Eclinical Solutions Market Size 2024-2028
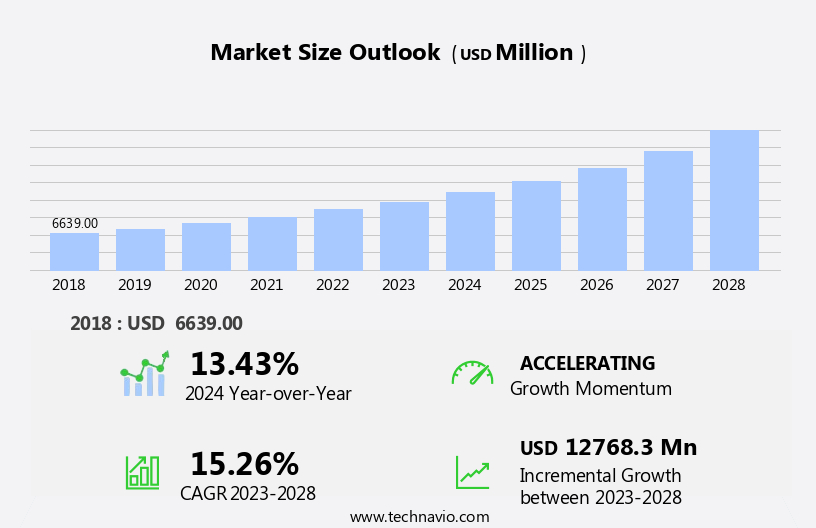
The eclinical solutions market size is forecast to increase by USD 12.77 billion, at a CAGR of 15.26% between 2023 and 2028.
- The market is experiencing significant growth, driven by the surge in clinical trials for new drugs. This trend is fueled by the increasing outsourcing of clinical trial processes to specialized organizations, providing cost savings and efficiency gains for pharmaceutical companies and enabling faster and more efficient drug development. However, this market also faces challenges, with the rising cost of clinical trials being a major obstacle. The complexity and intricacy of clinical trials, coupled with the need for advanced technology and expertise, contribute to escalating costs.
- To navigate these challenges, companies must focus on optimizing their clinical trial processes and exploring cost-effective solutions. Strategic collaborations, technology innovation, and operational efficiency are key areas of focus for market participants seeking to capitalize on the opportunities presented by the growing market.
What will be the Size of the Eclinical Solutions Market during the forecast period?
Explore in-depth regional segment analysis with market size data - historical 2018-2022 and forecasts 2024-2028 - in the full report.
Request Free Sample
The market continues to evolve, with dynamic market activities unfolding across various sectors. Integration of ECOA (Electronic Clinical Outcome Assessment) into data warehousing solutions is a notable trend, enhancing data security measures and streamlining clinical trial workflows. Data migration strategies are being refined to ensure seamless transition between systems, while eClinical company selection is increasingly focused on advanced features such as ETMF software, data quality metrics, user access management, and remote data capture. Study start-up timelines are under scrutiny as data integrity protocols and data standardization methods gain importance. Reporting modules and project management tools are being integrated into eClinical platforms to improve clinical trial management and regulatory compliance.
Medical device integration, electronic consent forms, and audit trail management are also key components of modern eClinical systems. Data privacy regulations and patient safety reporting are critical considerations in the eClinical landscape. System usability testing, security risk assessment, and EDC system validation are essential elements of eClinical system architecture. The ongoing evolution of clinical trial technology necessitates continuous adaptation to new requirements and regulatory guidelines.
How is this Eclinical Solutions Industry segmented?
The eclinical solutions industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2024-2028, as well as historical data from 2018-2022 for the following segments.
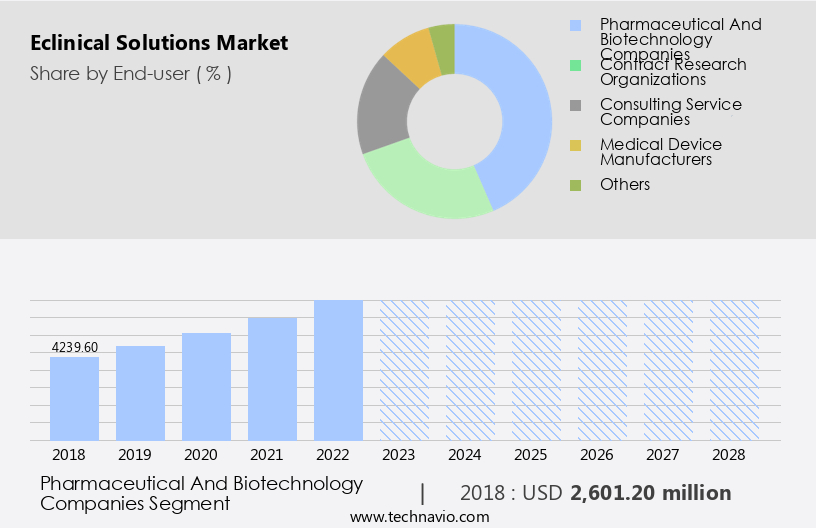
- End-user
- Pharmaceutical and biotechnology companies
- Contract research organizations
- Consulting service companies
- Medical device manufacturers
- Others
- Deployment
- Cloud-based
- On-premises
- Product
- Electronic Data Capture (EDC) and Clinical Data Management Systems (CDMS)
- Clinical Trial Management Systems (CTMS)
- Clinical Analytics Platforms
- Randomization and Trial Supply Management (RTSM)
- Clinical Data Integration Platforms
- Electronic Clinical Outcome Assessment (eCOA)
- Safety Solutions
- Electronic Trial Master File (eTMF)
- Electronic Consent (eConsent)
- Geography
- North America
- US
- Europe
- Germany
- UK
- APAC
- China
- Japan
- Rest of World (ROW)
- North America
By End-user Insights
The pharmaceutical and biotechnology companies segment is estimated to witness significant growth during the forecast period.
In the realm of clinical trials, eCoA integration and data warehousing solutions play a crucial role in streamlining processes and ensuring data accuracy. Data security measures are essential to protect sensitive information, while ETMF software features facilitate regulatory compliance. Clinical trial workflows are optimized with data migration strategies and case report forms, enabling seamless transitions between stages. company selection, randomization systems, and ePro data analysis contribute to efficient clinical trial management. Integration APIs, data quality metrics, user access management, and remote data capture enhance operational effectiveness. Data integrity protocols, data standardization methods, reporting modules, project management tools, and clinical trial technology form the backbone of eClinical systems.
Regulatory compliance tools, patient safety reporting, validation documentation, and data privacy regulations ensure transparency and trust. Medical device integration, electronic consent forms, audit trail management, and eClinical platforms facilitate site initiation visits and EDC system validation. System usability testing and security risk assessment are integral to maintaining system performance and reliability. The evolving clinical trial landscape emphasizes the importance of these integrated entities in addressing complexities and ensuring successful trial outcomes.
The Pharmaceutical and biotechnology companies segment was valued at USD 2.6 billion in 2018 and showed a gradual increase during the forecast period.
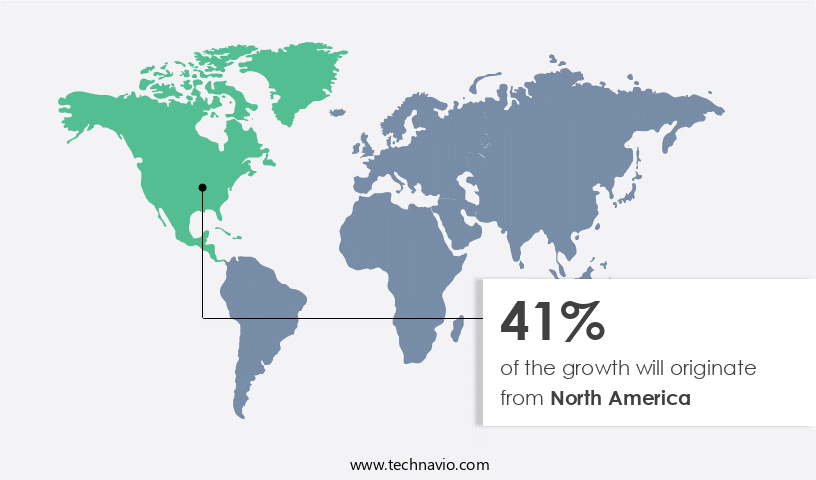
Regional Analysis
North America is estimated to contribute 41% to the growth of the global market during the forecast period. Technavio's analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
The market in North America is experiencing notable growth, driven by factors such as streamlined regulatory processes, a large presence of pharmaceutical and biotechnology companies and Contract Research Organizations (CROs), and substantial investments in clinical trial research. The US leads as the primary revenue contributor, with Canada and Mexico following suit. The number of clinical trials in these countries has risen, making North America an attractive region for clinical trial management systems (CTMS) market expansion. Clinical trial workflows are being optimized through advanced eClinical solutions, including data warehousing, data security measures, and data migration strategies.
Case report forms are being digitized, and ePro data analysis is facilitating real-time insights. company selection for eClinical systems is a critical process, with considerations given to randomization systems, integration APIs, data quality metrics, user access management, and remote data capture. Data integrity protocols, data standardization methods, and regulatory compliance tools are essential components of eClinical systems. Patient safety reporting, validation documentation, and data privacy regulations are also prioritized. Medical device integration and electronic consent forms are becoming increasingly important. Audit trail management and eClinical platforms are streamlining site initiation visits and EDC system validation.
System usability testing and security risk assessments are crucial elements in ensuring the success and reliability of eClinical solutions.
Market Dynamics
Our researchers analyzed the data with 2023 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
The market encompasses a range of technologies and services designed to streamline clinical research processes, focusing on data management, electronic data capture (EDC), and medical imaging. With considerations like increasing regulatory compliance, data security, and the need for real-time data access, eClinical Solutions have become essential for pharmaceutical and biotech companies. Key areas involve implementing advanced analytics and machine learning algorithms to improve clinical trial design, execution, and analysis. Through methods such as remote monitoring and real-time data capture, eClinical Solutions enable decentralized trials, reducing the need for on-site visits and accelerating trial timelines. Moreover, the market players focus on enhancing patient engagement and experience by integrating patient-reported outcomes (PROs) and electronic patient-reported outcomes (ePROs) into clinical trials. This approach allows for a more comprehensive understanding of patient experiences and treatment efficacy. Additionally, the integration of cloud technologies and mobile applications in eClinical Solutions facilitates global collaboration and data sharing among research sites, sponsors, and regulatory agencies. This not only expedites the clinical trial process but also ensures data consistency and accuracy. Furthermore, the market players invest in advanced technologies like artificial intelligence (AI) and natural language processing (NLP) to automate data processing, improve data quality, and enhance data interoperability. These advancements enable faster and more accurate data analysis, leading to better clinical trial outcomes. In conclusion, the market continues to evolve, addressing the complexities of clinical research and improving trial efficiency, data quality, and patient engagement. The integration of advanced technologies and a focus on data security and regulatory compliance make eClinical Solutions an indispensable component of the clinical research landscape.
What are the key market drivers leading to the rise in the adoption of Eclinical Solutions Industry?
- The increase in clinical trials for drugs serves as the primary catalyst for market growth.
- The market is experiencing significant growth due to the increasing number of clinical trials for drugs worldwide. With over 200,000 studies listed on clinicaltrials.Gov, the global registry of clinical trials, trials are being conducted in all 50 states of the US and over 150 countries. The annual number of registered trials has increased progressively in Asian countries like Japan and India, driven by local measures that encourage and enforce registration. Data security measures and eCOA integration are crucial features for eClinical companies in this market. Data warehousing solutions enable the collection, storage, and analysis of vast amounts of clinical trial data.
- Data migration strategies ensure seamless transition from one system to another. Case report forms are essential for data collection and management during clinical trials. Clinical trial workflows are optimized with the help of eClinical systems, including randomization systems. Selecting the right eClinical company is a critical decision for pharmaceutical and biotech companies. Companies offering advanced ETMF software features, such as document management, workflow automation, and regulatory compliance, are preferred. Effective data security measures, including encryption and access control, are essential to protect sensitive clinical trial data. The market is dynamic, with ongoing advancements in technology and regulatory requirements driving innovation and competition.
What are the market trends shaping the Eclinical Solutions Industry?
- The trend in the clinical trials industry is shifting towards increased outsourcing of processes. This outsourcing of tasks is becoming increasingly common and mandatory for many organizations to remain competitive and efficient.
- The market has witnessed significant growth in recent years due to the increasing focus on data-driven research and development in the pharmaceutical industry. Small companies are outsourcing their drug discovery process to subcontract laboratories, allowing them to concentrate on manufacturing and marketing activities. This trend is expected to continue, with the number of subcontract laboratories increasing since 2011. The clinical research industry has undergone a transformation in the last decade, moving from a vertical reporting model with a large in-house workforce to a matrix model involving both external and internal resources. The majority of clinical research work is now being outsourced to Contract Research Organizations (CROs).
- Data integrity and quality are crucial in the market. Data analysis is performed using advanced tools and methods such as ePRO data analysis and data standardization. Integration APIs facilitate seamless data transfer between systems, ensuring data consistency. User access management is implemented to maintain data security, and remote data capture is utilized to increase efficiency. Data quality metrics are monitored to ensure accuracy and reliability. Data integrity protocols are strictly adhered to, and reporting modules provide real-time access to data. These solutions enable sponsors and researchers to make informed decisions, reduce study start-up timelines, and improve overall study outcomes.
What challenges does the Eclinical Solutions Industry face during its growth?
- The escalating costs of clinical trials represent a significant challenge to the growth of the industry. In order to maintain profitability and bring new treatments to market, companies must effectively manage these expenses and explore cost-saving strategies.
- Clinical trial management has experienced significant evolution, with escalating costs being a major concern. Factors contributing to this increase include the complex and protracted nature of clinical trials. Patient enrollment is a significant challenge, followed by ensuring patient compliance and retention throughout the research study. According to recent research, the cost per patient in clinical trials in the US rose by over 150% between 2010 and 2015, while in the UK, the increase was around 85%-90%. To address these challenges and reduce costs, clinical trial technology has emerged as a crucial solution. Project management tools and eClinical system architecture are essential components of this technology.
- Regulatory compliance tools ensure adherence to stringent regulations, while patient safety reporting and validation documentation maintain data integrity. Data privacy regulations are also addressed, safeguarding sensitive patient information. Medical device integration is another critical aspect, enabling seamless communication between various devices and systems. These advancements contribute to more immersive and harmonious clinical trial management, ultimately striking a balance between efficiency and patient safety. By emphasizing these aspects, clinical trial management becomes more effective and cost-efficient.
Exclusive Customer Landscape
The eclinical solutions market forecasting report includes the adoption lifecycle of the market, covering from the innovator's stage to the laggard's stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the eclinical solutions market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape
Key Companies & Market Insights
Companies are implementing various strategies, such as strategic alliances, eclinical solutions market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Advarra Inc. - This company specializes in providing eClinical solutions, including CTMS and RTSM masters, enhancing clinical trial efficiency and data management for the global life sciences industry.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Advarra Inc.
- Aixial
- Anju Software Inc.
- ArisGlobal LLC
- Castor Research Inc.
- Dassault Systemes SE
- DATATRAK International Inc.
- eClinical Solutions LLC.
- eClinicalWorks LLC
- eResearchTechnology GmbH
- International Business Machines Corp.
- MedNet
- Medrio Inc.
- OpenClinica LLC
- Oracle Corp.
- Parexel International Corp.
- Saama Technologies Inc.
- Signant Health
- Veeva Systems Inc.
- YPrime LLC
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Eclinical Solutions Market
- In January 2024, Medidata Solutions, a leading global provider of cloud-based solutions for clinical research, announced the launch of its new eClinical platform, Medidata Rave 5.0. This advanced platform integrates electronic data capture, trial randomization and supply, and site payments, aiming to streamline clinical trials and improve operational efficiency (Medidata Press Release).
- In March 2024, IQVIA, a leading global provider of integrated clinical research services, entered into a strategic partnership with Oracle Health Sciences to offer a unified, end-to-end clinical trial solution. This collaboration combined IQVIA's clinical research services with Oracle Health Sciences' technology, providing customers with a more comprehensive and efficient clinical trial offering (IQVIA Press Release).
- In May 2024, Veeva Systems, a leading provider of industry-specific software for the global life sciences industry, raised USD 1.2 billion in a secondary public offering. The funds will be used to fuel Veeva's continued growth and innovation in the market (Veeva Systems SEC Filing).
- In January 2025, the U.S. Food and Drug Administration (FDA) announced the finalization of its eSource Data Elements and Data Standards for Electronic Source Data Amendments. This initiative aims to improve data quality and reduce the time and cost of clinical trials by promoting the use of electronic source data (FDA Press Release).
Research Analyst Overview
- In the clinical solutions market, effective budget management is crucial for optimizing resources during study execution. Query management processes are streamlined through project tracking software, enabling real-time visibility into system upgrade planning and clinical trial monitoring. Risk-based monitoring and system maintenance ensure data integrity, while database administration and compliance audit processes maintain regulatory standards. EDC system design, ETMF implementation, and adverse event reporting rely on centralized data storage and quality control processes.
- company relationship management enhances operational efficiency, and eCOA device compatibility is essential for patient recruitment strategies. System performance testing, clinical trial reporting, data validation rules, and data cleaning procedures all contribute to the data governance framework. Workflow automation and data anonymization techniques further improve overall market trends.
Dive into Technavio's robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Eclinical Solutions Market insights. See full methodology.
|
Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
175 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 15.26% |
|
Market growth 2024-2028 |
USD 12768.3 million |
|
Market structure |
Fragmented |
|
YoY growth 2023-2024(%) |
13.43 |
|
Key countries |
US, Germany, UK, China, and Japan |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
What are the Key Data Covered in this Eclinical Solutions Market Research and Growth Report?
- CAGR of the Eclinical Solutions industry during the forecast period
- Detailed information on factors that will drive the growth and forecasting between 2024 and 2028
- Precise estimation of the size of the market and its contribution of the industry in focus to the parent market
- Accurate predictions about upcoming growth and trends and changes in consumer behaviour
- Growth of the market across North America, Europe, Asia, and Rest of World (ROW)
- Thorough analysis of the market's competitive landscape and detailed information about companies
- Comprehensive analysis of factors that will challenge the eclinical solutions market growth of industry companies
We can help! Our analysts can customize this eclinical solutions market research report to meet your requirements.


 RIA -
RIA - 
