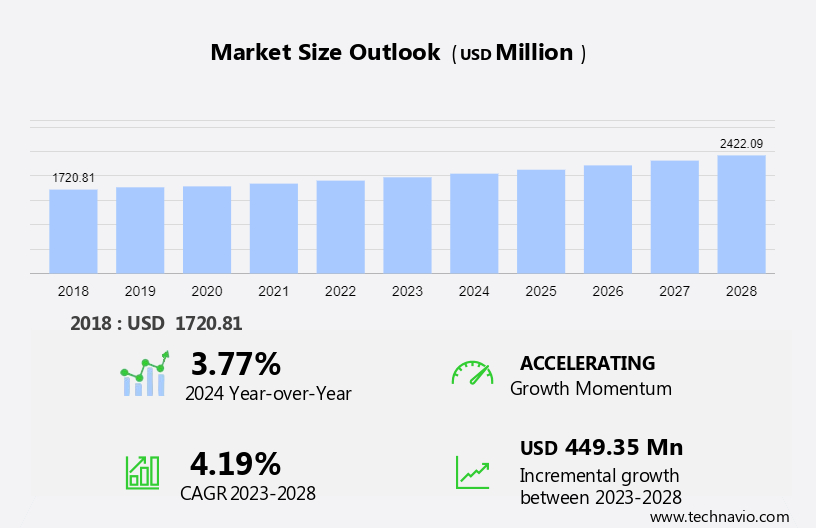
The APAC - Epilepsy Therapeutic Market size is forecast to increase by USD 449.35 million, at a CAGR of 4.19% between 2023 and 2028.
There has been a rising awareness regarding epilepsy across the world. The increasing awareness is a major factor driving the market growth by increasing demand for effective AEDs for the management of epilepsy. In addition, the campaign aims at increasing awareness and education about epilepsy and enhancing the social acceptance of people with epilepsy. In addition, it also aims to encourage governments and other organizations to extend their support to epilepsy patients and to identify the needs and requirements of epilepsy patients. Hence, such factors are positively impacting the market.
Technavio has segmented the market into Distribution Channel and Product
- The distribution channel segment is classified into hospital pharmacy, retail pharmacy, and online pharmacy
- The product segment is classified into first-generation epilepsy therapeutics, second-generation epilepsy therapeutics, and third-generation epilepsy therapeutics
It also includes an in-depth analysis of drivers, trends, and challenges. Our report examines historic data from 2018 - 2022, besides analyzing the current market scenario.
What will be the Size of the APAC Epilepsy Therapeutic Market During the Forecast Period?
To learn more about this report, Download Report Sample
APAC Epilepsy Therapeutic Market Segmentation by Distribution Channel and Product Analysis
Distribution Channel Analysis
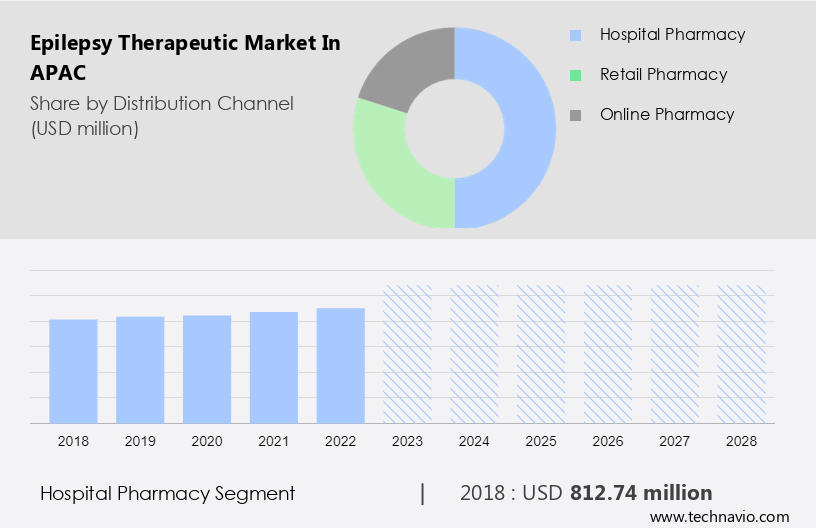
Hospital pharmacy
The hospital pharmacy segment is estimated to witness significant growth during the forecast period. This segment offers a wide variety of AEDs and related pharmaceuticals to meet the specific needs of epilepsy patients. In addition, it involves the direct involvement of healthcare professionals, such as pharmacists and doctors, who are involved in the prescription and dispensing of medications based on patient needs.
Get a glance at the market contribution of various segments Download PDF Sample
The hospital pharmacy segment was the largest segment and was valued at USD 812.74 million in 2018. Moreover, one of the biggest advantages of using a hospital pharmacy for epilepsy therapeutics is that it integrates seamlessly with the rest of the healthcare system. Therefore, healthcare providers can work closely together to create customized treatment plans and adjust medication regimens as needed based on patient feedback. In addition, hospital pharmacies also often carry a wide variety of epilepsy medications. Furthermore, this allows patients to have a single point of contact for all their pharmaceutical needs. Hence, such factors are fuelling the growth of this segment which in turn drives the market growth during the forecast period.
Product Analysis
First-generation epilepsy therapeutics
The growth of this segment is fuelled by its established adoption among end-users owing to the first-mover advantage in the market. In addition, some of these drugs, such as Dilantin and Tegretol, exert their pharmacologic effect by blocking the voltage-dependent sodium channels. Moreover, these drugs are highly efficacious; however, most of them cause severe adverse effects, which has led to their decreased adoption among people in developed and developing countries. For example, Dilantin (phenytoin) causes a rash, ataxia, hirsutism, gingival hypertrophy, and osteoporosis. Hence, these factors under the first-generation epilepsy products segment will drive the growth of the epilepsy therapeutics market in APAC during the forecast period.
Second-generation epilepsy therapeutics
The second-generation AEDs comprises zonisamide, topiramate, gabapentin, pregabalin, felbamate, tiagabine, lamotrigine, and levetiracetam. In addition, the second-generation AEDs offer various advantages such as simpler pharmacokinetic profiles over the first-generation AEDs. Moreover, the market is growing due to the availability of drugs with better safety and tolerability profiles when compared with their predecessors, thus driving the demand for these drugs in APAC. In addition, the developed infrastructure in Japan and Australia, coupled with the presence of major players such as Eisai Co. Ltd., H Lundbeck AS, and UCB, is likely to further drive market growth by increasing the production and commercialization of second-generation AEDs in APAC. Hence, such factors are fuelling the growth of this segment which in turn drives the market growth during the forecast period.
Key APAC Epilepsy Therapeutic MarketPlayers
Companies are implementing various strategies, such as strategic alliances, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the market. The report also includes detailed analyses of the competitive landscape of the market and information about 17 market companies, including:
Bausch Health Companies Inc: The company offers epilepsy therapeutics under the brand name Mysoline.
- Alkem Laboratories Ltd.
- BIAL
- Dr Reddys Laboratories Ltd.
- Eisai Co. Ltd.
- H Lundbeck AS
- Johnson and Johnson
- Novartis AG
- Pfizer Inc.
- Sanofi SA
- SK Inc.
- Sumitomo Pharma Co. Ltd.
- Sun Pharmaceutical Industries Ltd.
- Takeda Pharmaceutical Co. Ltd.
- Teva Pharmaceutical Industries Ltd.
- UCB SA
- GlaxoSmithKline Plc
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key market players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
APAC Epilepsy Therapeutic Market: Key Drivers, Trends, Challenges, and Customer Landscape
There are multiple factors influencing market growth. Our researchers analyzed the data with 2022 as the base year, along with the key drivers, trends, and challenges.
Key APAC Epilepsy Therapeutic Market Drivers
One of the key factors driving the APAC epilepsy therapeutic market growth is the increase in the patient population having epilepsy. There is an increasing prevalence of epilepsy with the annual incidence rate of epilepsy in developed countries is approximately 45-55 per 100,000 individuals, with higher rates in infants and older populations. In addition, in developing countries, the incidence rate is almost double, reaching about 95-105 per 100,000 individuals annually.
Moreover, the prevalence rate of epilepsy in developed countries is estimated between 5-10 per 1,000 individuals annually, whereas this rate is much higher in developing countries, reaching approximately 125-135 individuals annually. Furthermore, developing countries witness high incidence and prevalence rates due to the increasing population and lack of proper diagnostic tools to refrain from detecting the early symptoms of epilepsy. In addition, the increase in the patient population is expected to increase the consumption of AEDs, thereby driving the growth of the APAC epilepsy therapeutics market during the forecast period.
Significant APAC Epilepsy Therapeutic Market Trends
A key factor shaping the APAC epilepsy therapeutic market growth is the reformation of marketed epilepsy drugs. Drug delivery systems including transdermal patches, extended-release (ER) formulations, and orally disintegrating tablets increase the efficacy of drugs either by increasing patient compliance or by increasing the bioavailability of the drug. In addition, the reformulation of marketed drugs also provides an opportunity to grab patent term extensions, which can delay the entry of generic versions into the market.
Moreover, a single-loading dose was approved to provide an alternative to the standard titration schedule. Therefore, the reformulation of solid dosage forms such as tablets can provide a window for titrating the dose as per individual requirements. Furthermore, ER formulations such as KEPPRA XR reduce the dosing frequency, thereby increasing patient compliance. Therefore, it is positively impacting the APAC - epilepsy therapeutic market. Hence, such factors are driving the APAC epilepsy therapeutic market growth during the forecast period.
Major APAC Epilepsy Therapeutic Market Challenges
The social stigma associated with mental disorders is one of the key challenges hindering the APAC epilepsy therapeutic market growth. Epilepsy and associated seizures are perceived variably in different parts of the world, resulting in dejection, isolation, and denial of education. In addition, in some parts of the world, including APAC, public disclosure of symptoms triggers negative reactions from family and friends, leading to seclusion. Furthermore, misconceptions about seizures and epilepsy can induce premature loss of self-dependence and identity in the affected person.
Moreover, seizures associated with epilepsy are perceived variably in different parts of the world, including APAC, ranging from a component of aging to a supernatural event. In addition, inaccurate assumptions regarding the cause of epilepsy lead to wrong interpretations about the impact of this disease on family members. Furthermore, the social stigma associated with these diseases can have an adverse impact on the social and personal lives of caregivers, including family members. Hence, such factors are hindering the APAC epilepsy therapeutic market growth during the forecast period.
Key APAC Epilepsy Therapeutic Market Customer Landscape
The market report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their growth strategies.
APAC Epilepsy Therapeutic Market Customer Landscape
Segment Overview
The APAC epilepsy therapeutic market report forecasts market growth and provides a market growth analysis of the latest market trends and analysis and growth opportunities from 2018 to 2028.
- Distribution Channel Outlook
- Hospital pharmacy
- Retail pharmacy
- Online pharmacy
- Product Outlook
- First-generation epilepsy therapeutics
- Second-generation epilepsy therapeutics
- Third-generation epilepsy therapeutics
|
APAC Epilepsy Therapeutic Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
156 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 4.19% |
|
Market Growth 2024-2028 |
USD 449.35 million |
|
Market structure |
Fragmented |
|
YoY growth 2023-2024(%) |
3.77 |
|
Competitive landscape |
Leading Vendors, Market Positioning of Vendors, Competitive Strategies, and Industry Risks |
|
Key companies profiled |
Alkem Laboratories Ltd., Bausch Health Companies Inc., BIAL, Dr Reddys Laboratories Ltd., Eisai Co. Ltd., H Lundbeck AS, Johnson and Johnson, Novartis AG, Pfizer Inc., Sanofi SA, SK Inc., Sumitomo Pharma Co. Ltd., Sun Pharmaceutical Industries Ltd., Takeda Pharmaceutical Co. Ltd., Teva Pharmaceutical Industries Ltd., UCB SA, and GlaxoSmithKline Plc |
|
Market dynamics |
Parent market analysis, Market growth inducers and obstacles, Fast-growing and slow-growing segment analysis, COVID-19 impact and recovery analysis and future consumer dynamics, and Market condition analysis for the forecast period. |
|
Customization purview |
If our report has not included the data that you are looking for, you can reach out to our analysts and get segments customized. |
Download Sample PDF at your Fingertips
What are the Key Data Covered in this APAC Epilepsy Therapeutic Market in Regional Research Report?
- CAGR of the market during the forecast period
- Detailed information on factors that will drive the growth of the APAC epilepsy therapeutic market between 2023 and 2028
- Precise estimation of the APAC epilepsy therapeutic market size and its contribution to the market in focus on the parent market
- Detailed market growth and forecasting analysis of customer landscape covering Diver of price sensitivity, Adoption of the lifecycle, and key purchase criteria
- Companies landscape comparing criticality of inputs and factors of differentiation
- Exclusive Matrix on companies position and classification
- Accurate market forecast about upcoming market growth and trends and changes in consumer behavior
- Growth of the market across APAC
- A thorough market research and growth analysis of the market’s competitive landscape and detailed information about companies
- Comprehensive market analysis and report of factors that will challenge the growth of the APAC epilepsy therapeutic market companies
We can help! Our analysts can customize this market research report to meet your requirements. Get in touch

 RIA -
RIA - 
