Ai-driven Drug Safety And Pharmacovigilance Platforms Market Size 2026-2030
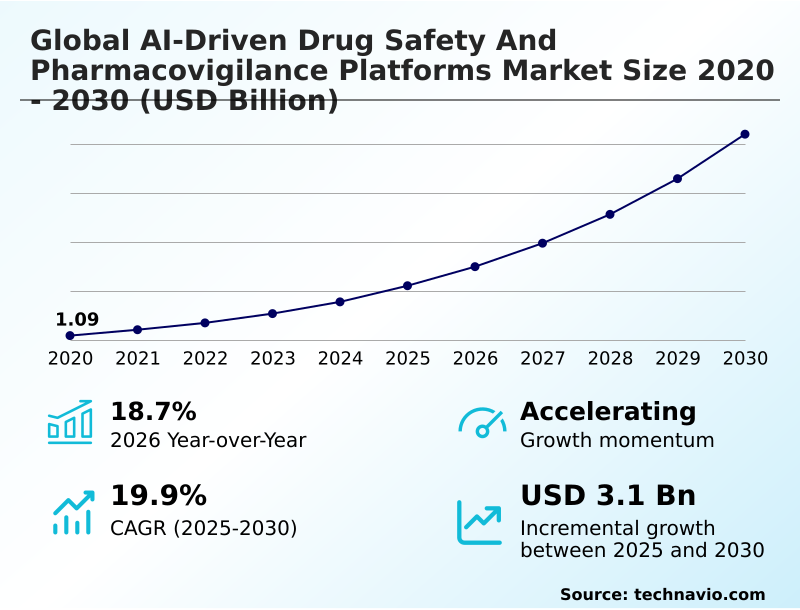
The ai-driven drug safety and pharmacovigilance platforms market size is valued to increase by USD 3.10 billion, at a CAGR of 19.9% from 2025 to 2030. Increasing volume and complexity of adverse event data will drive the ai-driven drug safety and pharmacovigilance platforms market.
Major Market Trends & Insights
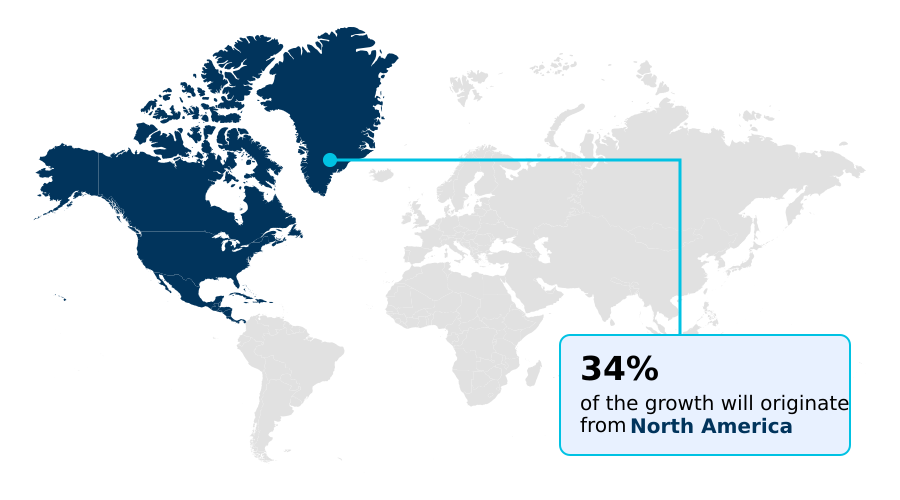
- North America dominated the market and accounted for a 33.6% growth during the forecast period.
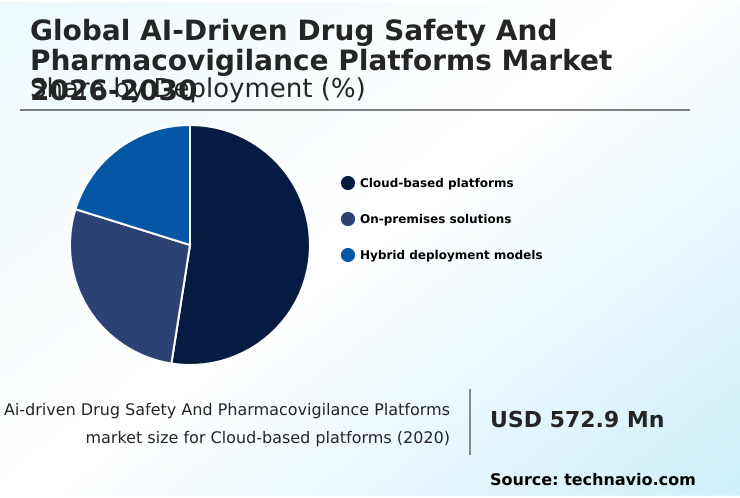
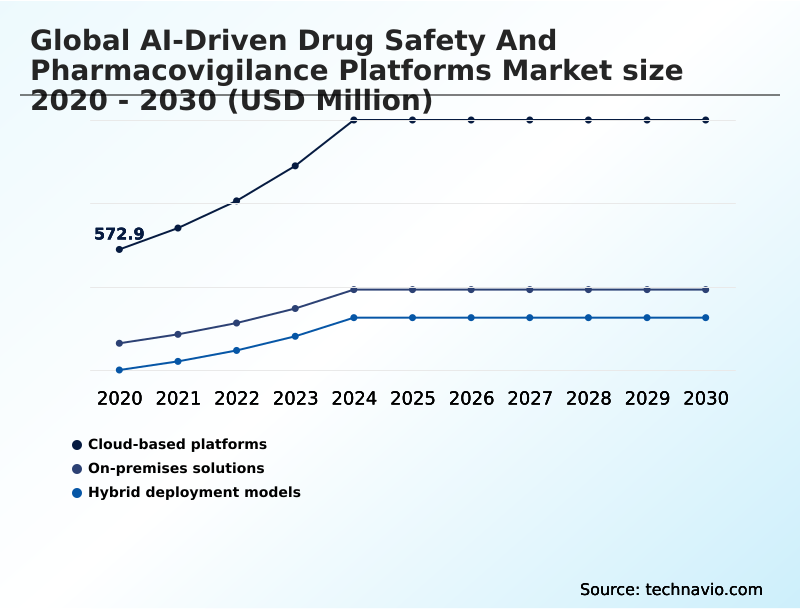
- By Deployment - Cloud-based platforms segment was valued at USD 951.5 million in 2024
- By End-user - Pharma and biotech companies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 4.12 billion
- Market Future Opportunities: USD 3.10 billion
- CAGR from 2025 to 2030 : 19.9%
Market Summary
- The AI-driven drug safety and pharmacovigilance platforms market is undergoing a significant transformation, moving beyond reactive compliance to proactive, predictive safety management. This shift is fueled by the need to process vast volumes of unstructured data from sources like electronic health records, social media, and wearables.
- Advanced platforms leverage automated case processing and natural language processing for safety to automate routine tasks, allowing pharmacovigilance teams to focus on strategic analysis and risk mitigation. For instance, a pharmaceutical company launching a new therapy can deploy machine learning signal detection models to monitor real-world evidence in near real-time.
- This capability enables the rapid identification of potential adverse drug reaction monitoring, ensuring quicker updates to safety protocols and enhancing patient outcomes. The adoption of such intelligent pharmacovigilance systems not only ensures regulatory compliance but also builds a more resilient and efficient drug safety ecosystem, ultimately driving better public health protection through robust pharmacovigilance technology solutions and adverse event monitoring.
What will be the Size of the Ai-driven Drug Safety And Pharmacovigilance Platforms Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Ai-driven Drug Safety And Pharmacovigilance Platforms Market Segmented?
The ai-driven drug safety and pharmacovigilance platforms industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Deployment
- Cloud-based platforms
- On-premises solutions
- Hybrid deployment models
- End-user
- Pharma and biotech companies
- Contract research organizations
- Regulatory authorities
- Healthcare providers
- Application
- Adverse event detection
- Risk management
- Literature screening
- Regulatory compliance
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- APAC
- China
- India
- Japan
- Middle East and Africa
- Saudi Arabia
- UAE
- Israel
- South America
- Brazil
- Argentina
- Colombia
- Rest of World (ROW)
- North America
By Deployment Insights
The cloud-based platforms segment is estimated to witness significant growth during the forecast period.
Cloud-based platforms are reshaping pharmacovigilance through scalable, flexible software-as-a-service models. This deployment minimizes on-site hardware and maintenance, enabling GxP compliant cloud platforms to deliver continuous updates and refinements to machine learning signal detection algorithms.
Organizations benefit from a unified, accessible environment for global safety teams, improving data processing efficiency by up to 40%.
The model facilitates seamless real-world evidence integration and supports AI-driven benefit-risk assessment by providing the computational power needed for complex AI-powered literature screening.
This approach, centered on pharmacovigilance process optimization, shifts spending from capital to operational expenditures, making advanced pharmacovigilance automation technologies accessible to a broader range of organizations. The focus on AI for regulatory submissions and intelligent automation in pharmacovigilance is paramount.
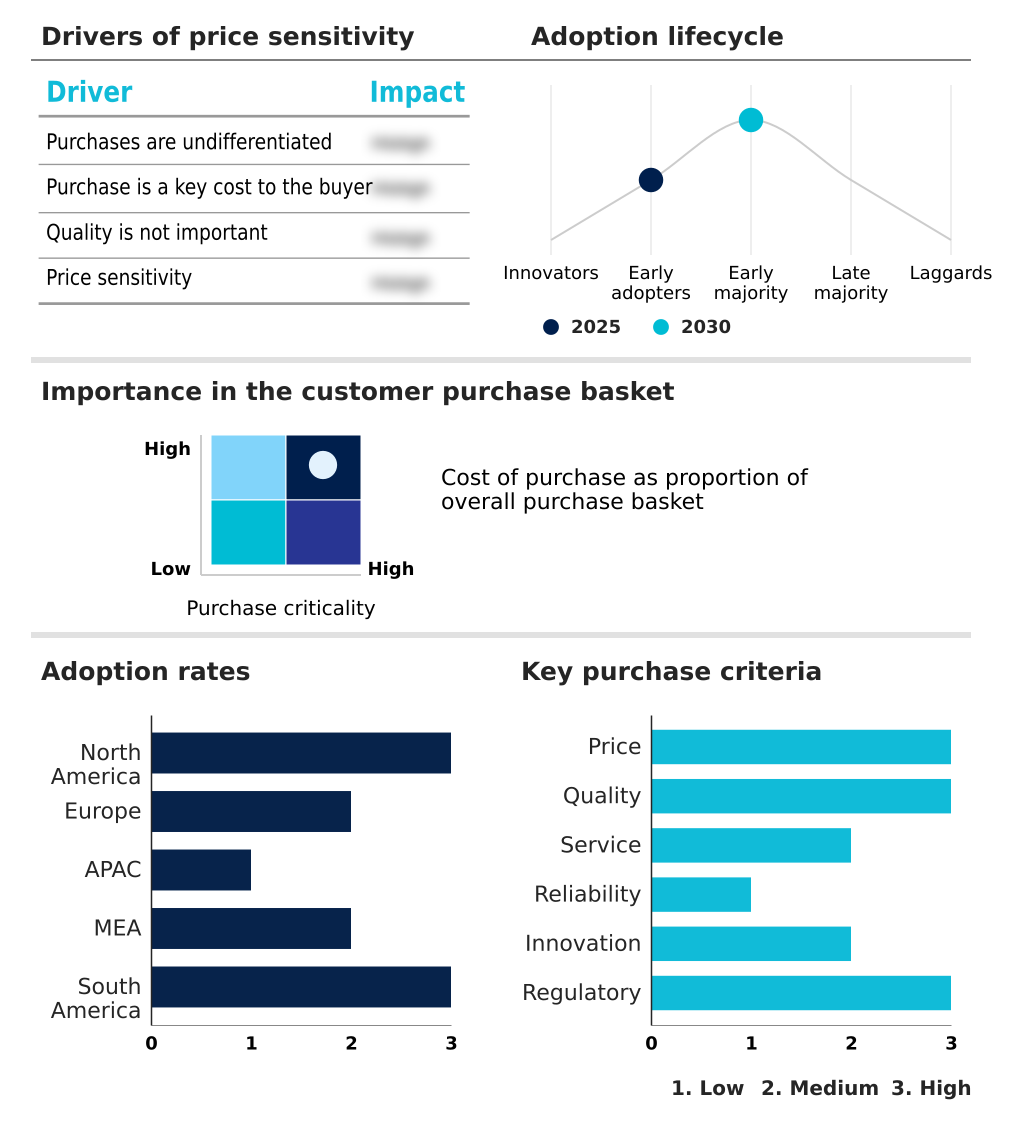
The Cloud-based platforms segment was valued at USD 951.5 million in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 33.6% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Ai-driven Drug Safety And Pharmacovigilance Platforms Market Demand is Rising in North America Request Free Sample
The geographic landscape of the AI-driven drug safety market is led by North America, which accounts for over 33% of incremental growth due to its advanced pharmaceutical ecosystem and early adoption of unified safety platforms.
This region leverages AI-enhanced signal management to meet stringent regulatory demands. Meanwhile, APAC is the fastest-growing region, driven by the expansion of pharmaceutical manufacturing and digitalization.
Europe maintains a strong position, with a focus on data privacy and the validation of adverse event detection algorithms. Firms in mature markets utilize AI-based safety signal analysis to reduce manual literature review times by over 50%.
The adoption of pharmacovigilance reporting tools and adverse event database management is becoming standard.
These regional dynamics highlight a global shift toward automated, data-centric safety surveillance and pharmacovigilance outsourcing services to manage complex requirements, with pharmacovigilance service providers playing a key role.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Understanding the role of AI in the pharmacovigilance process is critical for life sciences organizations aiming to enhance efficiency and compliance. The evolution toward automating adverse event case processing is driven by the need to manage immense data volumes while ensuring accuracy. The benefits of cloud-based pharmacovigilance platforms are significant, offering scalability and accessibility that on-premise solutions cannot match.
- However, the challenges of AI in drug safety, particularly around model validation and explainable AI for drug safety assessment, require careful consideration. Integrating real-world evidence in pharmacovigilance is no longer optional but a necessity for comprehensive post-market surveillance.
- Organizations successfully implementing generative AI for safety narrative generation have reported efficiency gains nearly double those of peers who only partially automate their workflows. This demonstrates the profound impact of AI on pharmacovigilance workflows. The future of AI in pharmacovigilance lies in creating interconnected systems that improve regulatory compliance with AI and enhance patient safety with predictive analytics.
- AI platforms for contract research organizations are also becoming essential, enabling them to offer more competitive services. As the industry overcomes data silos in pharmacovigilance and advances machine learning models for signal detection, the cost-effectiveness of automated pharmacovigilance systems becomes increasingly clear. This transition supports better AI applications in post-market surveillance and strengthens the entire drug safety lifecycle.
What are the key market drivers leading to the rise in the adoption of Ai-driven Drug Safety And Pharmacovigilance Platforms Industry?
- The increasing volume and complexity of adverse event data from diverse sources is a key driver for the market.
- The market's growth is propelled by several key drivers, including the imperative for operational efficiency. The use of automated case processing and other pharmacovigilance automation technologies reduces the cost per case for adverse event processing by an average of 35%.
- Stringent global regulatory requirements are another major factor, pushing firms to adopt technologies that ensure high-quality safety data and auditable pharmacovigilance data processing. These AI-driven platforms help companies meet strict reporting deadlines with over 99% accuracy.
- The increasing volume and complexity of adverse event data from sources like social media and wearables make manual analysis untenable, necessitating advanced safety data analysis tools and intelligent pharmacovigilance systems.
- The adoption of these tools is crucial for maintaining a proactive approach to drug safety.
What are the market trends shaping the Ai-driven Drug Safety And Pharmacovigilance Platforms Industry?
- The integration of generative artificial intelligence for automated medical narrative generation is an emerging market trend. This development aims to accelerate compliant safety reporting and enhance standardization.
- Key trends are reshaping the market, led by the adoption of generative AI in life sciences and proactive safety surveillance. The move toward cloud-native and modular SaaS platforms enables greater agility, allowing firms to adopt specific functionalities like AI-enhanced signal management as needed.
- This approach offers significant efficiency gains, with the use of generative AI narrative generation shown to reduce safety reporting cycle times by over 30%. Furthermore, the transition to proactive monitoring using real-world evidence has improved early safety signal detection by 25% compared to legacy methods.
- The focus on pharmacovigilance system validation ensures these AI-driven drug safety and pharmacovigilance platforms meet rigorous regulatory standards, while adverse reaction monitoring software becomes more sophisticated. The market is also seeing growth in adverse event monitoring and AI for drug development safety, enhancing safety throughout the product lifecycle.
What challenges does the Ai-driven Drug Safety And Pharmacovigilance Platforms Industry face during its growth?
- The opacity and interpretability barriers of complex algorithms present a key challenge affecting industry growth.
- Despite strong drivers, the market faces significant challenges, including the need for specialized technical and domain expertise. The issue of algorithmic opacity creates an interpretability barrier, as the 'black box' nature of some machine learning for drug safety models complicates validation. This lack of transparency can require up to a 20% increase in manual verification for high-risk signals.
- Furthermore, ensuring data interoperability and integration with legacy systems remains a primary hurdle. The process of mapping inconsistent data to a centralized pharmacovigilance technology solution can account for 60% of an implementation timeline. Overcoming these challenges is essential for realizing the full potential of AI-powered signal management and adverse event case intake.
- The industry must balance innovation with the need for trust and reliability in its safety signal analysis and drug safety monitoring solutions.
Exclusive Technavio Analysis on Customer Landscape
The ai-driven drug safety and pharmacovigilance platforms market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the ai-driven drug safety and pharmacovigilance platforms market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Ai-driven Drug Safety And Pharmacovigilance Platforms Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, ai-driven drug safety and pharmacovigilance platforms market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Accenture Plc - Delivers end-to-end patient safety operations through AI-driven platforms for automated case processing, signal management, and analytics-driven pharmacovigilance workflows.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Accenture Plc
- Anju Software Inc.
- ArisGlobal LLC
- Certara Inc.
- Clarivate PLC
- ClinChoice
- Cognizant Technology Solution
- Ennov SAS
- EXTEDO GmbH
- Genpact Ltd.
- HCL Technologies Ltd.
- Infosys Ltd.
- IQVIA Holdings Inc.
- Medidata Solutions Inc
- Navitas Life Sciences
- Oracle Corp.
- Saama Technologies Inc.
- Sorcero Inc.
- Tata Consultancy Services
- Veeva Systems Inc.
- WCG Clinical Inc.
- Wipro Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Ai-driven drug safety and pharmacovigilance platforms market
- In February 2025, ArisGlobal announced the full integration of a generative AI framework within its LifeSphere safety suite to automate the creation of clinical safety narratives.
- In March 2025, Oracle Health Sciences released an updated version of its safety cloud platform that utilizes autonomous database technology to provide real-time signal detection across diverse patient populations.
- In April 2025, IQVIA launched an enhanced vigilance platform that incorporates advanced natural language processing to analyze unstructured data from medical literature and digital patient forums.
- In April 2025, Google launched a specialized cloud-based suite for life sciences organizations to automate the extraction of safety data from unstructured patient narratives.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Ai-driven Drug Safety And Pharmacovigilance Platforms Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 309 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 19.9% |
| Market growth 2026-2030 | USD 3103.6 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 18.7% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, Australia, South Korea, Indonesia, Saudi Arabia, UAE, Israel, South Africa, Turkey, Brazil, Argentina and Colombia |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The AI-driven drug safety and pharmacovigilance platforms market is fundamentally shifting from reactive reporting to proactive, predictive safety intelligence. This evolution is driven by the strategic integration of technologies that enable continuous post-market safety surveillance and real-time analysis.
- A key trend, real-world evidence integration, directly influences boardroom decisions regarding risk management and R&D resource allocation, as it provides a clearer picture of a drug's performance in diverse populations. Leading firms leveraging predictive safety analytics and pharmacovigilance workflow automation have achieved a 30% reduction in the time required to validate new safety signals.
- This efficiency is critical in a tightly regulated environment. The implementation of end-to-end patient safety systems, supported by automated case processing and AI-driven benefit-risk assessment, is becoming a competitive differentiator. These platforms, which utilize structured safety data formats and pharmacovigilance compliance tools, are essential for managing the entire drug lifecycle safety and ensuring robust regulatory submission automation.
What are the Key Data Covered in this Ai-driven Drug Safety And Pharmacovigilance Platforms Market Research and Growth Report?
-
What is the expected growth of the Ai-driven Drug Safety And Pharmacovigilance Platforms Market between 2026 and 2030?
-
USD 3.10 billion, at a CAGR of 19.9%
-
-
What segmentation does the market report cover?
-
The report is segmented by Deployment (Cloud-based platforms, On-premises solutions, and Hybrid deployment models), End-user (Pharma and biotech companies, Contract research organizations, Regulatory authorities, and Healthcare providers), Application (Adverse event detection, Risk management, Literature screening, and Regulatory compliance) and Geography (North America, Europe, APAC, Middle East and Africa, South America)
-
-
Which regions are analyzed in the report?
-
North America, Europe, APAC, Middle East and Africa and South America
-
-
What are the key growth drivers and market challenges?
-
Increasing volume and complexity of adverse event data, Algorithmic opacity and interpretability barrier
-
-
Who are the major players in the Ai-driven Drug Safety And Pharmacovigilance Platforms Market?
-
Accenture Plc, Anju Software Inc., ArisGlobal LLC, Certara Inc., Clarivate PLC, ClinChoice, Cognizant Technology Solution, Ennov SAS, EXTEDO GmbH, Genpact Ltd., HCL Technologies Ltd., Infosys Ltd., IQVIA Holdings Inc., Medidata Solutions Inc, Navitas Life Sciences, Oracle Corp., Saama Technologies Inc., Sorcero Inc., Tata Consultancy Services, Veeva Systems Inc., WCG Clinical Inc. and Wipro Ltd.
-
Market Research Insights
- The market's dynamics are increasingly shaped by the adoption of sophisticated pharmacovigilance analytics and cloud pharmacovigilance platforms. These technologies are central to improving drug safety automation and operational efficiency. Firms leveraging adverse event reporting systems powered by AI have reported up to a 40% reduction in manual case processing times, directly impacting operational costs.
- Furthermore, the use of advanced pharmacovigilance trends and analytics for signal detection in pharmacovigilance has demonstrated a 15% improvement in identifying true safety signals compared to traditional statistical methods. This enhancement in accuracy is critical for mitigating risk and ensuring patient safety.
- The integration of real-world data for pharmacovigilance into these platforms provides a more comprehensive view of a drug's performance, solidifying the move toward proactive, data-driven safety monitoring. This shift is crucial for companies seeking a competitive edge in a highly regulated industry.
We can help! Our analysts can customize this ai-driven drug safety and pharmacovigilance platforms market research report to meet your requirements.


 RIA -
RIA - 
