Contract Research Organization (CRO) Market Size 2026-2030
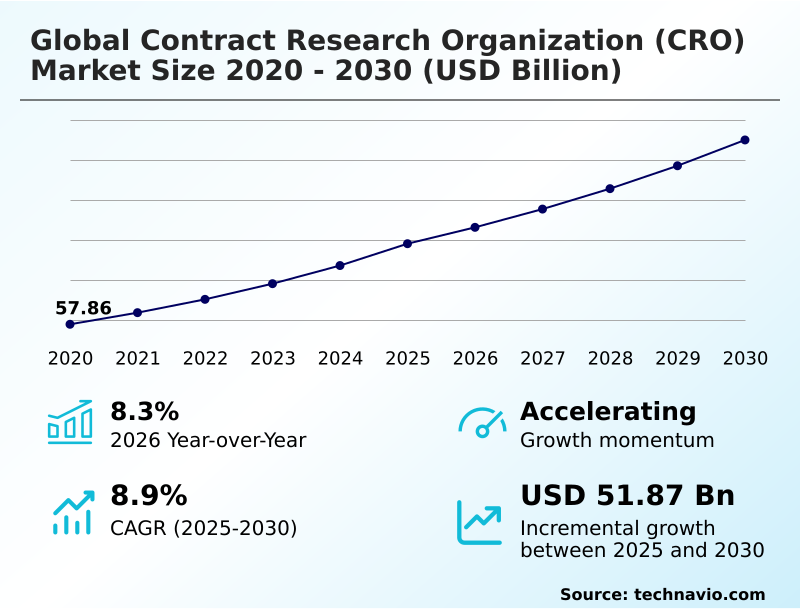
The contract research organization (cro) market size is valued to increase by USD 51.87 billion, at a CAGR of 8.9% from 2025 to 2030. Escalating research and development investments as primary catalyst will drive the contract research organization (cro) market.
Major Market Trends & Insights
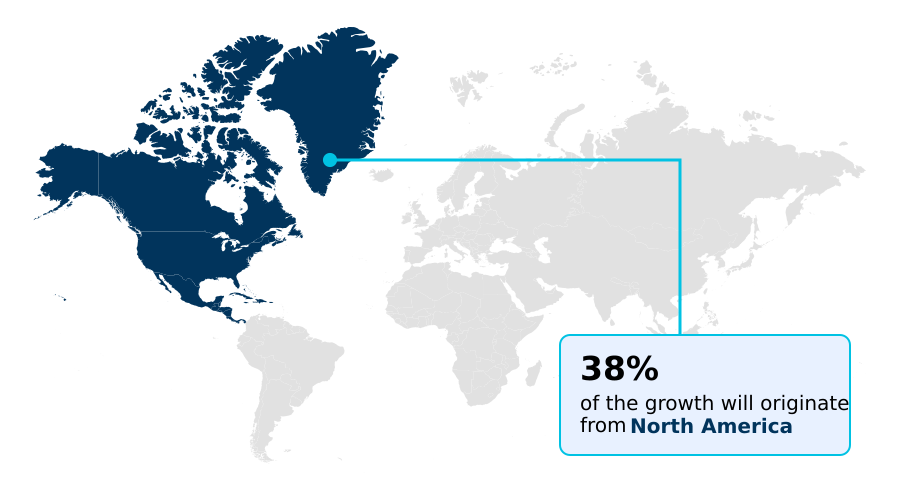
- North America dominated the market and accounted for a 38.2% growth during the forecast period.
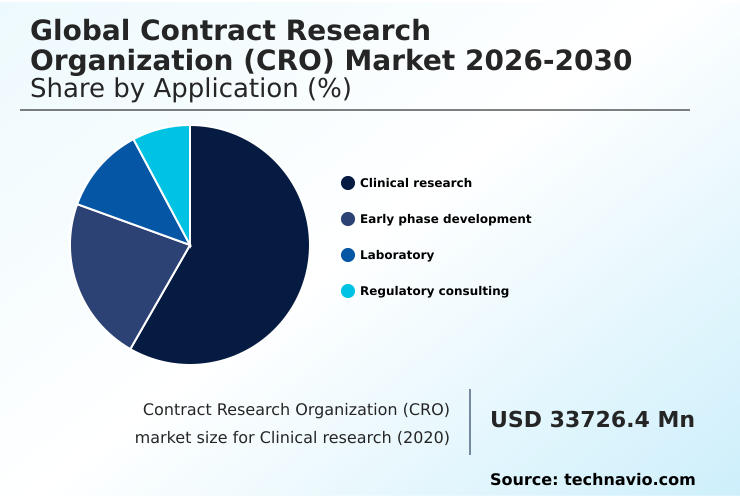
- By Application - Clinical research segment was valued at USD 50.58 billion in 2024
- By End-user - Pharmaceutical and biopharmaceutical companies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 92.16 billion
- Market Future Opportunities: USD 51.87 billion
- CAGR from 2025 to 2030 : 8.9%
Market Summary
- The contract research organization (CRO) market is undergoing significant expansion, driven by the biopharmaceutical industry's strategic shift toward outsourcing to enhance efficiency and access specialized expertise. CROs provide end-to-end solutions across the drug development lifecycle, from preclinical research and toxicology studies to full-scale clinical trial management. This partnership model allows sponsors to navigate complex regulatory landscapes and accelerate timelines.
- For instance, a mid-sized biotech firm can leverage a CRO for its entire clinical program, including pharmacovigilance services, bioanalytical testing, and biostatistics and data management, for a new oncology drug. This avoids the high fixed costs of building in-house infrastructure for services like central laboratory services and allows the firm to focus on its core discovery competencies.
- The adoption of digital health technologies, decentralized clinical trials (DCT), and the use of real world evidence (RWE) are key trends, forcing CROs to innovate. Success hinges on therapeutic area expertise, a global operational footprint, and a commitment to quality and regulatory compliance, ensuring data integrity for submissions to bodies like the FDA and EMA.
- This evolution transforms CROs from mere vendors to integral strategic partners in medical innovation.
What will be the Size of the Contract Research Organization (CRO) Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Contract Research Organization (CRO) Market Segmented?
The contract research organization (cro) industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Application
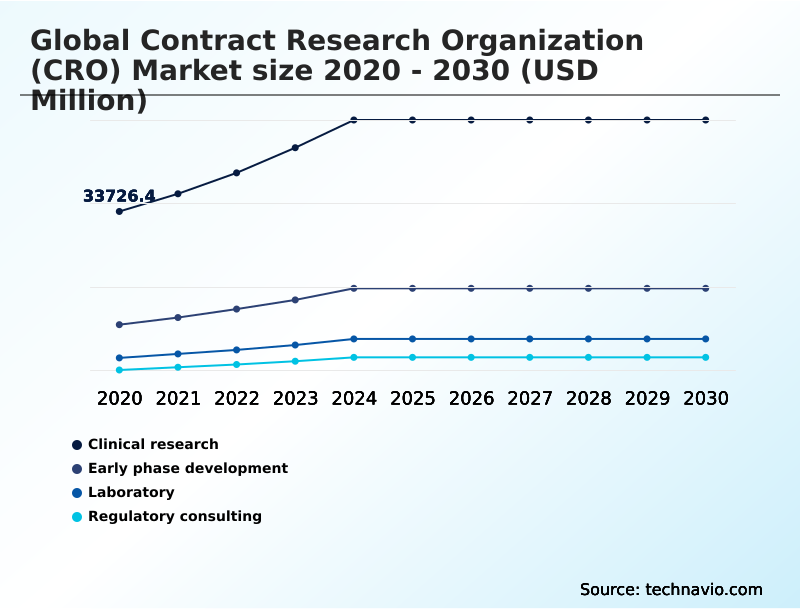
- Clinical research
- Early phase development
- Laboratory
- Regulatory consulting
- End-user
- Pharmaceutical and biopharmaceutical companies
- Medical device companies
- Academic institutes
- Therapeutic area
- Oncology
- Cardiovascular diseases
- CNS disorders
- Infectious diseases
- Others
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Application Insights
The clinical research segment is estimated to witness significant growth during the forecast period.
The clinical research segment is the largest application in the contract research organization (CRO) market, managing human trials from phase I to IV. Growth is driven by complex trial designs and the need for operational efficiency.
CROs in this domain provide a spectrum of services, including protocol design, global site selection and management, and clinical endpoint adjudication. A core competency is patient recruitment and retention, where CROs leverage sophisticated data analytics.
Effective clinical data management systems (CDMS) and adherence to good clinical practice (GCP) are essential. By partnering with CROs for these services, sponsors can achieve a reduction in trial startup times of over 25%, a significant value-based pricing advantage.
This strategic outsourcing model enables a patient-centric approach and ensures data integrity and security for successful regulatory submissions.
The Clinical research segment was valued at USD 50.58 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 38.2% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Contract Research Organization (CRO) Market Demand is Rising in North America Request Free Sample
The geographic landscape of the contract research organization (CRO) market is characterized by established dominance and rapid evolution.
North America, accounting for 38.18% of incremental growth, leads due to high R&D investment and a sophisticated regulatory environment that fosters innovation in areas like cell and gene therapy and pharmacokinetics (PK).
Europe remains a mature market, while Asia is the fastest-growing region, driven by large patient pools and improving infrastructure. The Rest of World (ROW) segment offers unique advantages; for instance, Australia provides rapid five-week study start-up times for first-in-human studies.
CROs must demonstrate a strong global operational footprint and local expertise, offering services from investigational new drug (IND) support to marketing authorization application (MAA) preparation, to effectively serve sponsors' global clinical development services needs across these diverse regions.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- The contract research organization (CRO) market is evolving to meet the complex demands of modern drug development, extending far beyond traditional trial management. A critical area of expansion involves CRO services for cell and gene therapy, which require specialized infrastructure and regulatory knowledge.
- The cost-benefit analysis of outsourcing preclinical studies consistently favors CRO partnerships, as they provide access to advanced capabilities without substantial capital investment. For later-stage development, the use of real world evidence in post-market surveillance is becoming standard practice, offering deeper insights into long-term safety and effectiveness.
- Firms leveraging specialized CRO expertise in oncology trials report data submission timelines that are up to 20% faster than those managing similar complex trials in-house. Success in this environment depends on a CRO's ability to offer integrated platforms for clinical data management and provide robust regulatory submission support for novel biologics.
- The role of CROs in personalized medicine is also growing, as they manage the complexities of biomarker-driven trials. Furthermore, AI for patient recruitment in CNS trials is a key innovation addressing historical bottlenecks.
- The ability to manage data integrity in hybrid trials, navigate both FDA and EMA regulatory pathways, and offer comprehensive pharmacovigilance for small molecule drugs are all hallmarks of a leading CRO. This shift towards strategic partnerships in drug development is redefining the CRO selection criteria for biotech startups, which now prioritize partners offering end-to-end support.
What are the key market drivers leading to the rise in the adoption of Contract Research Organization (CRO) Industry?
- Escalating research and development investments by pharmaceutical and biotechnology companies serve as the primary catalyst for market growth.
- The growth of the contract research organization (CRO) market is propelled by fundamental economic and scientific drivers.
- Escalating R&D outsourcing, a strategic imperative for biopharma, allows sponsors to adopt a flexible variable cost model, reallocating up to 30% of their operational budget to core discovery activities.
- This focus on capital efficiency is paramount as sponsors navigate increasingly complex therapeutic specialization. Access to deep therapeutic knowledge in areas like oncology and CNS disorders, provided by niche CRO providers and boutique CRO expertise, is a critical driver.
- Furthermore, the increasing complexity of clinical research, including the rise of advanced modalities, necessitates specialized preclinical research and bioanalytical testing capabilities.
- This demand for expert-driven end-to-end solutions accelerates the reliance on CROs to manage the intricacies of modern clinical development and achieve greater speed to market.
What are the market trends shaping the Contract Research Organization (CRO) Industry?
- The ascent of decentralized and hybrid clinical trial models is reshaping clinical research. This shift represents a move toward more patient-centric, technology-enabled frameworks.
- Key trends reshaping the contract research organization (CRO) market center on digitalization and strategic alignment. The pervasive integration of artificial intelligence (AI) in drug development is a dominant trend, with machine learning algorithms optimizing everything from site selection and management to risk-based monitoring.
- Hybrid trial models are now commonplace, enhancing the patient-centric approach and improving retention rates in long-term studies by over 25%. This shift requires CROs to offer seamless eClinical platform integration and advanced health economics and outcomes research (HEOR). The evolution toward strategic partnerships continues, with a focus on risk sharing partnerships that align sponsor and CRO goals.
- This dynamic fosters deeper collaboration across the drug development lifecycle, demanding more from a CRO’s supply chain management and regulatory compliance framework to deliver value.
What challenges does the Contract Research Organization (CRO) Industry face during its growth?
- An acute talent shortage and persistent workforce management pressures represent a key challenge affecting industry growth.
- The contract research organization (CRO) market faces significant operational and financial challenges that temper its growth. An acute shortage of skilled professionals leads to intense talent acquisition and retention pressures, with industry turnover rates for clinical research associates exceeding 25% annually. This workforce management issue directly impacts service quality and timelines.
- Simultaneously, CROs face relentless pricing pressure and margin compression from sponsors, forcing a delicate balance between cost containment and investment in innovative technologies like electronic data capture (EDC) and decentralized clinical trials. Upholding data privacy regulations is another major challenge, as a single breach can result in compliance fines representing up to 4% of a company’s annual revenue.
- These hurdles demand sophisticated strategies in both human capital and commercial operations.
Exclusive Technavio Analysis on Customer Landscape
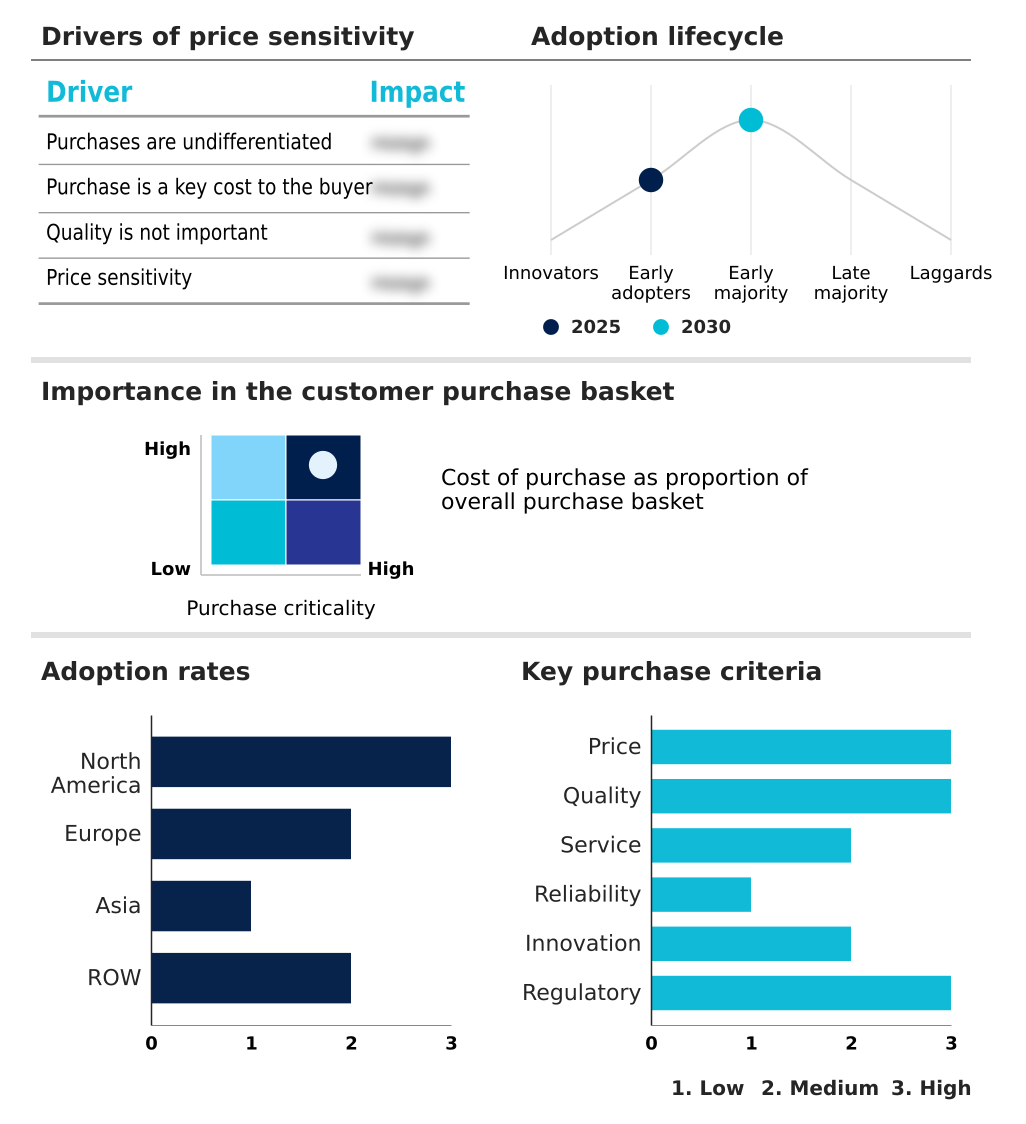
The contract research organization (cro) market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the contract research organization (cro) market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Contract Research Organization (CRO) Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, contract research organization (cro) market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Berries Health and Genetics - Delivers comprehensive contract research services, spanning clinical trial management and regulatory consulting, to support the entire drug development lifecycle.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Berries Health and Genetics
- BioAgile Therapeutics Pvt. Ltd.
- Charles River Laboratories International Inc.
- CTI Clinical Trial and Consulting Inc.
- Diagnostic Laboratory Services Inc.
- EmVenio Research
- Ergomed Plc
- Eurofins Scientific SE
- Hangzhou Tigermed Consulting Co. Ltd.
- ICON plc
- IQVIA Holdings Inc.
- Laboratory Holdings Inc.
- Medpace Holdings Inc.
- Novotech Health Holdings
- Parexel International Corp.
- Pharmaron Beijing Co. Ltd.
- Syneos Health Inc.
- Thermo Fisher Scientific Inc.
- Worldwide Clinical Trials Holdings Inc.
- WuXi AppTec Co. Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Contract research organization (cro) market
- In August 2024, ICON plc acquired KCR, a clinical trial solutions provider, to enhance its operational presence in Europe, particularly in Central and Eastern Europe.
- In October 2024, Thermo Fisher Scientific Inc. launched its Accelerator Drug Development service, integrating its contract development and manufacturing with contract research services into a unified offering.
- In January 2025, Lindus Health, a technology-focused CRO, raised $55 million in a Series B funding round to enhance its AI-driven eClinical platform.
- In January 2025, Avance Clinical announced it holds over 52% of the market share for active early-phase clinical trials in Australia, with a majority of its clients being US-based biotechs.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Contract Research Organization (CRO) Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 336 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 8.9% |
| Market growth 2026-2030 | USD 51874.2 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 8.3% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, Switzerland, China, Japan, India, South Korea, Singapore, Taiwan, Brazil, Israel, South Africa, Saudi Arabia, UAE, Argentina, Turkey and Egypt |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The contract research organization (CRO) market is evolving from a transactional service model to one of deep strategic partnership, driven by the increasing complexity of clinical development. Core functions such as clinical trial management, pharmacovigilance services, and regulatory consulting are now table stakes; differentiation comes from advanced capabilities.
- The integration of digital health technologies is central to this shift, with the adoption of decentralized clinical trials (DCT) being a key boardroom consideration impacting budgeting and risk management. This move toward virtual and hybrid models is compelling CROs to invest in eConsent platforms and robust cybersecurity postures to ensure data integrity and security.
- Companies successfully implementing these technologies have achieved a 30% reduction in data processing times. Service providers must also offer deep therapeutic area expertise in high-growth fields like cell and gene therapy and provide specialized services such as biomarker development and immunogenicity testing.
- Navigating the stringent requirements of good clinical practice (GCP) and good laboratory practice (GLP) while managing global cold chain logistics for advanced biologics remains a critical operational challenge. This dynamic environment necessitates a flexible, technology-forward approach to delivering comprehensive clinical development services.
What are the Key Data Covered in this Contract Research Organization (CRO) Market Research and Growth Report?
-
What is the expected growth of the Contract Research Organization (CRO) Market between 2026 and 2030?
-
USD 51.87 billion, at a CAGR of 8.9%
-
-
What segmentation does the market report cover?
-
The report is segmented by Application (Clinical research, Early phase development, Laboratory, and Regulatory consulting), End-user (Pharmaceutical and biopharmaceutical companies, Medical device companies, and Academic institutes), Therapeutic Area (Oncology, Cardiovascular diseases, CNS disorders, Infectious diseases, and Others) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Escalating research and development investments as primary catalyst, Acute talent shortage and workforce management pressures
-
-
Who are the major players in the Contract Research Organization (CRO) Market?
-
Berries Health and Genetics, BioAgile Therapeutics Pvt. Ltd., Charles River Laboratories International Inc., CTI Clinical Trial and Consulting Inc., Diagnostic Laboratory Services Inc., EmVenio Research, Ergomed Plc, Eurofins Scientific SE, Hangzhou Tigermed Consulting Co. Ltd., ICON plc, IQVIA Holdings Inc., Laboratory Holdings Inc., Medpace Holdings Inc., Novotech Health Holdings, Parexel International Corp., Pharmaron Beijing Co. Ltd., Syneos Health Inc., Thermo Fisher Scientific Inc., Worldwide Clinical Trials Holdings Inc. and WuXi AppTec Co. Ltd.
-
Market Research Insights
- The contract research organization (CRO) market is defined by a dynamic shift toward strategic outsourcing models, where sponsors seek end-to-end solutions to improve capital efficiency. This has led to an increase in preferred provider agreements, with sponsors consolidating work to achieve operational consistency. This trend has been shown to improve study startup times by over 15%.
- Simultaneously, the integration of AI in drug development is transforming key processes; advanced machine learning algorithms for patient identification can boost enrollment rates by up to 30% compared to traditional methods. While large providers offer a global operational footprint, niche CRO providers thrive by offering deep therapeutic specialization.
- This competitive environment, marked by risk sharing partnerships and a focus on speed to market, pushes all firms to enhance their eClinical platform capabilities and data privacy regulations adherence.
We can help! Our analysts can customize this contract research organization (cro) market research report to meet your requirements.


 RIA -
RIA - 
