Cell Therapy Market Size 2026-2030
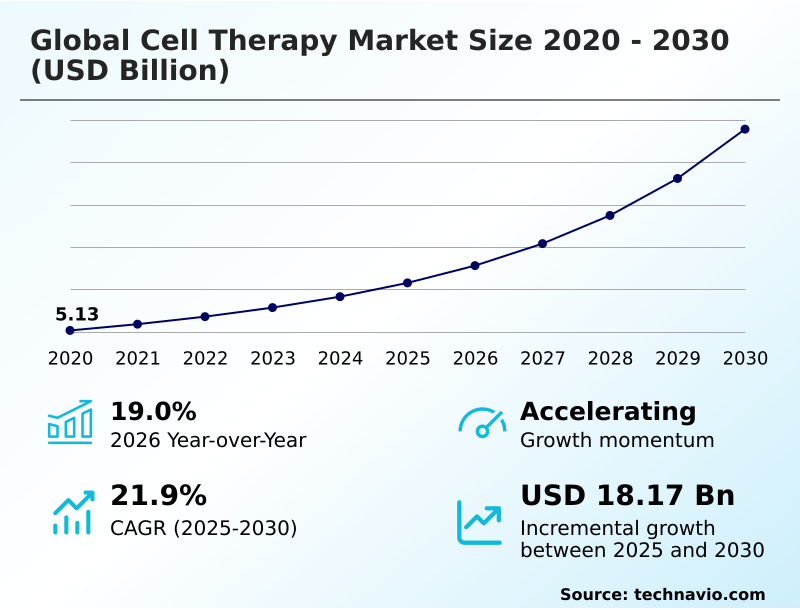
The cell therapy market size is valued to increase by USD 18.17 billion, at a CAGR of 21.9% from 2025 to 2030. Increasing prevalence of chronic and degenerative diseases will drive the cell therapy market.
Major Market Trends & Insights
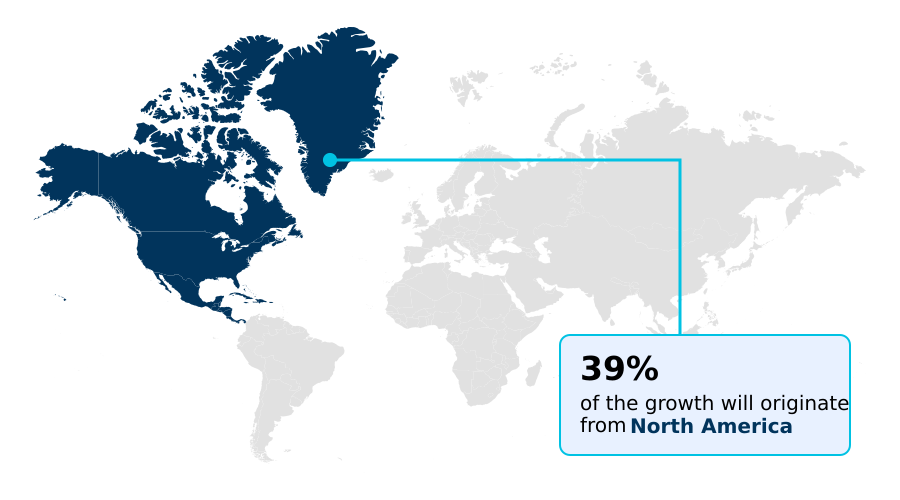
- North America dominated the market and accounted for a 39.3% growth during the forecast period.
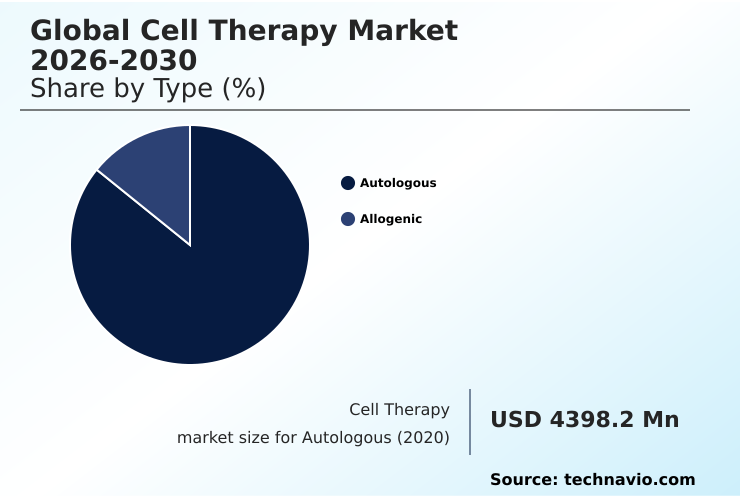
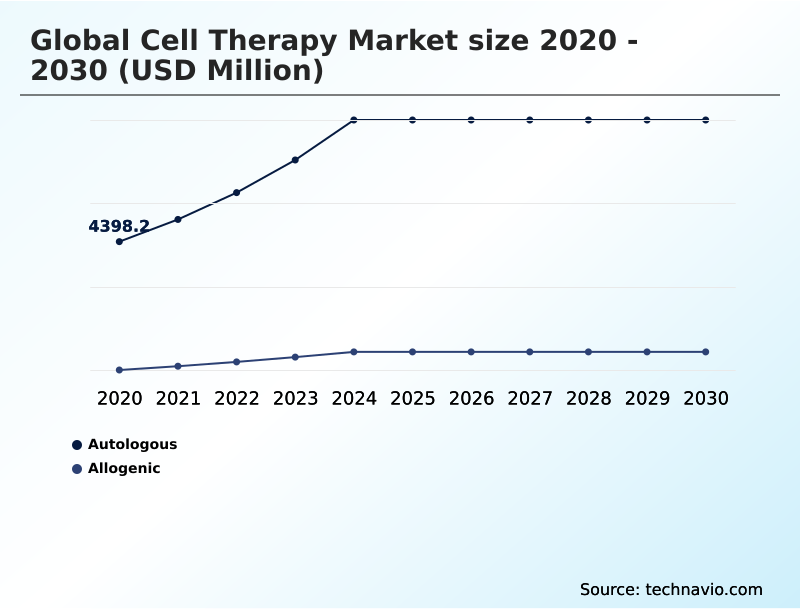
- By Type - Autologous segment was valued at USD 7.88 billion in 2024
- By Application - Malignancies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 23.79 billion
- Market Future Opportunities: USD 18.17 billion
- CAGR from 2025 to 2030 : 21.9%
Market Summary
- The cell therapy market is advancing from niche applications to a pivotal role in mainstream medicine. This evolution is propelled by technological progress in cell engineering and a deeper biological understanding, which together broaden the therapeutic scope from oncology to autoimmune and neurodegenerative conditions.
- A key trend is the industry's strategic pivot from complex autologous models to more scalable allogeneic or 'off-the-shelf' therapies, a move aimed at reducing costs and logistical burdens.
- For instance, a biopharmaceutical firm developing a therapy for a rare genetic disorder must navigate a complex value chain; this involves managing a cryopreserved supply chain where temperature deviations of just a few degrees can compromise a batch, necessitating advanced tracking systems to ensure a 99.9% success rate in maintaining the chain of custody from manufacturing to patient infusion.
- However, significant hurdles remain, including navigating divergent global regulatory pathways and establishing reimbursement models that can accommodate high upfront treatment costs, which are critical for ensuring patient access and sustaining innovation.
What will be the Size of the Cell Therapy Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Cell Therapy Market Segmented?
The cell therapy industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Type
- Autologous
- Allogenic
- Application
- Malignancies
- Musculoskeletal
- Cardiovascular
- Others
- End-user
- Biopharmaceutical companies
- CMOs
- CROs
- Academic and research institutes
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Type Insights
The autologous segment is estimated to witness significant growth during the forecast period.
The autologous segment operates on a highly personalized, one-patient-one-batch model, representing the foundational approach to cell therapy. This method involves harvesting a patient's own cells for subsequent ex vivo cell manipulation and reinfusion.
The primary advantage of autologous cell therapy is the elimination of immunological rejection, but these patient-specific treatment models present significant operational hurdles.
The vein-to-vein time can extend for weeks, and manufacturing success rates can vary by up to 15%, influenced by factors like the quality of a patient's starting biological material.
Maintaining a strict chain of identity under good manufacturing practice conditions is paramount. The process demands rigorous aseptic technique and adherence to strict release criteria, with analytical method validation ensuring product safety and consistency for each individual batch.
The Autologous segment was valued at USD 7.88 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 39.3% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Cell Therapy Market Demand is Rising in North America Request Free Sample
The geographic landscape is dominated by North America, which accounts for 39.3% of incremental growth, driven by a high concentration of research and investment.
However, Asia is forecast to have the highest growth rate, with its market expanding due to government initiatives and a large patient population.
In this region, local firms are advancing capabilities in aseptic processing and bioreactor technology, leading to a 15% reduction in contamination events in some advanced facilities.
European markets are focusing on harmonizing health technology assessment for gene-corrected cell treatments and other advanced therapies.
Across all major regions, there is a focus on refining quality control testing and scaling cell banking operations to support both patient-specific treatment models and allogeneic approaches, with significant research into immunogenicity risk mitigation.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- The future trajectory of the global cell therapy market 2026-2030 is being shaped by several interconnected long-term developments. The key drivers for growth in the global cell therapy market 2026-2030 are clear, but realizing this potential requires overcoming significant hurdles. A critical area is the advancements in CAR-T cell engineering for solid tumors, which involves overcoming the immunosuppressive tumor microenvironment.
- This contrasts with the more established comparison of autologous and allogeneic therapy platforms in hematology. The challenges in scaling autologous cell therapy manufacturing remain a primary concern, directly linked to the strategies for reducing vein-to-vein time in CAR-T. Consequently, the industry is closely watching the regulatory pathways for allogeneic cell therapy approval and the cost-effectiveness analysis of off-the-shelf cell therapies.
- Firms adopting manufacturing automation report up to a 2x improvement in throughput compared to manual processes. The investment trends in the global cell therapy market 2026-2030 reflect a strong interest in gene editing techniques to create universal donor cells, which is central to addressing immunogenicity in allogeneic cell products.
- Success hinges on developing potency assays for mesenchymal stem cells and navigating global regulatory divergence in advanced therapies. The role of CMOs and CROs in cell therapy development has become integral, especially for managing supply chain solutions for cryogenic cell therapy logistics.
- Beyond oncology, the future of cell therapy in treating neurodegenerative disorders and emerging trends in cell therapy for cardiovascular disease represent significant long-term growth frontiers, provided that reimbursement models for high-cost cell and gene therapies can be established.
What are the key market drivers leading to the rise in the adoption of Cell Therapy Industry?
- The increasing prevalence of chronic and degenerative diseases worldwide is a key driver fueling the growth of the cell therapy market.
- The market's expansion is driven by advancements in gene editing and the success of therapies like chimeric antigen receptor (CAR-T) and T-cell receptor therapy.
- These platforms have shown remarkable efficacy in hematological malignancies, with some CAR-T cell therapies for cancer achieving complete response rates over 80% in certain patient populations.
- The focus is now expanding to include TCR therapies for solid tumors and treatments for neurodegenerative disorders. This clinical pipeline expansion is supported by sophisticated regenerative medicine platforms and improved clinical trial logistics.
- Advanced gene editing technologies are also enabling the development of cell therapy for autoimmune diseases and strategies to mitigate risks like host versus graft disease, further broadening the therapeutic potential and attracting significant investment.
What are the market trends shaping the Cell Therapy Industry?
- A growing emphasis on allogenic and 'off-the-shelf' therapies represents a significant upcoming trend. This shift aims to address the scalability and cost challenges of personalized treatments.
- Key industry trends are pivoting toward allogeneic cell therapy and the development of off-the-shelf cell products to overcome logistical challenges. These cell engineering innovations focus on creating universal donor cells and hypoimmunogenic cell lines to minimize rejection.
- A major push involves tackling solid tumors by improving solid tumor infiltration with multi-targeted constructs designed to prevent antigen escape, a common failure mode. Concurrently, manufacturing 4.0 in cell therapy is gaining traction, with firms adopting automated closed-system manufacturing to standardize production, leading to a 20% improvement in process consistency.
- Optimizing bioprocessing parameters is central to these cost of goods reduction strategies, essential for scaling up cell therapy production to meet future demand.
What challenges does the Cell Therapy Industry face during its growth?
- Prohibitive costs and complex reimbursement frameworks pose a key challenge affecting the growth of the cell therapy industry.
- Significant challenges persist, primarily the high costs and navigating complex reimbursement frameworks for cell therapy. Payers are increasingly demanding value-based payment models to justify expenditures. The intricate distribution and supply chain logistics for living drugs require specialized cryogenic storage solutions and flawless execution to maintain product viability. Adherence to strict donor screening protocols is non-negotiable for safety.
- Furthermore, evolving regulatory landscapes create uncertainty, with companies needing to manage disparate regulatory pathways for ATMPs globally. The technical difficulty in establishing standardized potency assays and robust cryopreservation protocols adds another layer of complexity, where inconsistencies can impact product efficacy by up to 10% between different processing methods.
Exclusive Technavio Analysis on Customer Landscape
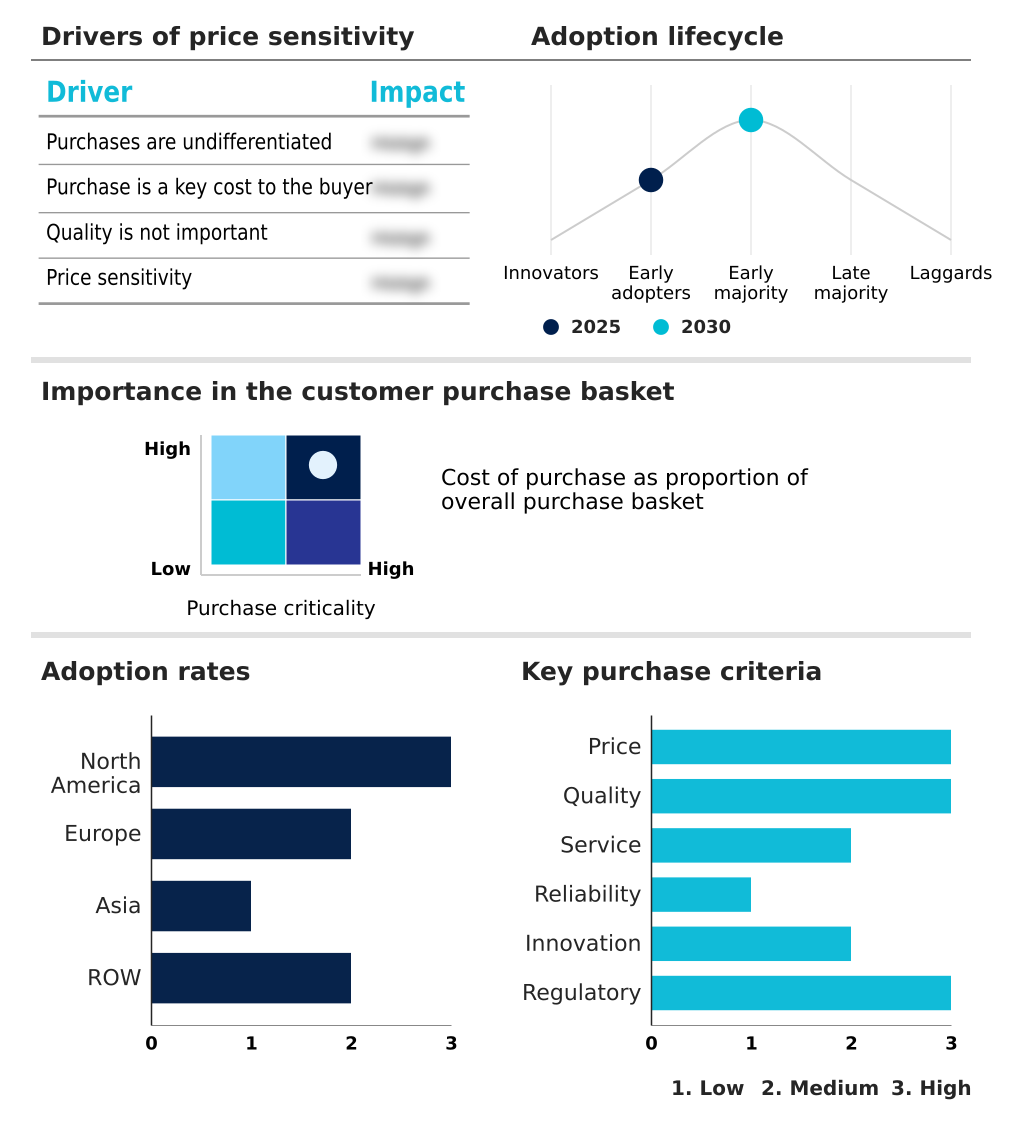
The cell therapy market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the cell therapy market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Cell Therapy Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, cell therapy market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
AbelZeta Pharma Inc. - Developing next-generation CAR-T and TCR-T cell therapies to address solid tumors and hematological cancers.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- AbelZeta Pharma Inc.
- Astellas Pharma Inc.
- Athersys Inc.
- Avita Medical Inc.
- Bayer AG
- Bristol Myers Squibb Co.
- Castle Creek Biosciences Inc.
- Celyad Oncology SA
- CRISPR Therapeutics AG
- Gilead Sciences Inc.
- Johnson and Johnson Services
- Kite Pharma Inc.
- Lineage Cell Therapeutics Inc.
- Novartis AG
- Pharmicell Co. Ltd.
- Sanpower Group Co. Ltd.
- Sarepta Therapeutics Inc.
- Vericel Corp.
- Vertex Pharmaceuticals Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Cell therapy market
- In October, 2025, Bristol Myers Squibb announced its acquisition of Orbital Therapeutics, gaining access to a next-generation RNA platform and an investigational in vivo CAR T-cell therapy.
- In November, 2025, Aspen Neuroscience secured $115 million in a Series C financing round to advance clinical development of its autologous stem cell-derived therapy for Parkinson disease.
- In April, 2025, Abeona Therapeutics received United States Food and Drug Administration (FDA) approval for Zevaskyn, a cell-based gene therapy for recessive dystrophic epidermolysis bullosa (RDEB).
- In June, 2025, Vertex Pharmaceuticals presented clinical trial data for zimislecel (VX-880), showing that a majority of participants with type 1 diabetes no longer required daily insulin.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Cell Therapy Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 293 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 21.9% |
| Market growth 2026-2030 | USD 18168.0 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 19.0% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, South Korea, Indonesia, Thailand, Brazil, Saudi Arabia, UAE, Turkey, Argentina, Colombia, South Africa and Israel |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The market's foundation is built upon complex science, from autologous cell therapy and allogeneic cell therapy to the intricacies of chimeric antigen receptor and T-cell receptor therapy. Central to development are induced pluripotent stem cells and mesenchymal stem cells, modified using advanced gene editing technologies.
- A key boardroom consideration is the investment in bioprocessing parameters and bioreactor technology to standardize ex vivo cell manipulation. The transition to automated closed-system manufacturing is critical for maintaining good manufacturing practice standards and ensuring aseptic processing. For autologous treatments, managing the vein-to-vein time is a core operational metric, with integrated digital platforms reducing logistical delays by up to 20%.
- The scientific challenge includes overcoming the immunosuppressive microenvironment to improve solid tumor infiltration and preventing antigen escape with multi-targeted constructs. Success requires validated potency assays, robust cryopreservation protocols, and stringent quality control testing against clear release criteria to ensure the safety and efficacy of treatments for conditions ranging from hematological malignancies to recessive dystrophic epidermolysis bullosa.
What are the Key Data Covered in this Cell Therapy Market Research and Growth Report?
-
What is the expected growth of the Cell Therapy Market between 2026 and 2030?
-
USD 18.17 billion, at a CAGR of 21.9%
-
-
What segmentation does the market report cover?
-
The report is segmented by Type (Autologous, and Allogenic), Application (Malignancies, Musculoskeletal, Cardiovascular, and Others), End-user (Biopharmaceutical companies, CMOs, CROs, and Academic and research institutes) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Increasing prevalence of chronic and degenerative diseases, Prohibitive costs and complex reimbursement frameworks
-
-
Who are the major players in the Cell Therapy Market?
-
AbelZeta Pharma Inc., Astellas Pharma Inc., Athersys Inc., Avita Medical Inc., Bayer AG, Bristol Myers Squibb Co., Castle Creek Biosciences Inc., Celyad Oncology SA, CRISPR Therapeutics AG, Gilead Sciences Inc., Johnson and Johnson Services, Kite Pharma Inc., Lineage Cell Therapeutics Inc., Novartis AG, Pharmicell Co. Ltd., Sanpower Group Co. Ltd., Sarepta Therapeutics Inc., Vericel Corp. and Vertex Pharmaceuticals Inc.
-
Market Research Insights
- The market's dynamic is shaped by a strategic push toward scaling up cell therapy production, driven by cell engineering innovations. This involves a clear distinction in autologous vs allogeneic therapy approaches, with a focus on off-the-shelf cell products and the creation of universal donor cells.
- Key regenerative medicine platforms are enabling new CAR-T cell therapies for cancer and expanding into cell therapy for autoimmune diseases. Managing supply chain logistics for living drugs requires advanced cryogenic storage solutions and strict aseptic technique.
- Navigating regulatory pathways for ATMPs and varied reimbursement frameworks for cell therapy is critical, with firms showing a 25% greater success rate when engaging regulators early. Adopting digital tools for bioprocessing and robust analytical method validation improves consistency, while effective donor screening protocols and cell culture media optimization are fundamental to safety and efficacy.
We can help! Our analysts can customize this cell therapy market research report to meet your requirements.

 RIA -
RIA - 
