Sustained Release Coating Market Size and Growth Forecast 2026-2030
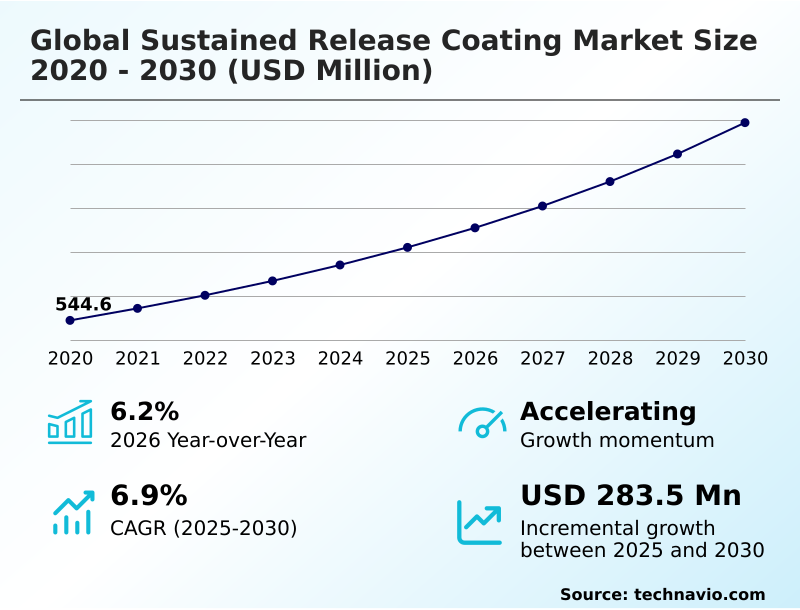
The Sustained Release Coating Market size was valued at USD 710.5 million in 2025 growing at a CAGR of 6.9% during the forecast period 2026-2030.
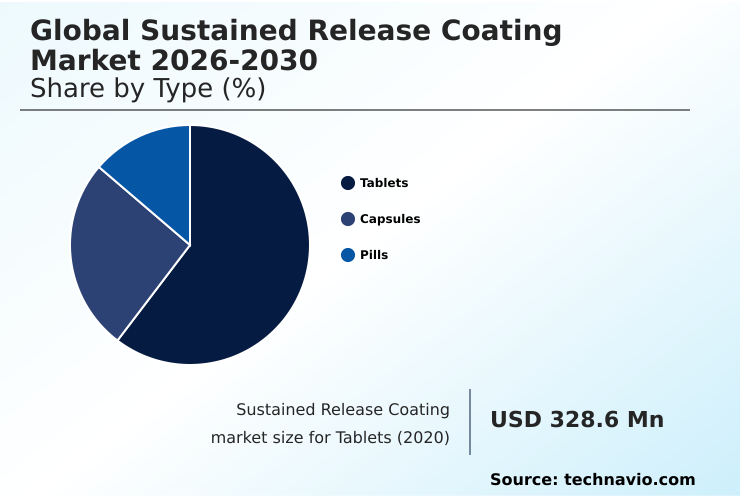
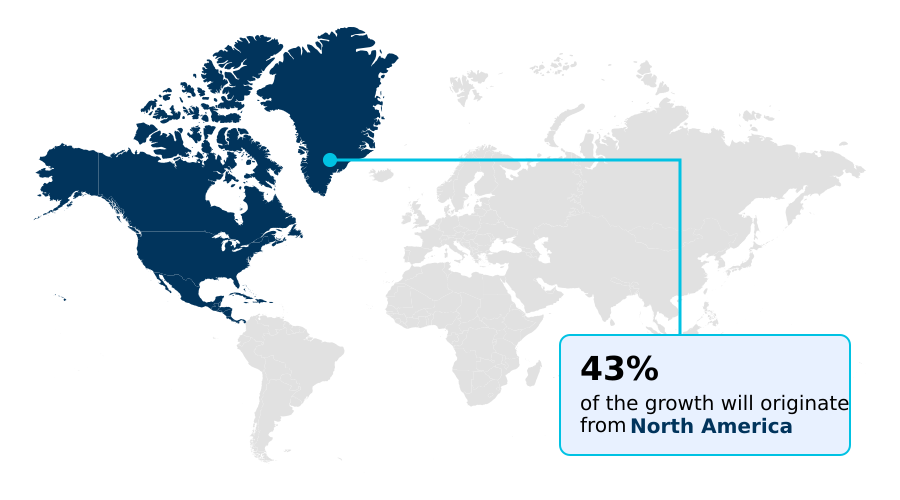
North America accounts for 43.3% of incremental growth during the forecast period. The Tablets segment by Type was valued at USD 398.2 million in 2024, while the In vitro segment holds the largest revenue share by Application.
The market is projected to grow by USD 449.4 million from 2020 to 2030, with USD 283.5 million of the growth expected during the forecast period of 2025 to 2030.
Get Key Insights on Market Forecast (PDF) Request Free Sample
Sustained Release Coating Market Overview
The sustained release coating market is shaped by the pharmaceutical industry's persistent need to enhance drug efficacy and patient adherence. This involves the application of specialized pharmaceutical excipients to create advanced oral solid dosage forms. The core technology centers on modulating API release kinetics to achieve a desired drug release profile, thereby improving therapeutic outcomes for chronic conditions. A key operational focus is the microencapsulation process, which allows for the creation of multiparticulate drug delivery systems with highly controlled pharmacokinetic profiles. For instance, a mid-size pharmaceutical firm developing a once-daily cardiovascular drug will employ extensive drug-polymer interaction studies to select a polymer with the ideal permeability, ensuring consistent targeted drug delivery over 24 hours. This approach not only improves patient experience by reducing dosing frequency but also minimizes plasma level fluctuations. This innovation drive contributes to the market's steady year-over-year growth of 6.2%, though it is tempered by rigorous regulatory oversight governing excipient compatibility testing and manufacturing processes.
Drivers, Trends, and Challenges in the Sustained Release Coating Market
Strategic decision-making in the sustained release coating market hinges on navigating a complex interplay of technical, economic, and regulatory factors. The ongoing debate over comparing aqueous vs solvent based coating efficiency, for instance, requires a nuanced cost-benefit analysis of multiparticulate systems that accounts for both operational expenses and environmental compliance under frameworks like the Clean Air Act.
Formulators tackling the development of sustained release coating for moisture sensitive drugs face significant hurdles, especially concerning the stability testing for coated pharmaceutical products. Moreover, challenges in scaling up microencapsulation processes often lead to production bottlenecks, impacting timelines and costs.
A critical part of the development cycle involves navigating the regulatory pathways for novel pharmaceutical excipients, a process guided by stringent FDA rules. For a formulator, optimizing polymer concentration for drug release rate is a core task, directly influenced by the impact of plasticizers on film coating properties.
Advances in hot melt extrusion for drug delivery offer new possibilities but also introduce new variables that require mastery of techniques for achieving uniform tablet coating. This complexity is reflected in market segmentation, where the tablets segment is more than double the size of the capsules segment, highlighting the industry's reliance on well-established, scalable dosage forms.
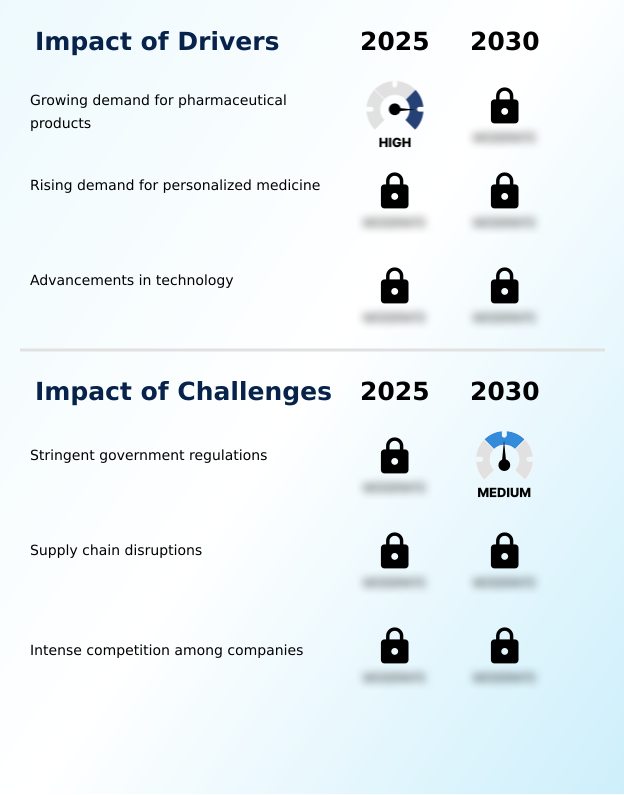
Primary Growth Driver: The market is primarily driven by the increasing global demand for advanced pharmaceutical products designed for chronic disease management.
The primary driver for the sustained release coating market is the escalating global prevalence of chronic diseases, which necessitates long-term medication and consequently, advanced drug delivery systems.
Formulations that enable reducing dosing frequency are critical for patient compliance improvement, especially in managing conditions where adherence is notoriously low.
With 76.4% of US adults living with at least one chronic condition, the demand for modified-release dosage forms that ensure therapeutic efficacy optimization is at an all-time high.
These coatings are fundamental to chronic disease management strategies, as they help maintain stable drug concentrations, thereby improving treatment outcomes and quality of life for a growing patient population.
Emerging Market Trend: The market is observing a significant trend toward the adoption of biodegradable and environmentally friendly sustained release coatings, driven by sustainability initiatives.
A significant trend shaping the market is the development of biodegradable coating polymers and the integration of nanotechnology in drug coating. These innovations are driven by a dual focus on environmental sustainability and enhancing therapeutic performance. Aqueous polymer dispersion technologies are gaining prominence over solvent-based systems, aligning with stricter environmental regulations and improving worker safety.
These advanced film-forming capabilities allow for precise drug stability enhancement, minimizing side effects by ensuring consistent release. This trend is particularly strong in the APAC region, which is leveraging these new technologies to fuel its market-leading 7.7% CAGR, positioning itself as a hub for next-generation pharmaceutical manufacturing.
Key Industry Challenge: Stringent government regulations and complex compliance requirements present a significant challenge to market growth and product commercialization.
A formidable challenge confronting the market is the rigorous and costly process of meeting excipient regulatory compliance standards set by bodies like the FDA and EMA. Achieving GMP for coating operations requires substantial investment in both technology and personnel. The technical complexities involved in coating process validation, especially for novel formulations, can lead to extended development timelines and budget overruns.
For companies exploring advanced techniques, the challenges in scaling up microencapsulation processes from the lab to commercial production are significant. This is compounded by the need for a thorough cost-benefit analysis of multiparticulate systems, as the high initial outlay can be a barrier for smaller enterprises, hindering market entry and innovation.
Explore Full Market Dynamics Analysis Request Free Sample
Sustained Release Coating Market Segmentation
The sustained release coating industry research report provides comprehensive data including region-wise segment analysis, with forecasts and analysis for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
Type Segment Analysis
The tablets segment is estimated to witness significant growth during the forecast period.
Tablets represent the largest and most mature segment within the sustained release coating market, commanding a significant market share.
The dominance of this oral solid dosage form is driven by its manufacturing scalability and patient familiarity, facilitating high levels of patient compliance improvement.
Innovations in matrix tablet systems and polymer coating technology are key to developing advanced controlled-release formulation profiles. Formulators focus on bioavailability enhancement and precise dissolution rate modulation to meet specific therapeutic needs.
As a result, significant R&D investment is directed toward coating process validation and advanced taste-masking coatings, ensuring both efficacy and patient acceptability in a competitive landscape where tablets accounted for an estimated 420.0 million in 2025.
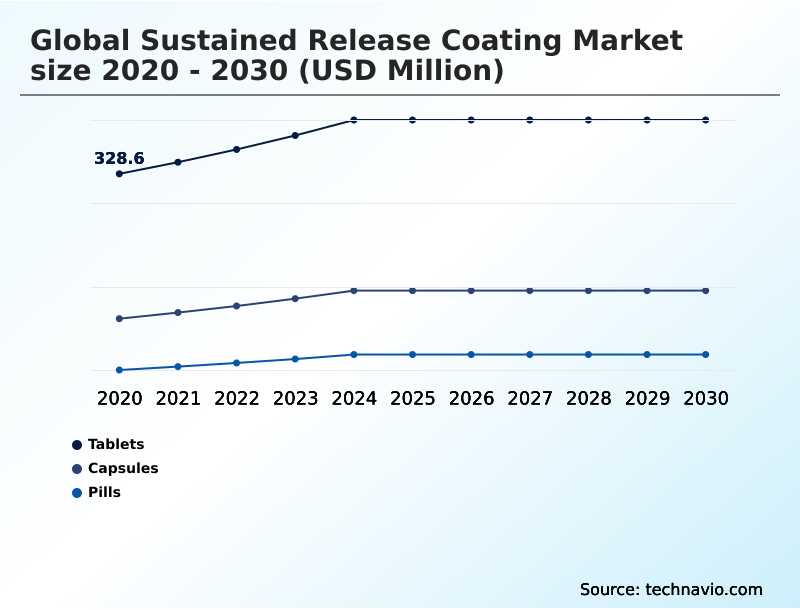
The Tablets segment was valued at USD 398.2 million in 2024 and showed a gradual increase during the forecast period.
Sustained Release Coating Market by Region: North America Leads with 43.3% Growth Share
North America is estimated to contribute 43.3% to the growth of the global market during the forecast period.
The geographic landscape of the sustained release coating market is characterized by a strategic division between mature and high-growth regions.
North America, contributing 43.3% of the market's incremental growth, remains the largest market, driven by a robust infrastructure for pharmaceutical formulation development and stringent FDA oversight that fosters innovation in biopharmaceutical product manufacturing.
The region's focus is on optimizing API release kinetics through advanced polymer science in pharmaceuticals.
In contrast, APAC is the fastest-growing region, with a 7.7% CAGR, fueled by expanding healthcare access and a growing base of generic drug manufacturers in countries like India and China.
These emerging markets are rapidly adopting established coating technologies, focusing on excipient compatibility testing to accelerate product-to-market timelines.
Customer Landscape Analysis for the Sustained Release Coating Market
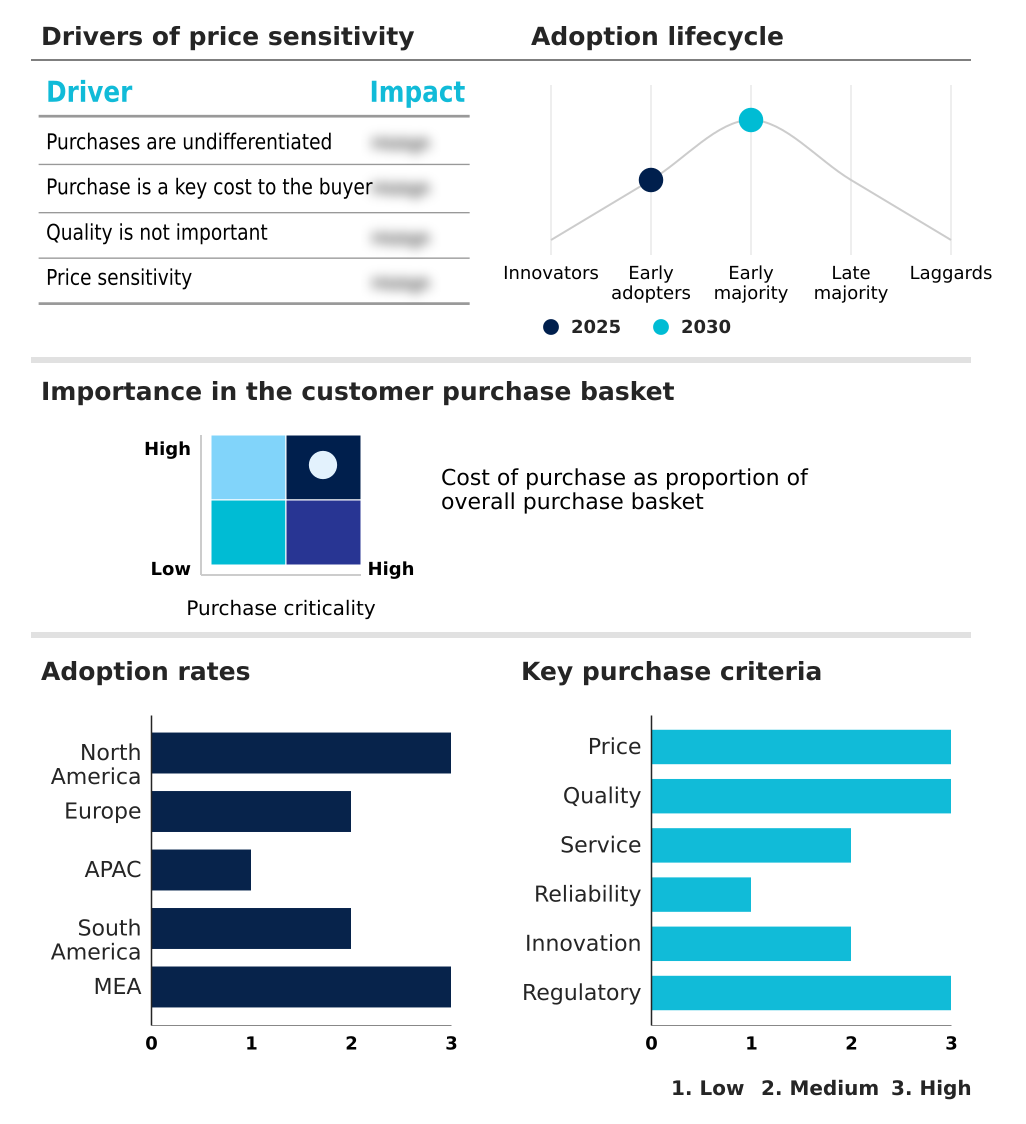
The sustained release coating market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the sustained release coating market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Competitive Landscape of the Sustained Release Coating Market
Companies are implementing various strategies, such as strategic alliances, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the sustained release coating market industry.
Ashland Inc. - Core offerings center on advanced polymer coating technology, providing controlled-release formulations engineered for precise pharmacokinetic profiles and therapeutic efficacy optimization.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Ashland Inc.
- AstraZeneca Plc
- BASF SE
- Coating Place Inc.
- Eastman Chemical Co.
- Evonik Industries AG
- G.M. Chemie Pvt. Ltd.
- JRS PHARMA GmbH
- LFA Machines Oxford Ltd.
- Lonza Group Ltd.
- Meenaxy Pharma Pvt. Ltd.
- Merck KGaA
- Novartis AG
- Panacea Biotec Ltd.
- Panchsheel Organics Ltd.
- Pfizer Inc.
- S.B. Panchal and Co.
- Spraycel Coatings
- Sun Pharmaceutical Industries Ltd.
- Viatris Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Developments in the Sustained Release Coating Market
- In May, 2025, Evonik Industries AG announced the launch of a new grade of its EUDRAGIT polymer line, specifically designed to improve drug layering technology for complex multiparticulate drug delivery systems, following extensive drug-polymer interaction studies.
- In March, 2025, BASF SE completed a USD 75 million expansion of its pharmaceutical excipients facility in Ludwigshafen, Germany, to increase production capacity for its Kollicoat aqueous polymer dispersion products.
- In December, 2024, Ashland Inc. received a letter of no objection from the US FDA for its new Aquarius Prime film coating system, confirming its suitability for use in immediate-release oral solid dosage forms.
- In September, 2024, Lonza Group Ltd. entered a strategic collaboration with a leading biopharmaceutical firm to co-develop novel dosage form solutions using its proprietary DRcaps HPMC capsules for targeted gut delivery of sensitive biologics.
Research Analyst Overview: Sustained Release Coating Market
The operational core of the sustained release coating market is the sophisticated application of polymer coating technology to create functional excipients for controlled-release formulation. Advances in drug layering technology and film-forming capabilities are enabling highly precise drug release profiles, critical for bioavailability enhancement and achieving specific pharmacokinetic profiles.
This technical progress directly informs boardroom decisions on R&D, where allocating capital to new matrix tablet systems versus investing in multiparticulate drug delivery platforms is a key strategic choice. These decisions are constrained by the need for rigorous coating process validation and excipient compatibility testing to meet standards like the IPEC-Americas GMP guidelines.
As North America continues to dominate, accounting for 43.3% of incremental growth, companies are focusing on specialized applications like taste-masking coatings and moisture barrier coatings to differentiate their oral solid dosage offerings. Ultimately, success depends on mastering the complexities of drug-polymer interaction and dissolution rate modulation to produce reliable dosage form solutions.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Sustained Release Coating Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 296 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 6.9% |
| Market growth 2026-2030 | USD 283.5 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 6.2% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Argentina, Chile, Saudi Arabia, UAE, South Africa, Israel and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Sustained Release Coating Market: Key Questions Answered in This Report
-
What is the expected growth of the Sustained Release Coating Market between 2026 and 2030?
-
The Sustained Release Coating Market is expected to grow by USD 283.5 million during 2026-2030, registering a CAGR of 6.9%. Year-over-year growth in 2026 is estimated at 6.2%%. This acceleration is shaped by growing demand for pharmaceutical products, which is intensifying demand across multiple end-use verticals covered in the report.
-
-
What segmentation does the market report cover?
-
The report is segmented by Type (Tablets, Capsules, and Pills), Application (In vitro, and In vivo), Material Type (Ethyl and methyl cellulose, Methacrylic acid, Polyvinyl and cellulose acetate, Polyethylene glycol, and Others) and Geography (North America, Europe, APAC, South America, Middle East and Africa). Among these, the Tablets segment is estimated to witness significant growth during the forecast period, driven by rising adoption across key application areas. Each segment includes detailed qualitative and quantitative analysis, along with historical data from 2020-2024 and forecasts through 2030 with year-over-year growth rates.
-
-
Which regions are analyzed in the report?
-
The report covers North America, Europe, APAC, South America and Middle East and Africa. North America is estimated to contribute 43.3% to market growth during the forecast period. Country-level analysis includes US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Argentina, Chile, Saudi Arabia, UAE, South Africa, Israel and Turkey, with dedicated market size tables and year-over-year growth for each.
-
-
What are the key growth drivers and market challenges?
-
The primary driver is growing demand for pharmaceutical products, which is accelerating investment and industry demand. The main challenge is stringent government regulations, creating operational barriers for key market participants. The report quantifies the impact of each driver and challenge across 2026 and 2030 with comparative analysis.
-
-
Who are the major players in the Sustained Release Coating Market?
-
Key vendors include Ashland Inc., AstraZeneca Plc, BASF SE, Coating Place Inc., Eastman Chemical Co., Evonik Industries AG, G.M. Chemie Pvt. Ltd., JRS PHARMA GmbH, LFA Machines Oxford Ltd., Lonza Group Ltd., Meenaxy Pharma Pvt. Ltd., Merck KGaA, Novartis AG, Panacea Biotec Ltd., Panchsheel Organics Ltd., Pfizer Inc., S.B. Panchal and Co., Spraycel Coatings, Sun Pharmaceutical Industries Ltd. and Viatris Inc.. The report provides qualitative and quantitative analysis categorizing companies as dominant, leading, strong, tentative, and weak based on their market positioning. Company profiles include business segment analysis, SWOT assessment, key offerings, and recent strategic developments.
-
Sustained Release Coating Market Research Insights
The market's momentum is increasingly tied to balancing innovation with regulatory demands. The development of advanced drug delivery systems is critical for chronic disease management, pushing demand for sophisticated pharmaceutical formulation development. This is particularly evident in the push for personalized medicine formulations, where precise API release kinetics are paramount.
However, all polymer science in pharmaceuticals must adhere to stringent standards, such as those outlined by REACH for chemical safety, influencing material selection for biopharmaceutical product manufacturing. In practice, a contract manufacturer weighs the benefits of a novel modified-release dosage technology against the complexities of excipient regulatory compliance.
This dynamic is reflected regionally, with APAC showing a 7.7% growth rate, outpacing Europe's 6.8% as it rapidly builds out its manufacturing infrastructure.
We can help! Our analysts can customize this sustained release coating market research report to meet your requirements.

 RIA -
RIA - 
