Pharmaceutical Contract Manufacturing Market Size 2026-2030
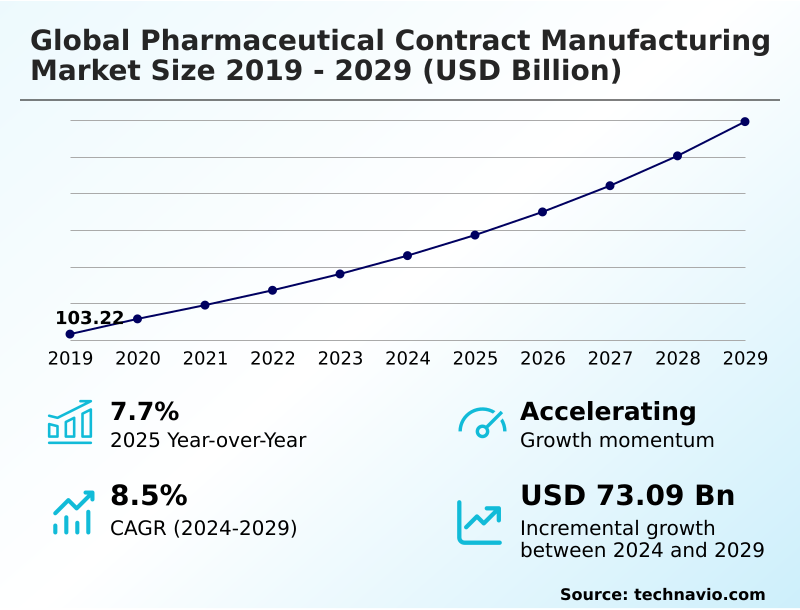
The pharmaceutical contract manufacturing market size is valued to increase by USD 76.40 billion, at a CAGR of 8.2% from 2025 to 2030. Increasing complexity and cost of research and development will drive the pharmaceutical contract manufacturing market.
Major Market Trends & Insights
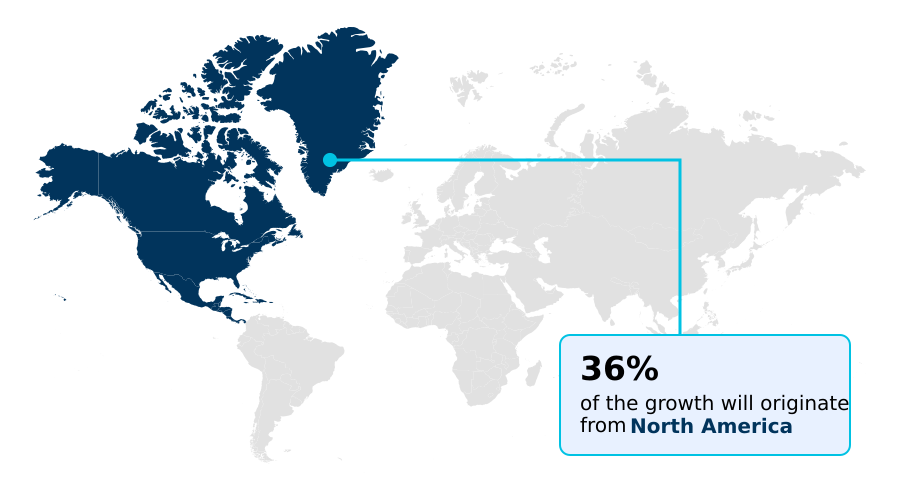
- Asia dominated the market and accounted for a 36.6% growth during the forecast period.
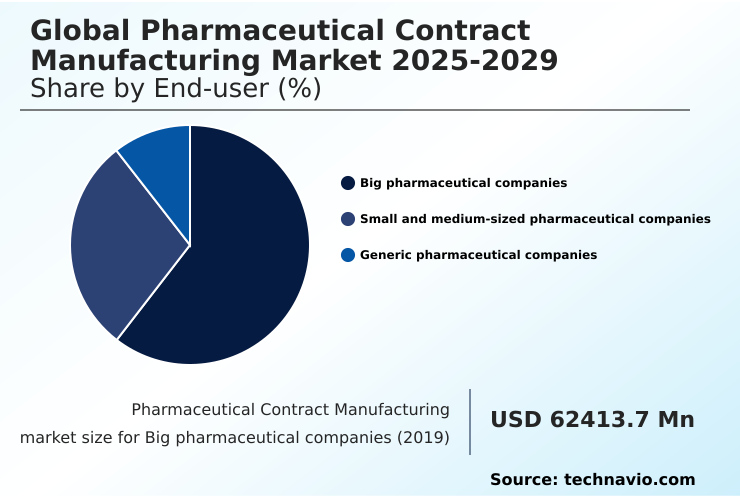
- By End-user - Big pharmaceutical companies segment was valued at USD 85.61 billion in 2024
- By Service - API and bulk drug manufacturing segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 122.11 billion
- Market Future Opportunities: USD 76.40 billion
- CAGR from 2025 to 2030 : 8.2%
Market Summary
- The pharmaceutical contract manufacturing market is expanding as drug developers increasingly outsource production to enhance efficiency and access specialized technologies. This trend is driven by the growing complexity of biologics and advanced therapies, which require significant capital investment and expertise in areas like aseptic fill-finish services and cell and gene therapy CDMO operations.
- Contract Development and Manufacturing Organizations (CDMOs) now act as strategic partners, supporting the entire lifecycle from clinical trial material (CTM) manufacturing to commercial scale-up. A key business scenario involves emerging biotechs partnering with a CDMO to navigate complex tech transfer management and regulatory submission support, allowing them to focus on core R&D activities.
- This reliance on outsourcing mitigates financial risk and accelerates time-to-market. The industry is also shaped by a move toward supply chain regionalization and the adoption of advanced continuous manufacturing platforms to ensure quality and supply continuity, with a strong emphasis on maintaining GMP compliance auditing across all partnerships.
What will be the Size of the Pharmaceutical Contract Manufacturing Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Pharmaceutical Contract Manufacturing Market Segmented?
The pharmaceutical contract manufacturing industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
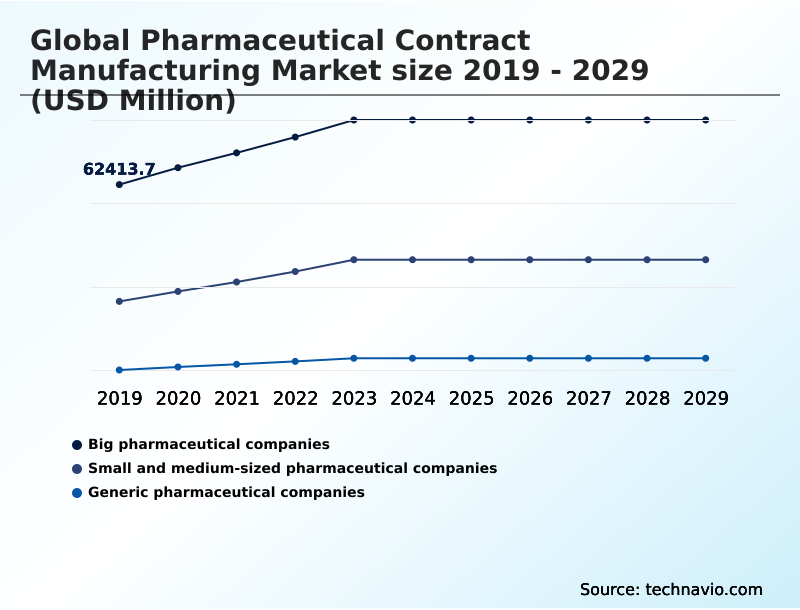
- End-user
- Big pharmaceutical companies
- Small and medium-sized pharmaceutical companies
- Generic pharmaceutical companies
- Service
- API and bulk drug manufacturing
- Final dosage form
- Secondary packaging
- Application
- Infectious diseases
- Cardiovascular diseases
- Oncology
- Geography
- North America
- US
- Canada
- Mexico
- Asia
- Europe
- Germany
- UK
- France
- Rest of World (ROW)
- North America
By End-user Insights
The big pharmaceutical companies segment is estimated to witness significant growth during the forecast period.
Big pharmaceutical companies are refining their approach to the global pharmaceutical contract manufacturing market 2026-2030, driven by persistent pharmaceutical outsourcing trends.
These firms leverage the specialized capabilities of a contract manufacturing organization (CMO) to achieve operational excellence in pharma and manage complex portfolios.
The focus is on long-term commercial drug production partners that can ensure robust pharmaceutical quality systems (PQS) and support the entire drug development outsourcing lifecycle.
This strategic shift facilitates commercial scale-up for both small molecule synthesis and complex oral solid dosage form manufacturing.
Partners are chosen based on their ability to conduct rigorous process validation services, quality control testing, and drug stability testing, ensuring that all outsourced activities pass strict GMP compliance auditing, with some firms seeing a 15% improvement in audit-readiness.
The Big pharmaceutical companies segment was valued at USD 85.61 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
Asia is estimated to contribute 36.6% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Pharmaceutical Contract Manufacturing Market Demand is Rising in Asia Request Free Sample
The geographic landscape of the global pharmaceutical contract manufacturing market 2026-2030 is shifting as regions specialize in high-value services.
North America is a leader in advanced therapy medicinal products (ATMP) manufacturing and specialty drug manufacturing, including complex antibody-drug conjugate (ADC) technology and cell and gene therapy CDMO services.
Investment in sterile injectable manufacturing and viral vector manufacturing is strong, with facilities improving lyophilization cycle development to cut processing times by over 15%. Europe remains a hub for high-potency API (HPAPI) manufacturing and innovative mRNA vaccine production.
The strategic importance of a reliable contract development and manufacturing organization (CDMO) is underscored by supply chain resilience initiatives, with firms diversifying their API sourcing strategy across continents to ensure stability.
This regional focus improves quality risk management (QRM) and operational excellence in pharma.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the global pharmaceutical contract manufacturing market 2026-2030 hinges on a nuanced understanding of outsourcing benefits and risks. When evaluating the cost of outsourcing pharma manufacturing, companies must look beyond initial pricing to the long-term benefits of continuous manufacturing pharma.
- The process of finding a reliable CDMO partner is critical, especially for organizations seeking small biotech manufacturing solutions, where overcoming cell therapy manufacturing challenges is paramount. The CDMO selection criteria for biologics are particularly stringent, demanding proven expertise in areas like biologics manufacturing cost analysis and gene therapy vector production scale-up.
- Successful tech transfer best practices pharma are essential for a smooth transition from development to commercialization. To ensure operational continuity, many firms now employ a dual sourcing API manufacturing strategy as a core tenet of improving pharma supply chain resilience. This includes optimizing cold chain for biologics and managing CDMO quality agreements effectively.
- Partners must demonstrate robust HPAPI handling and containment strategies and expertise in scaling up parenteral drug production. Exploring aseptic fill finish outsourcing costs and CDMO capacity reservation agreements early in development can prevent future bottlenecks.
- Firms that master regulatory compliance in contract manufacturing and mRNA manufacturing process development often reduce their time-to-market by a factor of two compared to those with less strategic oversight, especially when navigating challenges in ADC manufacturing.
What are the key market drivers leading to the rise in the adoption of Pharmaceutical Contract Manufacturing Industry?
- The increasing complexity and cost of research and development serve as a key driver for the market.
- The escalating complexity of new therapies is a major driver, expanding biopharmaceutical manufacturing capacity for large molecule manufacturing. The transition from clinical to commercial manufacturing requires sophisticated bioprocessing optimization and robust tech transfer management.
- Leading CDMOs that have embraced pharma 4.0 implementation report a 20% improvement in process yields for monoclonal antibody (mAb) production.
- Demand for continuous manufacturing platforms is rising to support biologics drug substance production through techniques like large-scale fermentation and downstream processing purification.
- The market is seeing more collaborative CDMO partnership models that integrate biopharmaceutical characterization early in the development cycle, reducing late-stage failures by over 15%. This integrated approach is essential for managing the intricate journey of advanced therapies.
What are the market trends shaping the Pharmaceutical Contract Manufacturing Industry?
- A significant market trend involves strategic onshoring to enhance supply chain resilience. This shift is particularly prominent in North America and Europe.
- A primary trend in the global pharmaceutical contract manufacturing market 2026-2030 is the strategic regionalization of supply chains to bolster pharmaceutical supply chain security. This has increased demand for US-based pharmaceutical manufacturing and strengthened the European CDMO market.
- To enhance supply chain agility, companies are investing in flexible manufacturing facilities capable of handling diverse products, including parenteral drug manufacturing and prefilled syringe filling. The focus on biologics has made cold chain logistics for biologics a critical competency, with specialized CDMOs demonstrating a 25% reduction in temperature excursions.
- Investments in single-use bioreactor systems are improving efficiency in clinical trial material (CTM) manufacturing. Furthermore, advanced supply chain serialization technologies are being implemented to ensure end-to-end traceability, with adoption rates increasing by 40% among top-tier CDMOs.
What challenges does the Pharmaceutical Contract Manufacturing Industry face during its growth?
- Heightened regulatory scrutiny and complexity present a key challenge affecting industry growth.
- Navigating stringent regulatory requirements and technical complexities presents a significant challenge. The industry faces persistent fill-finish capacity constraints, particularly for aseptic fill-finish services, which drives the need for partners with proven aseptic processing validation.
- Adhering to evolving aseptic processing standards and ensuring data integrity in manufacturing are critical, as regulators have increased scrutiny in these areas by over 30% in recent audits. The successful execution of pharmaceutical technology transfer requires meticulous planning to avoid costly delays. Many firms now seek end-to-end CDMO services to streamline regulatory submission support and analytical method validation.
- Implementing quality by design (QbD) principles and outsourcing complex peptide synthesis and purification or bioconjugation services are becoming standard strategies to mitigate compliance risks.
Exclusive Technavio Analysis on Customer Landscape
The pharmaceutical contract manufacturing market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the pharmaceutical contract manufacturing market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Pharmaceutical Contract Manufacturing Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, pharmaceutical contract manufacturing market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
AbbVie Inc. - Provides integrated solutions from oral dose development and biologics manufacturing to specialty drug services and clinical supply manufacturing, addressing complex pharmaceutical pipeline needs.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- AbbVie Inc.
- Aenova Holding GmbH
- Ajinomoto Co. Inc.
- Almac Group Ltd.
- Boehringer Ingelheim GmbH
- Cadila Pharmaceuticals Ltd.
- Cambrex Corp.
- Catalent Inc.
- Charles River Laboratories
- Cmic Holdings Co. Ltd.
- Corden Pharma Intl. GmbH
- Dr. Reddys Laboratories Ltd.
- Evonik Industries AG
- Lonza Group Ltd.
- PCI Pharma Services
- Recipharm AB
- Samsung Biologics Co. Ltd.
- Siegfried Holding AG
- Thermo Fisher Scientific Inc.
- WuXi Biologics Cayman Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Pharmaceutical contract manufacturing market
- In December, 2024, Novo Holdings finalized its USD 16.5 billion acquisition of Catalent, a strategic move to secure critical manufacturing capacity, which was followed by the sale of three key fill-finish sites to Novo Nordisk for USD 11 billion.
- In September, 2025, FUJIFILM Diosynth Biotechnologies inaugurated its new commercial-scale biomanufacturing facility in Holly Springs, North Carolina, a USD 3 billion investment designed to be one of North America's largest cell culture sites.
- In September, 2025, Moderna delivered the first mRNA vaccines manufactured at its new facility in Laval, Quebec, establishing localized production capabilities within Canada and demonstrating the trend of regionalizing vaccine supply chains.
- In April, 2025, Thermo Fisher Scientific Inc. announced the operational launch of its expanded St. Louis biologics site, a key part of its multi-billion dollar US investment, adding four 5,000L bioreactors to boost large-scale monoclonal antibody production.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Pharmaceutical Contract Manufacturing Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 296 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 8.2% |
| Market growth 2026-2030 | USD 76396.2 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 7.9% |
| Key countries | US, Canada, Mexico, China, India, Japan, South Korea, Singapore, Indonesia, Thailand, Germany, UK, France, The Netherlands, Italy, Spain, Switzerland, Brazil, Australia, Saudi Arabia, UAE, South Africa and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The global pharmaceutical contract manufacturing market 2026-2030 is defined by a strategic shift toward outsourcing complex manufacturing processes. Boardroom-level decisions increasingly focus on leveraging external expertise for biologics drug substance production, including monoclonal antibody (mAb) production and advanced antibody-drug conjugate (ADC) technology. The need for specialized capabilities in small molecule synthesis, peptide synthesis and purification, and bioconjugation services drives partnerships.
- Firms now rely on CDMOs for the entire product lifecycle, from drug product formulation development and clinical trial material (CTM) manufacturing to commercial scale-up and regulatory submission support. This includes critical services like sterile injectable manufacturing, aseptic fill-finish services using prefilled syringe filling, parenteral drug manufacturing, and liquid fill and finish.
- Specialized techniques such as lyophilization cycle development are outsourced to achieve efficiency gains, with some partners reducing cycle times by 20%. Expertise in high-potency API (HPAPI) manufacturing, cell and gene therapy CDMO services, viral vector manufacturing, and mRNA vaccine production is a key differentiator.
- Quality is paramount, governed by quality by design (QbD) principles, rigorous analytical method validation, process validation services, and GMP compliance auditing. Systems for tech transfer management, supply chain serialization, and drug stability testing ensure seamless operations, while continuous manufacturing platforms and single-use bioreactor systems offer flexibility in both large-scale fermentation and downstream processing purification.
What are the Key Data Covered in this Pharmaceutical Contract Manufacturing Market Research and Growth Report?
-
What is the expected growth of the Pharmaceutical Contract Manufacturing Market between 2026 and 2030?
-
USD 76.40 billion, at a CAGR of 8.2%
-
-
What segmentation does the market report cover?
-
The report is segmented by End-user (Big pharmaceutical companies, Small and medium-sized pharmaceutical companies, and Generic pharmaceutical companies), Service (API and bulk drug manufacturing, Final dosage form, and Secondary packaging), Application (Infectious diseases, Cardiovascular diseases, and Oncology) and Geography (North America, Asia, Europe, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Asia, Europe and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Increasing complexity and cost of research and development, Heightened regulatory scrutiny and complexity
-
-
Who are the major players in the Pharmaceutical Contract Manufacturing Market?
-
AbbVie Inc., Aenova Holding GmbH, Ajinomoto Co. Inc., Almac Group Ltd., Boehringer Ingelheim GmbH, Cadila Pharmaceuticals Ltd., Cambrex Corp., Catalent Inc., Charles River Laboratories, Cmic Holdings Co. Ltd., Corden Pharma Intl. GmbH, Dr. Reddys Laboratories Ltd., Evonik Industries AG, Lonza Group Ltd., PCI Pharma Services, Recipharm AB, Samsung Biologics Co. Ltd., Siegfried Holding AG, Thermo Fisher Scientific Inc. and WuXi Biologics Cayman Inc.
-
Market Research Insights
- The market dynamics are shaped by a strategic push toward outsourcing, where selecting the right CDMO partnership models is critical for success. Leveraging end-to-end CDMO services can accelerate clinical to commercial manufacturing timelines by up to 30%. The focus on large molecule manufacturing is intensifying, with biopharmaceutical manufacturing capacity being a key selection criterion.
- As companies diversify their API sourcing strategy to mitigate risk, the demand for partners with proven bioprocessing optimization capabilities grows. Adherence to strict aseptic processing standards and the implementation of robust pharmaceutical quality systems (PQS) are non-negotiable, as effective quality risk management (QRM) can reduce compliance-related delays by over 40%.
- The adoption of pharma 4.0 implementation further enhances operational excellence in pharma.
We can help! Our analysts can customize this pharmaceutical contract manufacturing market research report to meet your requirements.

 RIA -
RIA - 
