Pruritus Drugs Market Size 2026-2030
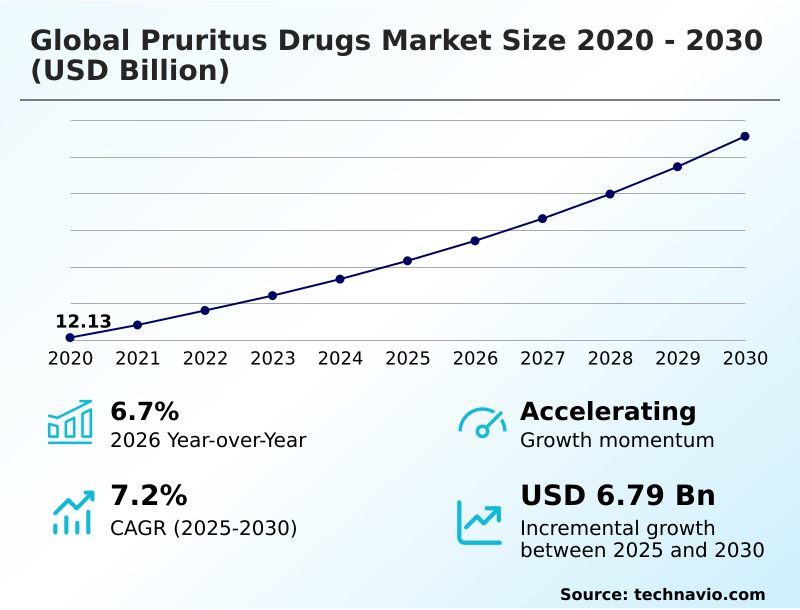
The pruritus drugs market size is valued to increase by USD 6.79 billion, at a CAGR of 7.2% from 2025 to 2030. Increasing prevalence of chronic systemic and dermatological diseases will drive the pruritus drugs market.
Major Market Trends & Insights
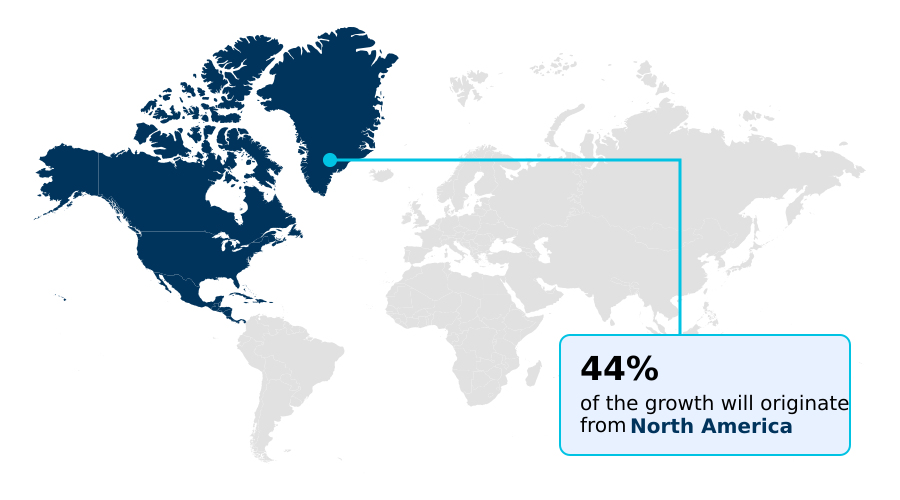
- North America dominated the market and accounted for a 44.1% growth during the forecast period.
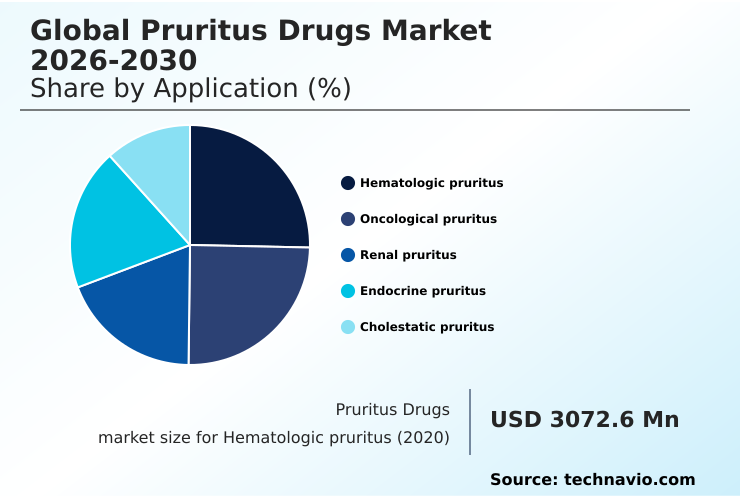
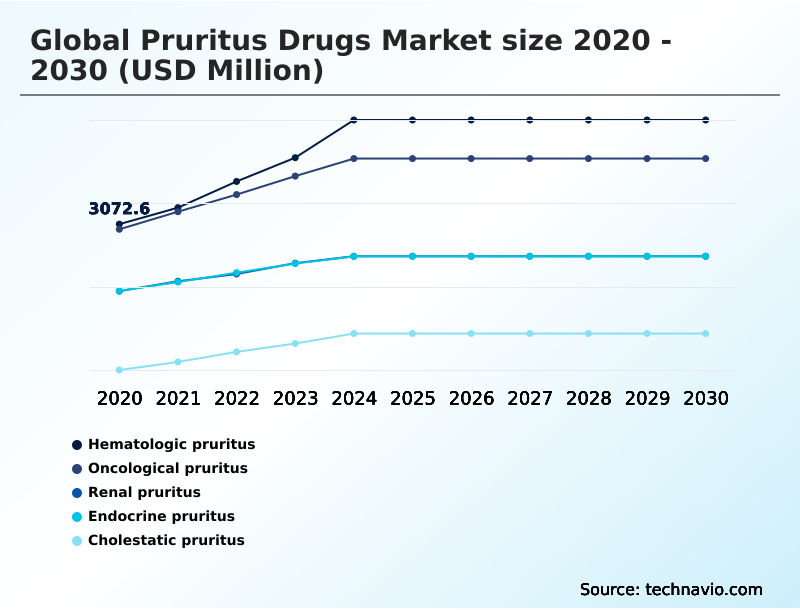
- By Application - Hematologic pruritus segment was valued at USD 4.26 billion in 2024
- By Route of Administration - Oral segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 10.98 billion
- Market Future Opportunities: USD 6.79 billion
- CAGR from 2025 to 2030 : 7.2%
Market Summary
- The pruritus drugs market is advancing beyond generalized symptom management toward precise, mechanism-based interventions. This evolution is driven by a deeper understanding of the neuroimmune pathways and cytokine cascades that mediate chronic itch in various dermatological and systemic diseases.
- As a result, the therapeutic landscape is increasingly defined by targeted biologics and small-molecule inhibitors that offer superior efficacy compared to traditional antihistamines or corticosteroids. A significant business challenge involves navigating complex reimbursement landscapes; for instance, a company launching a novel monoclonal antibody for atopic dermatitis must overcome restrictive step-therapy protocols that mandate failure on multiple older, less effective treatments.
- This requires generating robust pharmacoeconomic data to prove long-term value and justify premium pricing to payers. Strategic partnerships and innovative patient assistance programs are becoming critical for overcoming these access hurdles.
- Concurrently, the market is influenced by the rising prevalence of conditions like chronic kidney disease-associated pruritus and cholestatic pruritus, creating demand for specialized treatments that address the unique pathophysiology of these systemic conditions. The development of therapies with improved safety profiles, such as nonsteroidal topical agents, addresses the long-term risks associated with corticosteroid use.
What will be the Size of the Pruritus Drugs Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Get Free Sample
How is the Pruritus Drugs Market Segmented?
The pruritus drugs industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Application
- Hematologic pruritus
- Oncological pruritus
- Renal pruritus
- Endocrine pruritus
- Cholestatic pruritus
- Route of administration
- Oral
- Topical
- Injectable
- Others
- End-user
- Hospitals
- Homecare
- Specialty Clinics
- Others
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Application Insights
The hematologic pruritus segment is estimated to witness significant growth during the forecast period.
The hematologic pruritus segment of the pruritus drugs market addresses intense itching linked to blood disorders like polycythemia vera and Hodgkin's lymphoma.
The condition's complex pathogenesis, involving altered cytokine profiles and systemic histamine levels, makes it highly refractory to conventional antihistamines, creating a significant unmet clinical need.
This drives market demand for targeted therapeutic interventions, such as janus kinase inhibitors and other advanced immunomodulatory agents. The focus on personalized medicine, guided by molecular diagnostics, is transforming treatment paradigms.
Pharmaceutical development is increasingly centered on agents that address the underlying hematological drivers, with new therapies demonstrating over a 50% improvement in patient-reported itch severity, thereby improving quality of life and signaling a robust trajectory for therapeutic innovation.
The Hematologic pruritus segment was valued at USD 4.26 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 44.1% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Pruritus Drugs Market Demand is Rising in North America Get Free Sample
The geographic landscape of the pruritus drugs market is led by North America, which accounts for over 44% of the market, driven by high healthcare spending, favorable reimbursement for biologics, and a strong pipeline of janus kinase inhibitors.
Europe follows, with its market shaped by rigorous health technology assessments and centralized regulatory approvals. The Asia region is emerging as the fastest-growing market, with its expansion rate projected to be 15% higher than the global average.
This growth is fueled by rising disease prevalence, modernizing healthcare infrastructure in countries like China and India, and streamlined regulatory pathways for novel therapies.
The Rest of the World (ROW), encompassing South America, the Middle East, and Africa, presents opportunistic growth, driven by increasing awareness of conditions like atopic dermatitis and a gradual shift toward advanced therapeutics.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic development in the pruritus drugs market requires a nuanced approach to clinical trials and market access, particularly when considering the diverse etiologies of chronic itch. For companies developing targeted biologics for atopic dermatitis, demonstrating long-term safety and efficacy is paramount, especially when seeking approval for pediatric atopic dermatitis management.
- The commercial success of emerging treatments for neuropathic itch and novel therapies for idiopathic pruritus hinges on robust patient-reported outcomes in pruritus trials that can effectively communicate value to payers. In parallel, the advancement of oral small molecules for systemic itch and kappa opioid agonists for renal pruritus requires navigating distinct regulatory and reimbursement challenges.
- Firms specializing in therapies for cholestatic pruritus management, such as those with an ileal bile acid transporter inhibitors mechanism, must build strong relationships with hepatology specialists. An integrated strategy that addresses the pharmacology of neurokinin-1 receptor antagonists alongside the adverse effects of topical corticosteroids can yield a competitive advantage.
- Furthermore, a focus on the cost-effectiveness of pruritus biologics is critical, as well-formulated pharmacoeconomic data can shorten the path to formulary inclusion by up to 12 months compared to approaches lacking such evidence, thereby accelerating revenue generation for advances in prurigo nodularis treatment and other high-need indications like chronic urticaria.
What are the key market drivers leading to the rise in the adoption of Pruritus Drugs Industry?
- The increasing prevalence of chronic systemic and dermatological diseases that manifest with intense itch is a key driver fueling the expansion of the pruritus drugs market.
- The pruritus drugs market is propelled by the rising global prevalence of chronic diseases and a robust clinical pipeline.
- The increasing incidence of atopic dermatitis and other inflammatory skin disorders, coupled with an aging population susceptible to systemic conditions like cholestatic pruritus, creates a large, underserved patient base.
- In response, pharmaceutical innovation is centered on targeted biologic therapies, including janus kinase inhibitors and monoclonal antibodies, which demonstrate up to a 70% improvement in itch scores for refractory patients.
- These advancements are supported by strategic licensing agreements and acquisitions, which accelerate development timelines by an average of 18-24 months.
- These partnerships synergize the agility of biotech firms with the global commercial reach of major pharmaceutical companies, ensuring that novel treatments like ileal bile acid transporter inhibitors rapidly reach patients in need.
What are the market trends shaping the Pruritus Drugs Industry?
- A key market trend is the accelerated shift toward nonsteroidal topical innovations. This is driven by the need for safer, long-term alternatives to traditional corticosteroids for managing chronic itch.
- Key trends shaping the pruritus drugs market include a significant move toward safer, more targeted therapies. There is an accelerated adoption of nonsteroidal topical innovations, with topical phosphodiesterase inhibitors showing a 30% reduction in local adverse events compared to long-term corticosteroid use. This addresses concerns about skin atrophy and systemic absorption.
- Another major trend is the strategic expansion of existing biologics, such as interleukin-31 receptor antagonists, into idiopathic and unexplained itch syndromes, which could expand the addressable patient population by over 25%. Furthermore, the rising prominence of precision therapeutics for systemic conditions like uremic pruritus is evident.
- The development of selective kappa opioid receptor agonists, which target the peripheral nervous system without central side effects, represents a significant advancement in managing itch for patients with end-stage renal disease.
What challenges does the Pruritus Drugs Industry face during its growth?
- The high cost of advanced biologic therapies, compounded by stringent reimbursement policies and complex payer negotiations, presents a significant challenge to market growth and patient access.
- Significant challenges constrain the pruritus drugs market, led by the high cost of advanced biologic therapies and stringent reimbursement policies. Payers often implement restrictive step-therapy protocols, delaying patient access to novel treatments like monoclonal antibodies by an average of six months. This creates a substantial barrier to care, particularly for those with severe prurigo nodularis.
- Another critical issue is the widespread underdiagnosis of chronic itch, especially in developing regions, which leaves an estimated 30% of the potential market unaddressed due to a lack of standardized diagnostic tools and specialist access. Furthermore, complex health technology assessments and fragmented regulatory pathways, particularly in Europe, create significant hurdles.
- Gaining market access for a new interleukin-31 receptor antagonist can require separate, lengthy negotiations in each country, dampening revenue potential.
Exclusive Technavio Analysis on Customer Landscape
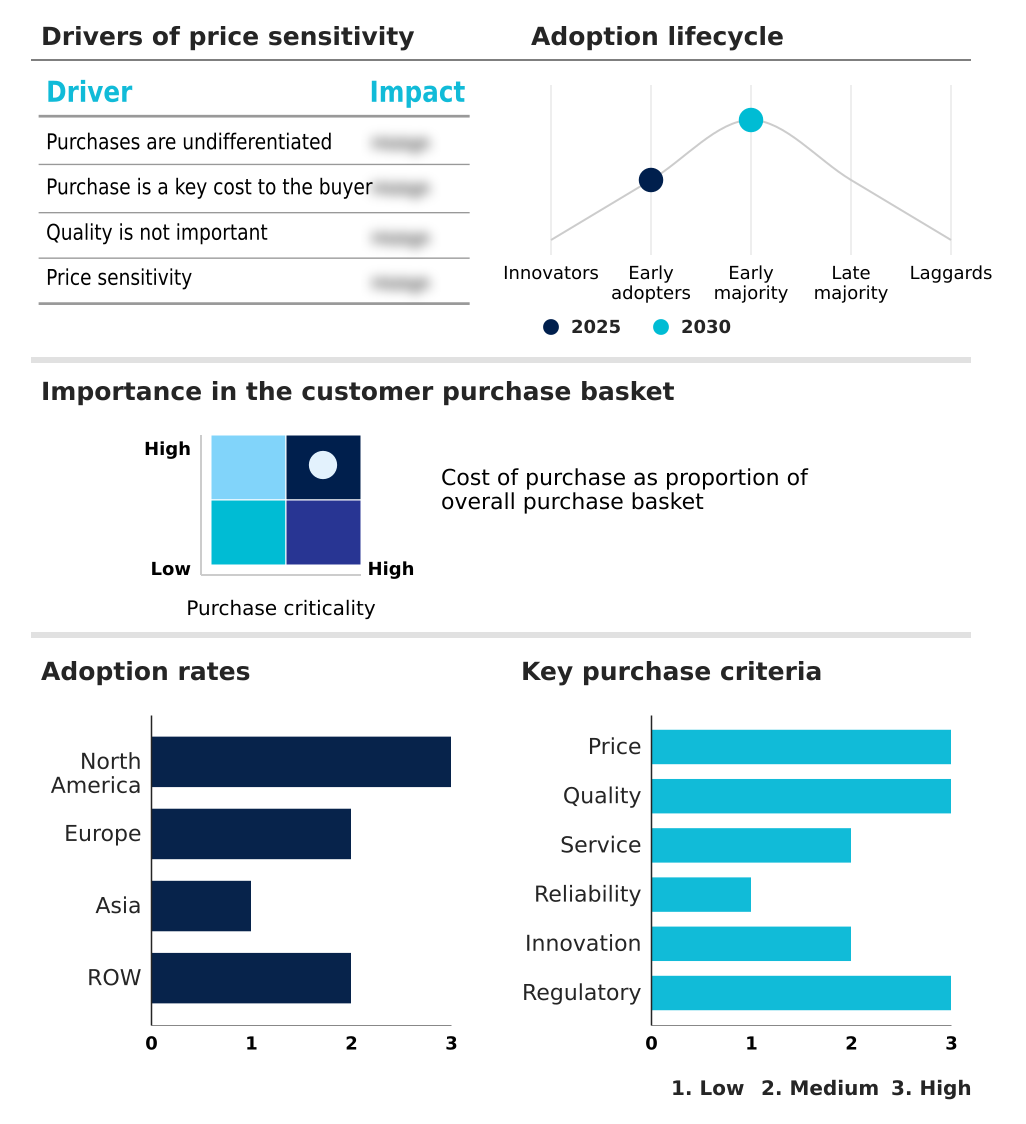
The pruritus drugs market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the pruritus drugs market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Pruritus Drugs Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, pruritus drugs market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
AbbVie Inc. - Key offerings in the pruritus drugs market are shifting toward targeted biologic therapies and specialized small-molecule inhibitors designed to address complex neuroimmune and systemic itch pathways.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- AbbVie Inc.
- Almirall SA
- Bausch Health Companies Inc.
- Bristol Myers Squibb Co.
- CSL Ltd.
- Escient Pharmaceuticals
- Galderma Group AG
- Incyte Corp.
- Kiniksa Pharmaceuticals Ltd.
- Kissei Pharmaceutical Co. Ltd.
- LEO Pharma AS
- Maruho Co. Ltd.
- Pfizer Inc.
- Regeneron Pharmaceuticals Inc.
- Sanofi SA
- Taiho Pharmaceutical Co. Ltd.
- Toray Industries Inc.
- Tvardi Therapeutics Inc.
- Vanda Pharmaceuticals Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Pruritus drugs market
- In August 2025, a comprehensive multicenter, double-blind, randomized phase three clinical trial published in The British Medical Journal demonstrated that anrikefon, an investigational intravenous kappa opioid receptor agonist, was highly safe and resulted in a noticeable reduction in severe itch intensity for patients undergoing hemodialysis.
- In October 2025, Arcutis Biotherapeutics presented compelling long-term clinical data demonstrating that Zoryve cream, a potent topical phosphodiesterase type four inhibitor, significantly reduced severe itch and sleep disturbances in patients with atopic dermatitis.
- In December 2025, Galderma announced the first patient enrollment in a groundbreaking phase two clinical study to evaluate the safety and pharmacokinetic profile of nemolizumab for treating adult patients with chronic pruritus of unknown origin.
- In February 2025, the European Commission granted widespread regulatory approval for Nemluvio, a targeted subcutaneous monoclonal antibody developed by Galderma, for the treatment of moderate-to-severe prurigo nodularis and atopic dermatitis.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Pruritus Drugs Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 307 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 7.2% |
| Market growth 2026-2030 | USD 6792.0 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 6.7% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, Japan, India, South Korea, Indonesia, Thailand, Brazil, Argentina, South Africa, UAE, Saudi Arabia, Turkey, Colombia and Egypt |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The pruritus drugs market is undergoing a fundamental transformation, driven by a deeper scientific understanding of the neuroimmune basis of chronic itch. This has catalyzed a shift from broad-spectrum corticosteroids and systemic immunosuppressants toward highly specific interventions. The current landscape is dominated by the development and commercialization of janus kinase inhibitors, monoclonal antibodies, and topical phosphodiesterase inhibitors.
- These advanced therapeutics, including interleukin-31 receptor antagonists, target the precise cytokine pathways involved in conditions like atopic dermatitis, prurigo nodularis, and chronic spontaneous urticaria.
- This focus on neuroimmune modulation and neuromodulators is also extending to systemic conditions such as cholestatic pruritus, uremic pruritus, and end-stage renal disease, where ileal bile acid transporter inhibitors and kappa opioid receptor agonists are setting new standards of care. Boardroom-level strategy is now centered on balancing the high costs of R&D with complex pharmacoeconomic evaluation and health technology assessments.
- Novel therapies are demonstrating a 50% greater improvement in itch-free days compared to legacy treatments, justifying their value amidst stringent step-therapy protocols and a competitive environment pushing for nonsteroidal topical innovations.
What are the Key Data Covered in this Pruritus Drugs Market Research and Growth Report?
-
What is the expected growth of the Pruritus Drugs Market between 2026 and 2030?
-
USD 6.79 billion, at a CAGR of 7.2%
-
-
What segmentation does the market report cover?
-
The report is segmented by Application (Hematologic pruritus, Oncological pruritus, Renal pruritus, Endocrine pruritus, and Cholestatic pruritus), Route of Administration (Oral, Topical, Injectable, and Others), End-user (Hospitals, Homecare, Specialty Clinics, and Others) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Increasing prevalence of chronic systemic and dermatological diseases, High cost of advanced biologic therapies and stringent reimbursement policies
-
-
Who are the major players in the Pruritus Drugs Market?
-
AbbVie Inc., Almirall SA, Bausch Health Companies Inc., Bristol Myers Squibb Co., CSL Ltd., Escient Pharmaceuticals, Galderma Group AG, Incyte Corp., Kiniksa Pharmaceuticals Ltd., Kissei Pharmaceutical Co. Ltd., LEO Pharma AS, Maruho Co. Ltd., Pfizer Inc., Regeneron Pharmaceuticals Inc., Sanofi SA, Taiho Pharmaceutical Co. Ltd., Toray Industries Inc., Tvardi Therapeutics Inc. and Vanda Pharmaceuticals Inc.
-
Market Research Insights
- The pruritus drugs market dynamics are shaped by a strategic pivot toward innovative delivery systems and specialized treatments that improve patient outcomes. The adoption of transdermal drug delivery and other novel administration routes improves bioavailability, with some formulations achieving a 20% faster onset of symptomatic relief compared to conventional methods.
- Development of disease-modifying therapies is critical, as they target the underlying neuroimmune pathways of the itch-scratch cycle, demonstrating a 40% greater reduction in flare-ups over a six-month period. Firms are increasingly focused on creating robust real-world evidence to support clinical practice guidelines and secure favorable positioning in specialty pharmacy tiers.
- This evidence-based approach helps navigate complex managed care strategies and prior authorization requirements, ultimately accelerating patient access to breakthrough treatments.
We can help! Our analysts can customize this pruritus drugs market research report to meet your requirements.

 RIA -
RIA - 
