Clinical Trial Decentralization Platforms Market Size 2026-2030
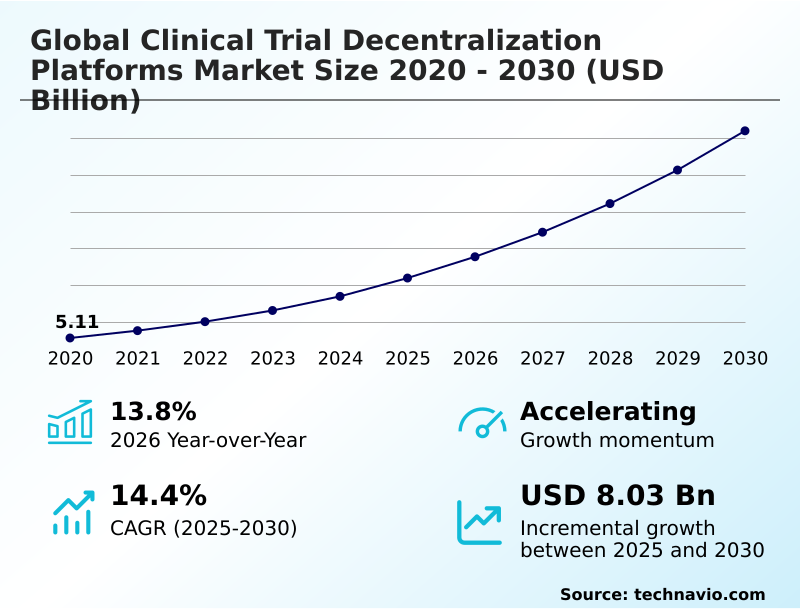
The clinical trial decentralization platforms market size is valued to increase by USD 8.03 billion, at a CAGR of 14.4% from 2025 to 2030. Enhancing patient accessibility and diversity through decentralized frameworks will drive the clinical trial decentralization platforms market.
Major Market Trends & Insights
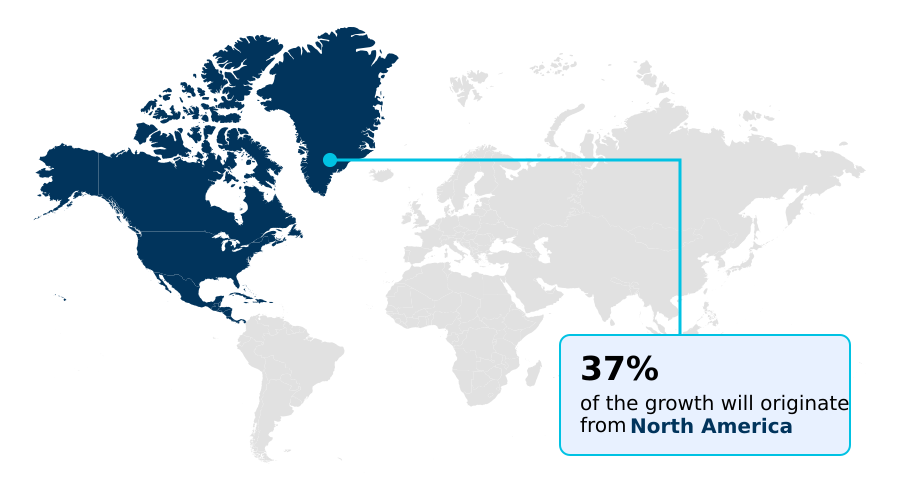
- North America dominated the market and accounted for a 37.3% growth during the forecast period.
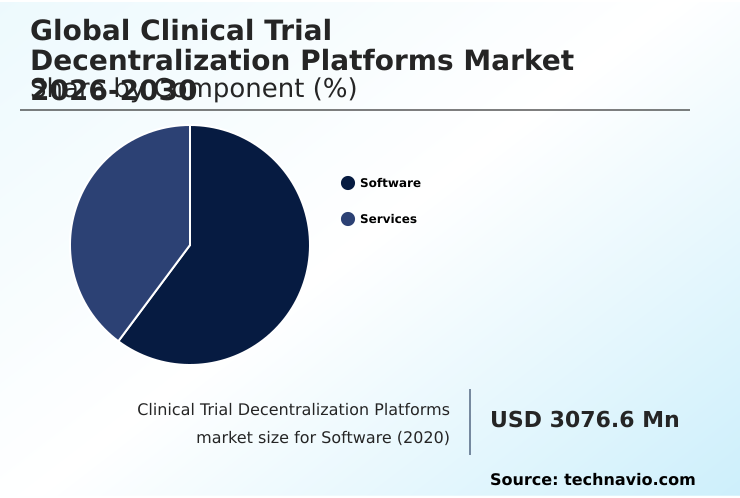
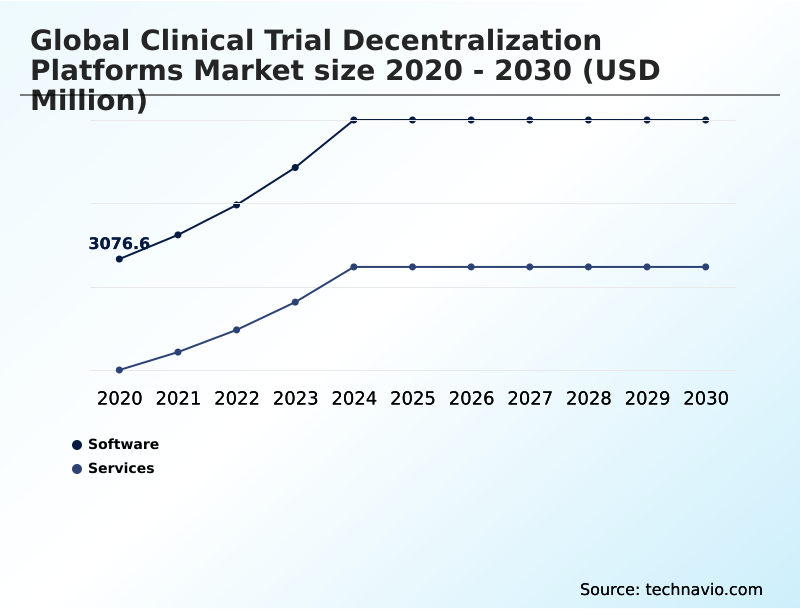
- By Component - Software segment was valued at USD 4.38 billion in 2024
- By End-user - Pharmaceutical companies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 11.30 billion
- Market Future Opportunities: USD 8.03 billion
- CAGR from 2025 to 2030 : 14.4%
Market Summary
- The clinical trial decentralization platforms market is fundamentally reshaping pharmaceutical R&D by prioritizing patient-centric trial design and operational efficiency. This shift is enabled by a suite of digital tools, including eConsent platforms for remote consenting processes and mobile health (mHealth) applications for real-world evidence (RWE) collection.
- By leveraging remote patient monitoring (RPM) and wearable sensor data, sponsors can implement continuous data collection, which is vital for digital biomarker validation and remote safety monitoring. For instance, a mid-sized biotech firm can now execute a complex, multi-national rare disease study using virtual trial platforms, thereby improving patient retention rates without the substantial capital investment required for physical sites.
- This model relies on robust decentralized data management and electronic data capture (EDC) systems to ensure data integrity. The evolution toward hybrid clinical trial models, which blend remote activities with necessary in-person visits, addresses the logistical challenges of direct-to-patient (DTP) supply chains and ensures comprehensive protocol adherence monitoring, ultimately accelerating the delivery of new therapies.
What will be the Size of the Clinical Trial Decentralization Platforms Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Clinical Trial Decentralization Platforms Market Segmented?
The clinical trial decentralization platforms industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Component
- Software
- Services
- End-user
- Pharmaceutical companies
- Contract research organizations
- Biotechnology companies
- Academic and research institutes
- Deployment
- Cloud based platforms
- Hybrid deployment
- On premises solutions
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- APAC
- China
- India
- Japan
- South America
- Brazil
- Argentina
- Colombia
- Middle East and Africa
- South Africa
- Israel
- Saudi Arabia
- Rest of World (ROW)
- North America
By Component Insights
The software segment is estimated to witness significant growth during the forecast period.
The software segment is central to enabling a patient-centric trial design that prioritizes patient accessibility improvement and participant diversity enhancement. Core to this are eConsent platforms and robust telehealth integration, which facilitate remote interactions while reducing participant burden.
Advanced electronic clinical outcome assessment (eCOA) solutions are crucial for continuous data collection, capturing everything from subjective feedback to objective wearable sensor data. The infrastructure supports extensive real-world evidence (RWE) collection and enables sophisticated digital biomarker validation.
These platforms improve data quality, with some achieving a 15% reduction in data entry errors compared to manual methods. This digital framework increasingly positions clinical research-as-a-care-option, capturing real-time physiological data to advance therapeutic development.
The Software segment was valued at USD 4.38 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 37.3% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Clinical Trial Decentralization Platforms Market Demand is Rising in North America Request Free Sample
The geographic landscape is led by North America, where a mature regulatory environment supports the use of advanced digital endpoint measurement and patient-facing technology.
This region serves as a primary market for integrated data platforms that facilitate complex remote trial orchestration and clinical operations automation. Meanwhile, the APAC region is the fastest-growing market, driven by the adoption of study-at-home models and virtual care integration.
The implementation of site-less clinical trials in this region has shown to reduce administrative overhead by up to 35%. Across all regions, a focus on risk-based quality management (RBQM) and comprehensive remote monitoring oversight is standard.
The use of telehealth-enabled trials and remote diagnostics is becoming universal, with platforms demonstrating a 20% improvement in data consistency across geographically dispersed participant groups.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic implementation of decentralized clinical trial platforms is becoming a competitive differentiator. The technology for remote patient reported outcomes is a cornerstone, particularly when combined with effective eCOA and ePRO integration strategies to capture high-quality data.
- In specialized fields, a decentralized clinical trial platform for oncology allows researchers to reach vulnerable populations, while the use of wearable sensor data for cardiovascular trials provides continuous physiological insights. Success depends on mastering operational complexities, including establishing best practices for direct-to-patient logistics and managing the investigational product in at-home trials.
- Firms must address the challenges of data privacy in BYOD trials and ensure regulatory compliance for digital biomarkers. The adoption of hybrid trial models for rare disease studies and the use of real-world evidence collection using mHealth apps are becoming standard. Key initiatives focus on improving patient retention with digital engagement tools and integrating telehealth into clinical trial protocols.
- Organizations performing a cost-benefit analysis of decentralized clinical trials find that integrated platforms can double data query resolution speed compared to siloed systems. Efficiently scaling virtual trials across different regions requires robust AI-powered patient recruitment for decentralized trials, diligent remote safety monitoring and adverse event reporting, and a sophisticated platform for remote monitoring of vital signs.
- Ultimately, success hinges on seamless data harmonization for multi-source EDC and effective clinical supply chain management for remote sites.
What are the key market drivers leading to the rise in the adoption of Clinical Trial Decentralization Platforms Industry?
- A key market driver is the enhancement of patient accessibility and diversity, which is achieved through the adoption of decentralized frameworks.
- Market growth is significantly driven by the imperative for global trial scalability and the need to generate robust regulatory submission data.
- The use of advanced patient recruitment algorithms has been shown to increase participant enrollment by over 30% in targeted populations.
- Platforms offering a seamless remote consenting process via digital informed consent are critical for improving the patient journey mapping from the outset. This digital-first approach enhances clinical data interoperability between systems.
- Mobile health (mHealth) applications are central to this paradigm, enabling remote safety monitoring and serving as a primary patient engagement platform. This facilitates better management of trial participation logistics.
- The integration of real-time data analytics into the clinical trial management system (CTMS) allows for continuous oversight and faster decision-making, which is essential for decentralized data management.
What are the market trends shaping the Clinical Trial Decentralization Platforms Industry?
- The integration of AI is emerging as a significant trend, aiming to enhance patient recruitment and improve retention rates in clinical trials.
- Key market trends are converging around the adoption of hybrid clinical trial models and fully virtual trial platforms, which rely on sophisticated electronic data capture (EDC) systems. These platforms facilitate seamless data harmonization protocols, which are essential for integrating information from disparate sources and enabling remote data verification.
- The implementation of these models has led to a 25% reduction in data cleaning timelines. Furthermore, advancements in direct-to-patient (DTP) logistics and investigational medicinal product (IMP) tracking technology are streamlining the clinical supply chain. This focus on decentralized study conduct improves patient-reported data integrity and protocol adherence monitoring.
- As a result, organizations are achieving greater operational efficiency through remote site management and virtual investigator meetings, with eSource data capture reducing source data verification needs by up to 40%.
What challenges does the Clinical Trial Decentralization Platforms Industry face during its growth?
- A key challenge affecting industry growth involves data privacy concerns and the complexities of ensuring robust cybersecurity in remote trial environments.
- A primary challenge in the market is ensuring data security in remote trials, particularly with the rise of Bring Your Own Device (BYOD) strategy. Implementing robust data privacy encryption on cloud-based clinical platforms is non-negotiable to protect electronic patient-reported outcomes (ePRO) and maintain the electronic trial master file (eTMF).
- The logistical complexities of clinical supply chain management for site-less clinical trials introduce risks that require advanced remote monitoring solutions. Moreover, ensuring successful digital health technology adoption requires comprehensive remote training and support, as poor usability can negatively impact patient-reported data integrity and efforts at improving patient retention rates.
- In fact, studies indicate that trials with inadequate connected device integration see a 20% higher participant drop-off rate compared to those with seamless tech support.
Exclusive Technavio Analysis on Customer Landscape
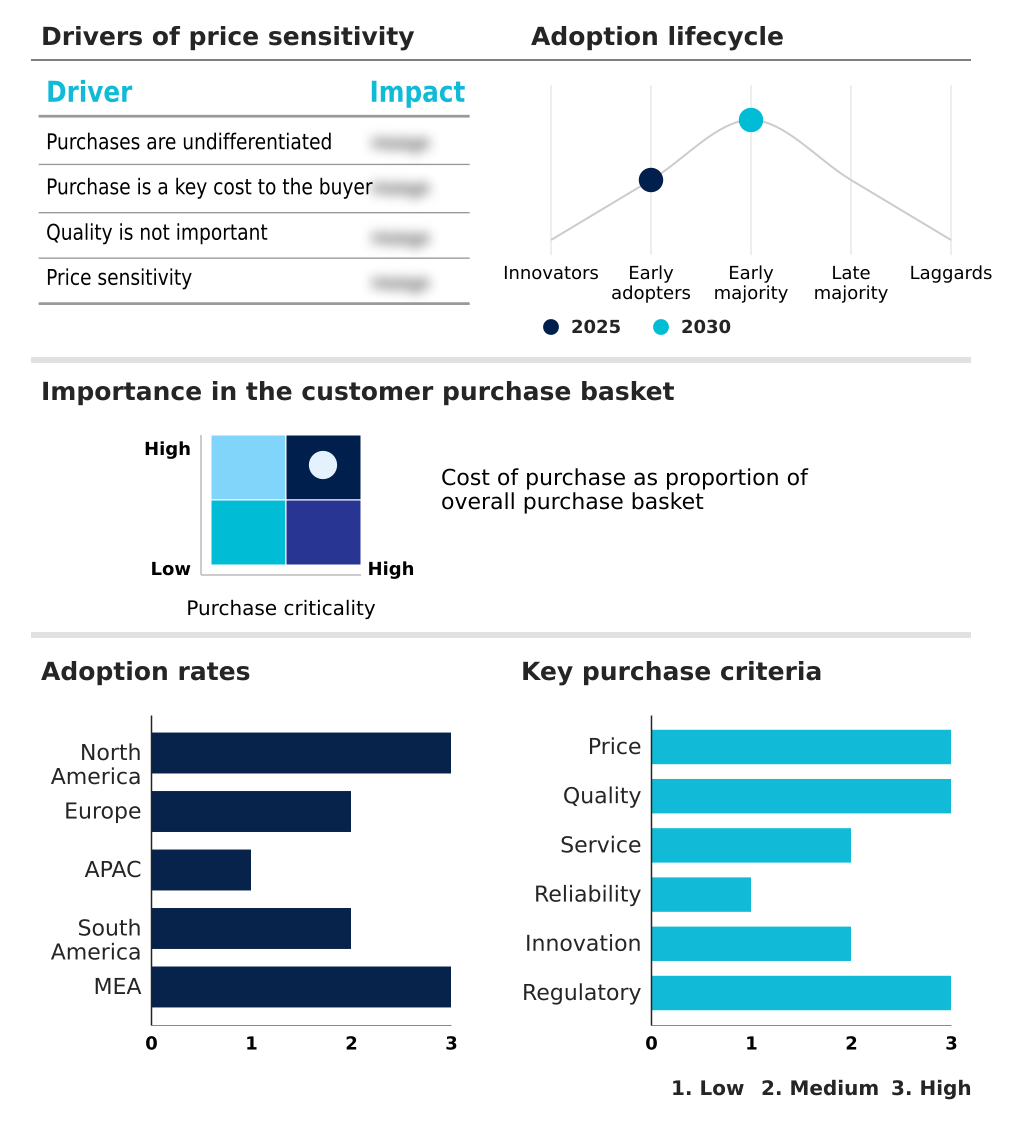
The clinical trial decentralization platforms market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the clinical trial decentralization platforms market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Clinical Trial Decentralization Platforms Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, clinical trial decentralization platforms market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Advarra Inc. - Modular software platforms are scaling remote and hybrid trials by prioritizing mobile-first patient interfaces for enhanced data collection and participant accessibility.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Advarra Inc.
- Castor Research Inc.
- Clinical Ink Inc.
- Crucial Data Solutions Inc.
- Florence Healthcare.
- ICON plc
- IQVIA Holdings Inc.
- KAYENTIS SAS
- Laboratory Corp.
- Lightship
- Medable Inc.
- Medidata
- Obvio Health USA Inc.
- Parexel International Corp.
- Reify Health
- Science 37 Inc.
- Signant Health
- Thermo Fisher Scientific Inc.
- Veeva Systems Inc.
- YPrime LLC
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Clinical trial decentralization platforms market
- In September 2024, Medable announced the launch of Agent Studio, an AI platform designed to enable life sciences companies to implement agentic AI in their clinical operations.
- In December 2024, AlphaTON Capital Corp. announced plans to introduce a decentralized, AI-driven biotech platform focusing initially on rare and immunocompromised cancers.
- In March 2025, the European Medicines Agency launched a new digital toolkit designed to assist trial sponsors in navigating the regulatory requirements for decentralized data collection across member states.
- In April 2025, a consortium of major pharmaceutical companies and software developers finalized a new universal data standard for decentralized platforms, ensuring compatibility between remote monitoring sensors and clinical databases.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Clinical Trial Decentralization Platforms Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 293 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 14.4% |
| Market growth 2026-2030 | USD 8026.0 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 13.8% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, The Netherlands, Spain, China, India, Japan, Australia, South Korea, Indonesia, Brazil, Argentina, Colombia, South Africa, Israel, Saudi Arabia, UAE and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The market's evolution is defined by the convergence of sophisticated software and patient-centric services. An integrated technology stack, including eConsent platforms, remote patient monitoring (RPM), and electronic clinical outcome assessment (eCOA), is now the standard for enabling hybrid clinical trial models and full virtual trial platforms.
- The shift toward decentralized data management, supported by robust electronic data capture (EDC) systems and eSource data capture, is driven by the need for real-time data analytics. This trend directly impacts boardroom decisions regarding capital expenditure, as investment shifts from physical site infrastructure to scalable, cloud-based clinical platforms.
- For example, firms adopting comprehensive remote monitoring solutions have achieved a 30% reduction in site visit costs. Key components such as digital informed consent, mobile health (mHealth) applications, and patient engagement platforms are critical for a successful patient-centric trial design.
- The entire ecosystem, from investigational medicinal product (IMP) tracking and direct-to-patient (DTP) logistics to the electronic trial master file (eTMF) and clinical trial management system (CTMS), is being optimized for remote trial orchestration and clinical operations automation.
What are the Key Data Covered in this Clinical Trial Decentralization Platforms Market Research and Growth Report?
-
What is the expected growth of the Clinical Trial Decentralization Platforms Market between 2026 and 2030?
-
USD 8.03 billion, at a CAGR of 14.4%
-
-
What segmentation does the market report cover?
-
The report is segmented by Component (Software, and Services), End-user (Pharmaceutical companies, Contract research organizations, Biotechnology companies, and Academic and research institutes), Deployment (Cloud based platforms, Hybrid deployment, and On premises solutions) and Geography (North America, Europe, APAC, South America, Middle East and Africa)
-
-
Which regions are analyzed in the report?
-
North America, Europe, APAC, South America and Middle East and Africa
-
-
What are the key growth drivers and market challenges?
-
Enhancing patient accessibility and diversity through decentralized frameworks, Data privacy concerns and complexity of cybersecurity in remote environments
-
-
Who are the major players in the Clinical Trial Decentralization Platforms Market?
-
Advarra Inc., Castor Research Inc., Clinical Ink Inc., Crucial Data Solutions Inc., Florence Healthcare., ICON plc, IQVIA Holdings Inc., KAYENTIS SAS, Laboratory Corp., Lightship, Medable Inc., Medidata, Obvio Health USA Inc., Parexel International Corp., Reify Health, Science 37 Inc., Signant Health, Thermo Fisher Scientific Inc., Veeva Systems Inc. and YPrime LLC
-
Market Research Insights
- The market is defined by a strategic move toward improving patient retention rates and enhancing participant diversity, directly influencing study outcomes. The adoption of digital health technology facilitates a decentralized study conduct, with some sponsors reporting a 25% reduction in patient dropout rates compared to traditional site-based models.
- Furthermore, the integration of remote diagnostics and telehealth-enabled trials streamlines trial participation logistics, contributing to a 15% faster recruitment timeline. This shift is not merely technological but also operational, as it enables remote site management and virtual investigator meetings, which are critical for global trial scalability.
- The focus on a patient-centric data collection model, where the patient journey mapping is central, ensures that the remote consenting process and subsequent data submission are seamless, supporting the broader goal of making clinical research-as-a-care-option a practical reality.
We can help! Our analysts can customize this clinical trial decentralization platforms market research report to meet your requirements.


 RIA -
RIA - 
