Antinuclear Antibody Test Market Size 2025-2029
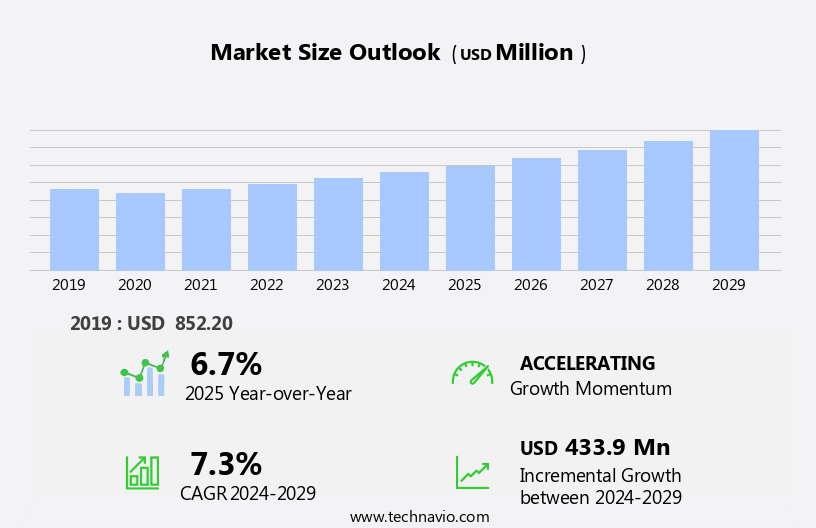
The antinuclear antibody test market size is forecast to increase by USD 433.9 million, at a CAGR of 7.3% between 2024 and 2029.
- The Antinuclear Antibody (ANA) test market is experiencing significant growth due to the increasing demand for these diagnostic tests. The rising prevalence of autoimmune diseases, which are detected through ANA tests, is a key driver for market expansion. Autoimmune diseases, such as systemic lupus erythematosus and rheumatoid arthritis, affect millions worldwide, necessitating accurate and timely diagnosis. However, the market faces challenges from stringent regulations for the approval of medical diagnostics. ANA (Antinuclear Antibody) tests have gained significant importance in the medical diagnostics industry due to the increasing prevalence of autoimmune diseases.
- Additionally, the high competition in the diagnostics industry can put pressure on prices, making it essential for companies to differentiate themselves through innovation and quality. To capitalize on market opportunities and navigate challenges effectively, companies must stay abreast of regulatory requirements and invest in research and development to offer innovative, high-quality ANA tests. The demand for ANA tests is on the rise as they are used to identify various autoimmune disorders such as Systemic Lupus Erythematosus (SLE), Rheumatoid Arthritis (RA), and Sjogren's syndrome. Regulatory bodies, including the Food and Drug Administration (FDA), require rigorous testing and validation before approving new diagnostic tools. This process can be lengthy and costly, potentially hindering market growth.
What will be the Size of the Antinuclear Antibody Test Market during the forecast period?
- In the antinuclear antibody (ANA) test market, early detection and effective management of autoimmune diseases are key priorities. Lifestyle modifications and environmental factors play a role in disease onset and progression, making predictive analytics and patient education essential. Big data analytics and artificial intelligence are transforming ANA testing, enabling the identification of unique autoantibody profiles, including anti-RNP, anti-DS DNA, anti-Scl-70, anti-Sm, and anti-centromere antibodies. Disease-modifying anti-rheumatic drugs, biological agents, and immunomodulatory therapies are crucial therapeutic strategies for managing autoimmune diseases.
- Immunoglobulin M, Immunoglobulin A, and Immunoglobulin G are essential components of immunotherapy. Targeted therapies and machine learning algorithms are revolutionizing treatment plans, tailored to individual genetic predispositions. Nonsteroidal anti-inflammatory drugs continue to be used for symptomatic relief, while personalized medicine and disease monitoring are the future of ANA testing and autoimmune disease prevention.
How is this Antinuclear Antibody Test Industry segmented?
The antinuclear antibody test industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2025-2029, as well as historical data from 2019-2023 for the following segments.
- Application
- Autoimmune diseases
- Infectious diseases
- Technique
- Enzyme-linked immunosorbent assay
- Indirect immunofluorescence
- Multiplex assay
- Others
- End-user
- Hospitals
- Clinical diagnostic laboratories
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- France
- Germany
- Italy
- UK
- APAC
- China
- Japan
- South America
- Brazil
- Rest of World (ROW)
- North America
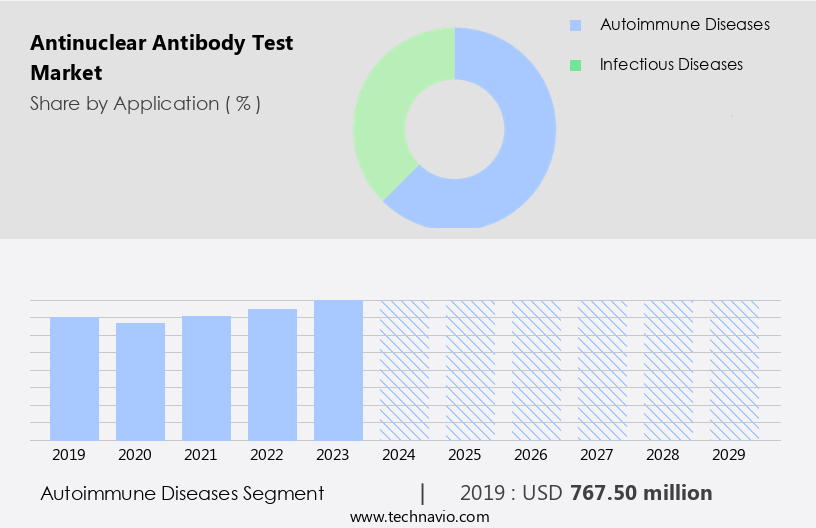
By Application Insights
The autoimmune diseases segment is estimated to witness significant growth during the forecast period. The antinuclear antibody (ANA) test market plays a pivotal role in the diagnosis and management of autoimmune diseases. This test is essential for healthcare providers to identify and monitor conditions such as rheumatoid arthritis, systemic lupus erythematosus, and Sjögren's syndrome. The homogeneous pattern of ANA tests ensures standardization and reliability, enabling accurate clinical diagnosis. New technologies, including point-of-care testing and laboratory automation, streamline the testing process, enhancing diagnostic efficiency. False positives and false negatives remain critical concerns, necessitating rigorous quality control measures. Preanalytical factors, such as patient preparation and specimen handling, significantly impact test results. Laboratories employ stringent procedures to minimize these errors, ensuring diagnostic accuracy.
Advanced analytical techniques, like cytoplasmic and nuclear staining, enable the detection of specific antinuclear antigens, such as centromere, RNP, Smith, and Scl-70. Regulatory guidelines and reference ranges influence the interpretation of ANA test results. The negative predictive value of these tests is high, making them valuable tools for ruling out autoimmune diseases. However, positive predictive value relies on differential diagnosis and clinical context. As disease progression and treatment response evolve, diagnostic companies continue to refine testing methodologies and analytical validation to improve diagnostic accuracy. Home testing and clinical trials expand access to ANA testing, enabling early detection and management of autoimmune diseases.
The Autoimmune diseases segment was valued at USD 767.50 million in 2019 and showed a gradual increase during the forecast period.
The integration of new technologies and advanced laboratory procedures enhances diagnostic precision, supporting more effective patient management. Despite these advancements, challenges persist, including interfering substances and postanalytical factors. Industry participants remain committed to addressing these issues, ensuring the continued relevance and reliability of ANA testing in healthcare. Assays used for ANA testing include Enzyme-linked Immunosorbent Assay (ELISA), Immunofluorescence Assay (IFA), and Multiplex Assay.
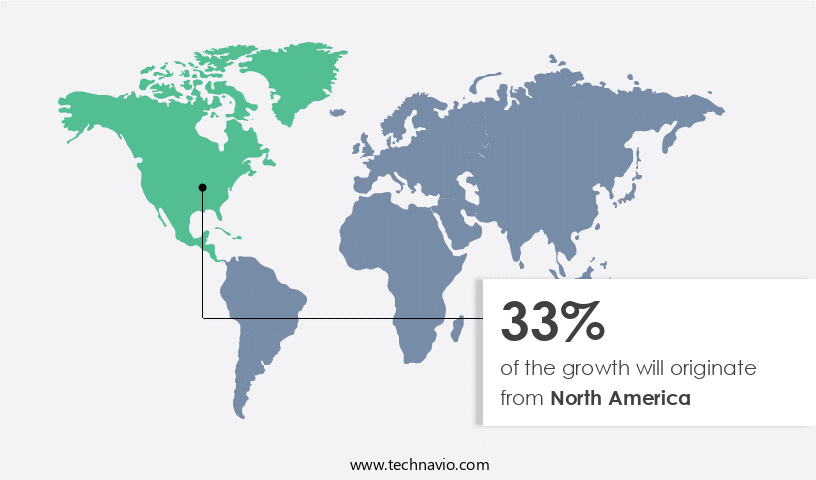
Regional Analysis
North America is estimated to contribute 33% to the growth of the global market during the forecast period. Technavio's analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
Market Dynamics
Our researchers analyzed the data with 2024 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
What are the Antinuclear Antibody Test market drivers leading to the rise in the adoption of Industry?
- The significant rise in the demand for ANA (Antinuclear Antibody) tests serves as the primary growth catalyst for the associated market. The Antinuclear Antibody (ANA) test is a crucial diagnostic tool for identifying autoimmune disorders, such as rheumatoid arthritis, Systemic Lupus Erythematosus, and Sjögren's Syndrome. This test detects the presence of antinuclear antibodies (ANAs) in a patient's blood, which bind to the body's own tissues. Interfering substances, such as certain medications and infections, can affect the diagnostic accuracy of ANA tests. The demand for ANA tests is driven by the increasing prevalence of autoimmune disorders and the need for early diagnosis and effective patient management. For instance, Sjögren's Syndrome, a chronic autoimmune disorder, affects an estimated 4 million Americans. Early diagnosis and appropriate drug therapy can help mitigate the symptoms and prevent complications.
- Laboratory automation and analytical validation have improved the efficiency and accuracy of ANA testing. However, challenges remain, including the presence of false-positive results due to analytical factors and the need for home testing to expand access to diagnostic services. Clinical trials and collaborations between diagnostic companies are ongoing to address these challenges and improve the overall quality and accessibility of ANA testing. The ANA test plays a vital role in identifying autoimmune disorders and ensuring effective patient management. The increasing prevalence of these disorders, advancements in laboratory technology, and ongoing research efforts are expected to fuel the demand for ANA testing in the coming years.
What are the Antinuclear Antibody Test market trends shaping the Industry?
- Autoimmune diseases are on the rise, representing a significant market trend in the healthcare industry. The increasing prevalence of these conditions warrants continued research and development in diagnostic tools and treatment options. The Antinuclear Antibody (ANA) test is a crucial diagnostic tool for identifying autoimmune disorders, particularly Systemic Lupus Erythematosus (SLE). This test detects the presence of antibodies against nuclear antigens, such as RNP antigen, Smith antigen, Scl-70 antigen, and double-stranded DNA. SLE patients typically exhibit a positive ANA test result, but other conditions, including viral infections and certain medications, may also cause a positive result. The rising prevalence of autoimmune disorders can be attributed to various environmental factors that disrupt the immune system. SLE, which affects women disproportionately (with a 7:1 female-to-male ratio), is believed to result from the overexpression of CD40LG and CXCR3 genes due to the lack of X Chromosome inactivation.
- The ANA test's accuracy depends on the reference ranges and the pattern of antibody distribution. A speckled pattern is indicative of a high titer of ANA, while a peripheral pattern suggests a lower titer. To ensure test accuracy and consistency, quality control measures are essential. General practitioners often use the ANA test as a first step in diagnosing autoimmune disorders. A positive ANA test result requires further testing to confirm the diagnosis and determine the specific autoimmune disorder. The positive predictive value of the ANA test depends on the prevalence of autoimmune disorders in the population being tested.
How does Antinuclear Antibody Test market faces challenges face during its growth?
- The stringent regulatory approvals process for medical diagnostics poses a significant challenge to the industry's growth trajectory. This hurdle, which requires extensive testing and validation to ensure accuracy and safety, can significantly delay product launch timelines and increase development costs for companies in this sector. Antinuclear Antibody (ANA) tests are essential diagnostic tools for identifying autoimmune diseases. Adherence to standard operating procedures is crucial in the production and implementation of ANA tests to ensure accurate clinical diagnosis. New technologies, such as homogeneous pattern assays and point-of-care testing, offer advantages in terms of speed and convenience. However, these methods may introduce false positives or false negatives due to postanalytical factors, necessitating careful laboratory procedures.
- Department of Health and Human Services. These agencies monitor the design, testing, manufacture, labeling, promotion, and marketing of medical diagnostic devices, including ANA test kits. Ensuring compliance with these regulations can be time-consuming and costly for manufacturers. Laboratory procedures for ANA testing involve specimen collection, nuclear staining, and interpretation of results. False positives and false negatives can occur due to various factors, necessitating differential diagnosis. Differential diagnosis involves considering other possible causes for the patient's symptoms and ruling out alternative diagnoses. ANA tests play a vital role in the diagnosis of autoimmune diseases. Regulations on the approval of medical diagnostics are stringently enforced by regulatory agencies, including the US Food and Drug Administration (FDA) and the Center for Medicare Services of the U.S.
Exclusive Customer Landscape
The antinuclear antibody test market forecasting report includes the adoption lifecycle of the market, covering from the innovator's stage to the laggard's stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the antinuclear antibody test market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape
Key Companies & Market Insights
Companies are implementing various strategies, such as strategic alliances, antinuclear antibody test market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Abbott Laboratories - The company introduces an advanced antinuclear antibody testing solution, featuring the Agility ELISA automation system.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Abbott Laboratories
- Abcam plc
- Antibodies Inc.
- Bio Rad Laboratories Inc.
- BioVision Inc.
- Grifols SA
- Immuno Concepts Ltd.
- Merck KGaA
- Orgentec Diagnostika GmbH
- Perkin Elmer Inc.
- QuidelOrtho Corp.
- Seramun Diagnostica GmbH
- Thermo Fisher Scientific Inc.
- Transasia Bio Medicals Ltd.
- Trinity Biotech Plc
- Werfenlife SA
- ZEUS Scientific Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Antinuclear Antibody Test Market
- In February 2023, Roche Diagnostics, a leading diagnostics company, announced the launch of its new Elecsys ANA Plus assay, which offers improved sensitivity and specificity for the detection of antinuclear antibodies (ANA), according to a Roche press release. This innovation is expected to enhance diagnostic accuracy and support earlier and more precise diagnosis of various autoimmune diseases.
- In November 2024, Thermo Fisher Scientific and F. Hoffmann-La Roche entered into a strategic collaboration to co-develop and commercialize a new range of in vitro diagnostic tests, including ANA tests, as stated in a Thermo Fisher Scientific press release. This partnership aims to leverage both companies' expertise and resources to expand their diagnostic offerings and improve patient care.
- In March 2025, Grifols, a global healthcare company, completed the acquisition of Bio-Rad Laboratories' Diagnostic Immunology Business for USD 1.35 billion, as reported by Reuters. This acquisition significantly strengthened Grifols' presence in the in vitro diagnostics market, particularly in the ANA test segment.
Research Analyst Overview
The Antinuclear Antibody (ANA) test market continues to evolve, driven by the constant emergence of new technologies and standard operating procedures. ANAs are a type of autoantibodies produced in response to various autoimmune diseases, including Systemic Lupus Erythematosus (SLE), Rheumatoid Arthritis (RA), and Sjögren's Syndrome. These antibodies can target different nuclear antigens such as RNP antigen, Smith antigen, centromere antigen, and Scl-70 antigen, among others. The test's interpretation requires a deep understanding of the various ANA patterns, including homogeneous, speckled, and cytoplasmic staining. Reference ranges and ANA titers play a crucial role in clinical diagnosis, disease monitoring, and treatment response assessment.
However, the test's accuracy is influenced by numerous factors, including preanalytical, analytical, and postanalytical factors. False positives and false negatives can occur due to interfering substances, specimen handling, and patient preparation. Laboratory automation and point-of-care testing are gaining popularity to improve diagnostic accuracy and reduce turnaround time. Quality control measures are essential to ensure reliable results and minimize variability. Healthcare providers rely on ANA testing for disease monitoring, differential diagnosis, and precision assessment. Drug therapy response evaluation and disease progression assessment are critical applications of ANA testing. Clinical trials and diagnostic companies are continually researching new methods and technologies to enhance the test's diagnostic accuracy and reduce the impact of interfering substances.
Negative predictive value and positive predictive value are essential performance indicators for ANA testing. Continuous method comparison and analytical validation are necessary to maintain diagnostic accuracy and ensure the test's reliability. The ongoing unfolding of market activities and evolving patterns underscore the importance of staying abreast of the latest developments in ANA testing. For instance, according to the Foundation for Peripheral Neuropathy report, approximately 3,000-6,000 people in the US are diagnosed with Guillain-Barre Syndrome (GBS) every year.
The Antinuclear Antibody (ANA) Test Market is expanding rapidly due to increasing awareness of autoimmune disorders and their early detection. This test is crucial for identifying conditions linked to genetic predisposition and guiding effective autoimmune disease management. Various antibodies, including anti-DNA antibody, antism antibody, antirnp antibody, anti-centromere antibody, and anti-SCL antibody, play a vital role in diagnosing specific autoimmune diseases. The demand for ANA tests is growing as healthcare professionals leverage immunomodulatory therapy to manage symptoms and improve patient outcomes.
Dive into Technavio's strong research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Antinuclear Antibody Test Market insights. See full methodology.
|
Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
207 |
|
Base year |
2024 |
|
Historic period |
2019-2023 |
|
Forecast period |
2025-2029 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 7.3% |
|
Market growth 2025-2029 |
USD 433.9 million |
|
Market structure |
Fragmented |
|
YoY growth 2024-2025(%) |
6.7 |
|
Key countries |
US, Germany, UK, China, Canada, France, Brazil, Japan, Italy, and Mexico |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
What are the Key Data Covered in this Antinuclear Antibody Test Market Research and Growth Report?
- CAGR of the Antinuclear Antibody Test industry during the forecast period
- Detailed information on factors that will drive the growth and forecasting between 2025 and 2029
- Precise estimation of the size of the market and its contribution of the industry in focus to the parent market
- Accurate predictions about upcoming growth and trends and changes in consumer behaviour
- Growth of the market across North America, Europe, Asia, and Rest of World (ROW)
- Thorough analysis of the market's competitive landscape and detailed information about companies
- Comprehensive analysis of factors that will challenge the antinuclear antibody test market growth of industry companies
We can help! Our analysts can customize this antinuclear antibody test market research report to meet your requirements.


 RIA -
RIA - 
