Biosimilars Market Size 2026-2030
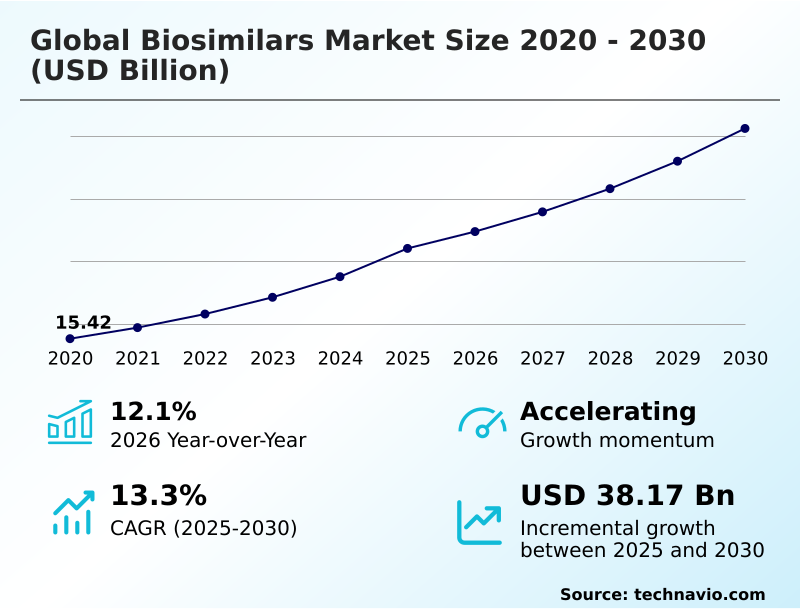
The biosimilars market size is valued to increase by USD 38.17 billion, at a CAGR of 13.3% from 2025 to 2030. Increasing patent expirations and intensifying focus on cost-effectiveness will drive the biosimilars market.
Major Market Trends & Insights
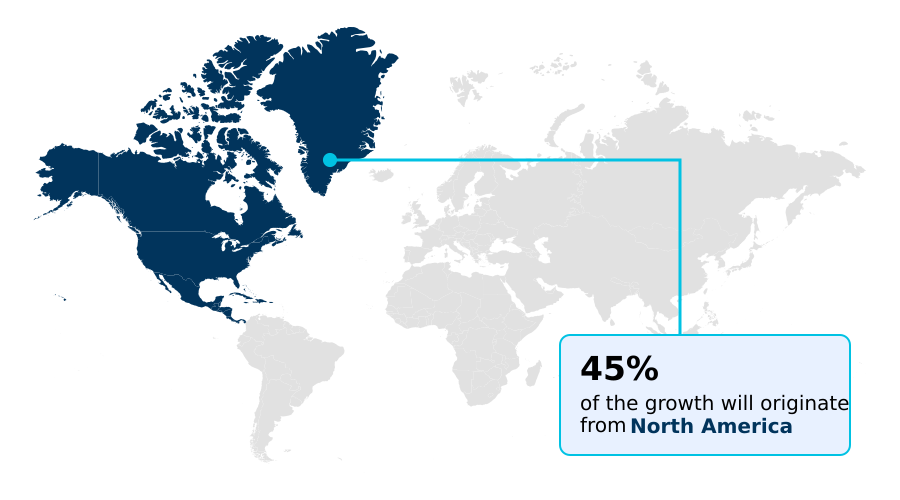
- North America dominated the market and accounted for a 45% growth during the forecast period.
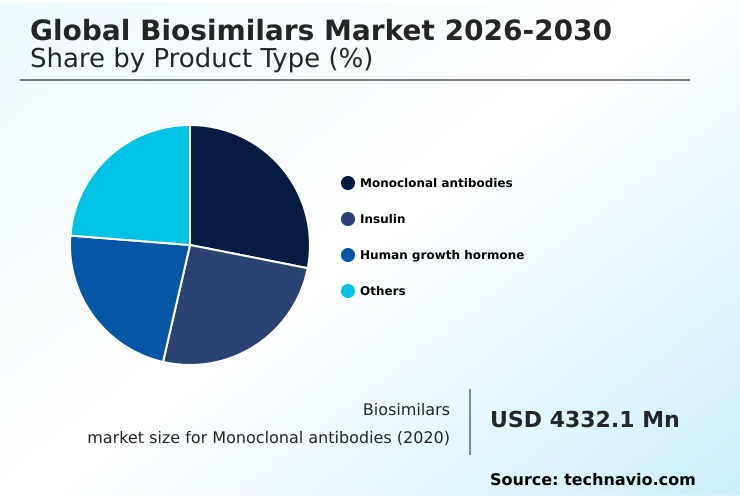
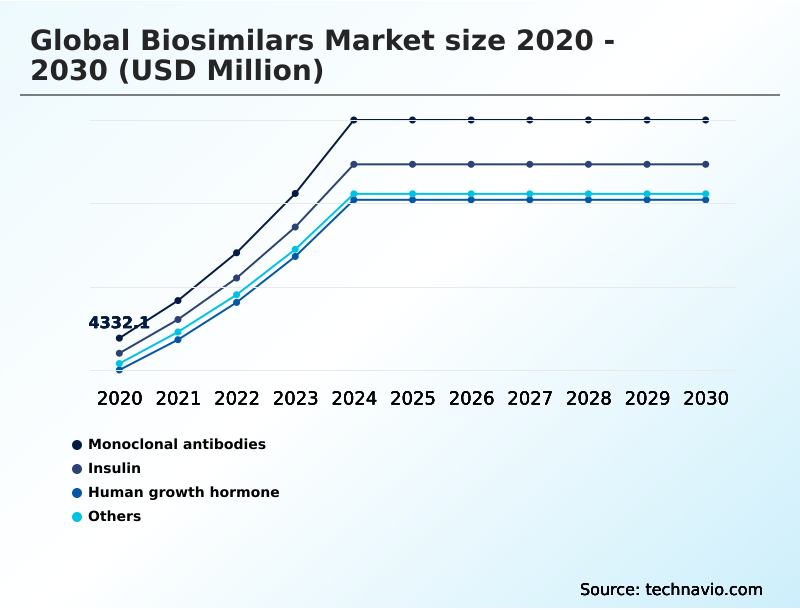
- By Product Type - Monoclonal antibodies segment was valued at USD 10.10 billion in 2024
- By Application - Oncology and hematology segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 66.90 billion
- Market Future Opportunities: USD 38.17 billion
- CAGR from 2025 to 2030 : 13.3%
Market Summary
- The biosimilars market is defined by the critical need to balance scientific rigor with commercial viability. The primary driver remains the expiration of patents on high-revenue originator biologics, which opens the door for more affordable yet therapeutically equivalent alternatives.
- This dynamic is fostering intense competition and pushing manufacturers toward strategic innovations, such as developing products with interchangeability designations to gain a competitive edge at the pharmacy level. However, the path to market is fraught with challenges, including navigating complex patent thickets and overcoming lingering physician and patient reluctance.
- For instance, a biopharmaceutical firm must leverage advanced analytical characterization and robust clinical data to create a compelling totality-of-the-evidence package for regulatory submission. Successfully demonstrating indistinguishable pharmacokinetic and pharmacodynamic profiles is not merely a scientific hurdle but a crucial commercial step.
- This rigorous validation process, essential for gaining prescriber confidence, directly impacts market penetration and the realization of cost savings for healthcare systems. As the industry matures, the focus is shifting from simple replication to creating bio-betters and expanding into new therapeutic areas, signaling a more sophisticated and value-driven future.
What will be the Size of the Biosimilars Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Get Free Sample
How is the Biosimilars Market Segmented?
The biosimilars industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Product type
- Monoclonal antibodies
- Insulin
- Human growth hormone
- Others
- Application
- Oncology and hematology
- Endocrinology
- Immunology
- Nephrology
- Manufacturing type
- In-house manufacturing
- Contract manufacturing
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Product Type Insights
The monoclonal antibodies segment is estimated to witness significant growth during the forecast period.
The monoclonal antibodies segment is a cornerstone of the biosimilars market, driven by patent expirations of high-revenue biologic drugs. These therapies, essential for oncology and hematology treatments and immunology autoimmune therapies, are subject to intense development focus.
Innovations in peptide mapping and protein aggregation analysis are crucial for demonstrating biosimilarity. Regulatory shifts, such as the increased reliance on a totality-of-the-evidence approach, have streamlined development, with some programs reporting a 20% reduction in pre-clinical validation timelines.
This evolution allows for faster market entry of cost-effective therapeutic alternatives. The emphasis on high-concentration formulations and advanced drug delivery systems further shapes the competitive dynamics within this critical market segment, supporting broader patient access expansion and public health sustainability.
The Monoclonal antibodies segment was valued at USD 10.10 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
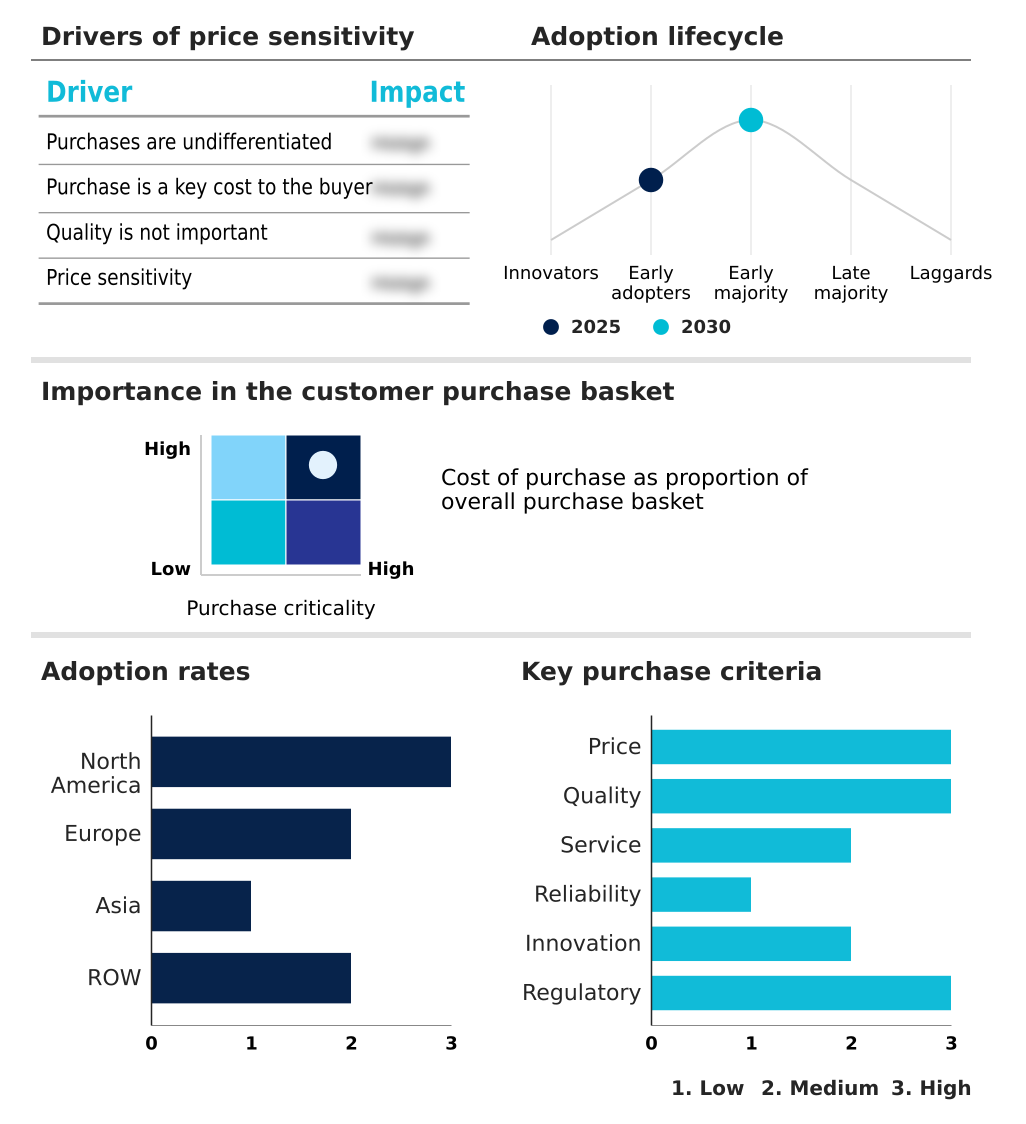
North America is estimated to contribute 45% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Biosimilars Market Demand is Rising in North America Get Free Sample
The geographic landscape of the market is diverse, with North America, Europe, and Asia representing distinct stages of market maturity.
North America leads in revenue, where regulatory advancements under the Biologics Price Competition and Innovation Act (BPCIA) have supported a steady influx of new products.
Europe is a mature market characterized by high biosimilar uptake, with some nations achieving over 80% market share for certain molecules due to established tender systems.
Asia is emerging as a critical growth hub for both consumption and manufacturing, with regional players leveraging advanced biomanufacturing technologies to produce biologics.
Government support for local production in countries like India and South Korea has helped reduce manufacturing costs by up to 25%, bolstering both domestic supply and export capabilities for off-patent biological medicines.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the biosimilars market requires a deep understanding of specific therapeutic and regulatory nuances. For instance, developing a monoclonal antibody biosimilar for oncology demands a different clinical and commercialization strategy than an interchangeable insulin glargine biosimilar, where pharmacy-level substitution is key.
- The denosumab biosimilar litigation impact analysis highlights the significant risk posed by legal challenges, which can delay market entry. Similarly, the successful launch of an adalimumab biosimilar high-concentration formulation depends on navigating a competitive landscape dominated by an established originator.
- In specialized fields, bringing an aflibercept biosimilar for macular degeneration or a ustekinumab biosimilar for Crohn's disease to market involves engaging with specific physician groups and patient advocates. Supportive care products, such as a filgrastim biosimilar for neutropenia management or an epoetin alfa biosimilar in chronic kidney disease, have different reimbursement dynamics.
- The industry is also focused on streamlining regulatory pathways for biosimilars to reduce development timelines. Firms must decide between leveraging the role of CDMOs in biosimilar manufacturing or pursuing the vertical integration benefits for biosimilar producers to control costs. Successfully navigating patent thickets for biologic drugs and boosting physician confidence in biosimilar adoption are critical for success.
- The ultimate goal is realizing significant cost savings from biosimilar substitution, where the impact of interchangeability on market uptake cannot be overstated. Companies must weigh a biobetter development versus a biosimilar strategy, considering the potential for biosimilar expansion in emerging markets.
- Advanced manufacturing for biologic production and the global harmonization of biosimilar regulations are essential for ensuring supply chain security for biologic medicines, with integrated supply chains showing up to 15% greater resilience to disruptions.
What are the key market drivers leading to the rise in the adoption of Biosimilars Industry?
- The market's growth is primarily driven by the increasing number of patent expirations for major biologics, coupled with an intensified focus on cost-effectiveness within global healthcare systems.
- Market growth is fundamentally driven by the continuing wave of patent expirations for major biologics, which enables the introduction of cost-effective therapeutic alternatives.
- This focus on healthcare expenditure reduction is a powerful incentive for payers, with biosimilar adoption capable of lowering treatment costs by over 35% in competitive therapeutic classes.
- The rising global incidence of chronic conditions further fuels demand for sustainable solutions in chronic disease management and nephrology patient care. Concurrently, the refinement of regulatory frameworks, including the totality-of-the-evidence approach, is streamlining development without compromising on immunogenicity assessment.
- These clearer regulatory submission pathways, combined with advancements in biomanufacturing, are lowering entry barriers and fostering a more dynamic and competitive market.
What are the market trends shaping the Biosimilars Industry?
- A key market trend is the strategic shift from standard biosimilar development to the pursuit of interchangeability designations. Concurrently, there is a growing focus on creating bio-betters, which offer enhanced therapeutic performance over existing biologics.
- Key market trends reflect a strategic evolution beyond direct replication toward value-added differentiation. A primary focus is on securing an interchangeability designation, a regulatory status that can boost market penetration by over 30% in its first year by enabling pharmacy-level substitution. Concurrently, some firms are pursuing biobetters development, creating enhanced versions of existing biologics.
- There is also a notable expansion into new therapeutic areas, including ophthalmology and neurology, diversifying from the initial focus on oncology and immunology. This is complemented by a move toward vertical integration and the adoption of advanced biomanufacturing technologies.
- In-house control over production can shorten development-to-market timelines by up to 20%, providing a significant competitive advantage and enhancing biopharmaceutical supply chain security.
What challenges does the Biosimilars Industry face during its growth?
- A key challenge affecting industry growth is the need for manufacturers to navigate complex and evolving regulatory frameworks across different global jurisdictions.
- Despite strong growth drivers, the market faces significant challenges that can impede progress. Aggressive legal strategies from originator companies, including the creation of patent thickets, present a persistent intellectual property litigation risk that can delay market entry by several years. The lack of complete regulatory framework harmonization across key global regions creates complexity and increases development costs.
- Another critical hurdle is overcoming physician and patient adoption hesitancy, which is often rooted in a lack of understanding of the rigorous science behind therapeutic equivalence demonstration. In some specialized fields, biosimilar uptake remains 40% lower than in primary care settings, highlighting the ongoing need for targeted education to build trust and ensure market access strategies are effective.
Exclusive Technavio Analysis on Customer Landscape
The biosimilars market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the biosimilars market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Biosimilars Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, biosimilars market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Abbott Laboratories - The firm’s offerings center on producing high-quality, cost-effective biologic alternatives to expand patient access in critical therapeutic areas through advanced scientific development and manufacturing.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Abbott Laboratories
- Biocon Ltd.
- Dr. Reddys Laboratories Ltd.
- Fresenius Kabi AG
- Intas Pharmaceuticals Ltd.
- Kashiv Biosciences LLC
- Mabxience Holding S.L.
- NeuClone Pty Ltd.
- Organon and Co.
- Pfizer Inc.
- Reliance Life Sciences Pvt. Ltd.
- Samsung Bioepis Co. Ltd.
- Sandoz Group AG
- STADA Arzneimittel AG
- Teva Pharmaceutical Ltd.
- Viatris Inc.
- Zentiva Group
- Zydus Lifesciences Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Biosimilars market
- In May 2025, Formycon and its partner Fresenius Kabi announced that their ustekinumab biosimilar, Otulfi (ustekinumab-aauz), was designated as interchangeable by the US Food and Drug Administration, allowing for substitution at the pharmacy level.
- In April 2025, Boehringer Ingelheim announced it had received the first interchangeable designation for its adalimumab biosimilar, Cyltezo, in a high-concentration formulation, directly challenging the market-dominant version of Humira.
- In April 2025, Biocon Biologics Ltd. announced that the US Food and Drug Administration approved Johevne (bevacizumab-nila), a biosimilar referencing Avastin, for treating various cancers including colorectal, lung, and certain brain tumors.
- In January 2025, Amgen initiated the US launch of Wezlana (ustekinumab-auub), the first biosimilar to reference Stelara, providing a lower-cost biologic option for patients with conditions such as Crohn's disease and plaque psoriasis.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Biosimilars Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 299 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 13.3% |
| Market growth 2026-2030 | USD 38171.7 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 12.1% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, South Korea, Thailand, Indonesia, Brazil, Saudi Arabia, UAE, Turkey, Argentina, South Africa, Colombia and Israel |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The biosimilars market is predicated on delivering high-quality biologic medical products as alternatives to established reference biologic drugs. Successful market entry hinges on meticulous analytical characterization, including peptide mapping and analysis of post-translational modifications, to prove similarity.
- The industry is witnessing a strategic shift toward vertical integration in biopharma, a boardroom-level decision involving significant capex to gain control over the entire production process, from cell line development and upstream fermentation to downstream purification and final fill-finish operations.
- This strategy, coupled with the adoption of process analytical technology (PAT), has enabled some manufacturers to improve batch-to-batch consistency by over 30%. Development pipelines are increasingly focused on complex molecules like monoclonal antibodies, insulin analogs, and human growth hormone (somatropin), alongside supportive care biologics such as granulocyte colony-stimulating factors (G-CSF) and erythropoiesis-stimulating agents (ESAs).
- Adherence to good manufacturing practice (gmp) compliance is non-negotiable, whether production is in-house or managed by a contract development and manufacturing organization (cdmo). As more anti-angiogenic agents and tumor necrosis factor (tnf) inhibitors come off patent, the competitive landscape will intensify, making manufacturing efficiency a key differentiator.
What are the Key Data Covered in this Biosimilars Market Research and Growth Report?
-
What is the expected growth of the Biosimilars Market between 2026 and 2030?
-
USD 38.17 billion, at a CAGR of 13.3%
-
-
What segmentation does the market report cover?
-
The report is segmented by Product Type (Monoclonal antibodies, Insulin, Human growth hormone, and Others), Application (Oncology and hematology, Endocrinology, Immunology, and Nephrology), Manufacturing Type (In-house manufacturing, and Contract manufacturing) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Increasing patent expirations and intensifying focus on cost-effectiveness, Navigating evolving and complex regulatory frameworks
-
-
Who are the major players in the Biosimilars Market?
-
Abbott Laboratories, Biocon Ltd., Dr. Reddys Laboratories Ltd., Fresenius Kabi AG, Intas Pharmaceuticals Ltd., Kashiv Biosciences LLC, Mabxience Holding S.L., NeuClone Pty Ltd., Organon and Co., Pfizer Inc., Reliance Life Sciences Pvt. Ltd., Samsung Bioepis Co. Ltd., Sandoz Group AG, STADA Arzneimittel AG, Teva Pharmaceutical Ltd., Viatris Inc., Zentiva Group and Zydus Lifesciences Ltd.
-
Market Research Insights
- The market's dynamics are shaped by a push for healthcare expenditure reduction, where value-based payment models are gaining traction and have been shown to increase biosimilar adoption by up to 15% in certain health systems. The development of cost-effective therapeutic alternatives is paramount, with strategic partnerships accelerating market entry by an average of 20%.
- Companies are focused on patient access expansion, leveraging streamlined regulatory pathways to serve unmet needs in chronic disease management. This competitive environment encourages biopharmaceutical innovation, including the creation of high-concentration formulations. Ensuring public health sustainability remains a core objective, with off-patent biological medicines playing a crucial role in managing costs while maintaining high standards of care.
We can help! Our analysts can customize this biosimilars market research report to meet your requirements.


 RIA -
RIA - 
