Gene Panel Market Size 2026-2030
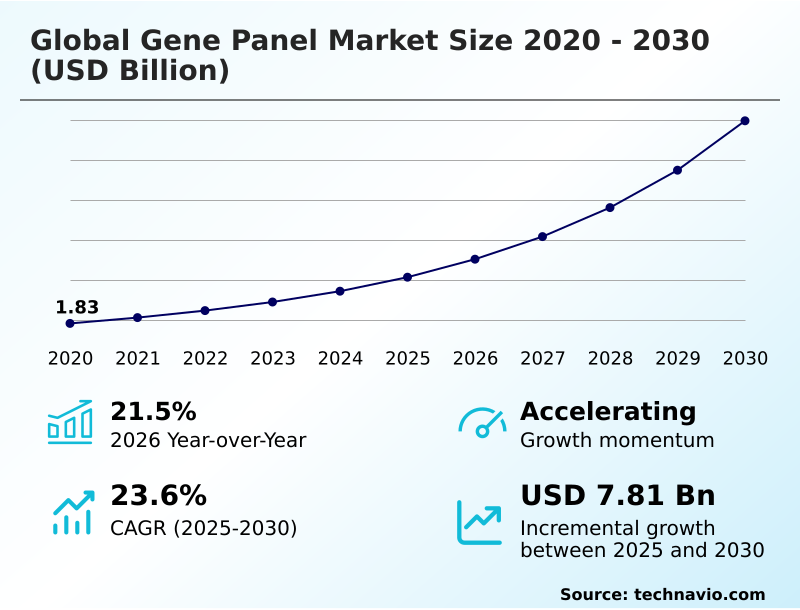
The gene panel market size is valued to increase by USD 7.81 billion, at a CAGR of 23.6% from 2025 to 2030. Ascendancy of precision oncology and proliferation of targeted therapies will drive the gene panel market.
Major Market Trends & Insights
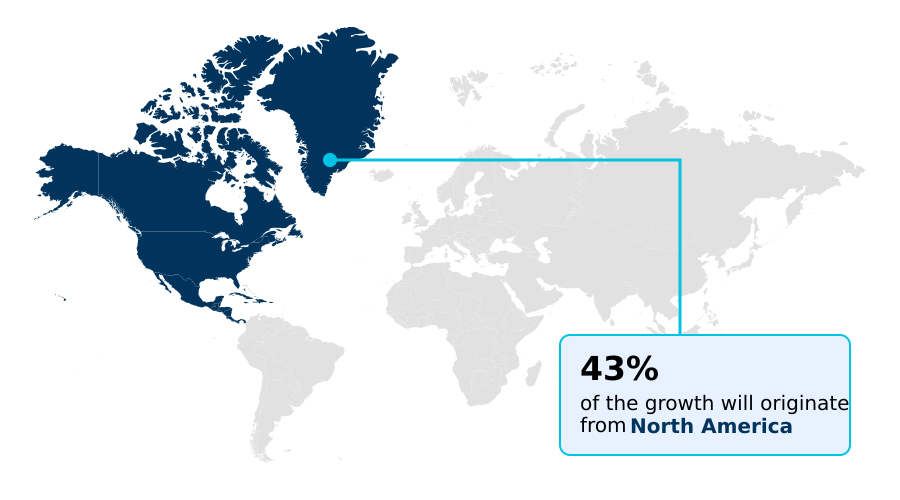
- North America dominated the market and accounted for a 42.6% growth during the forecast period.
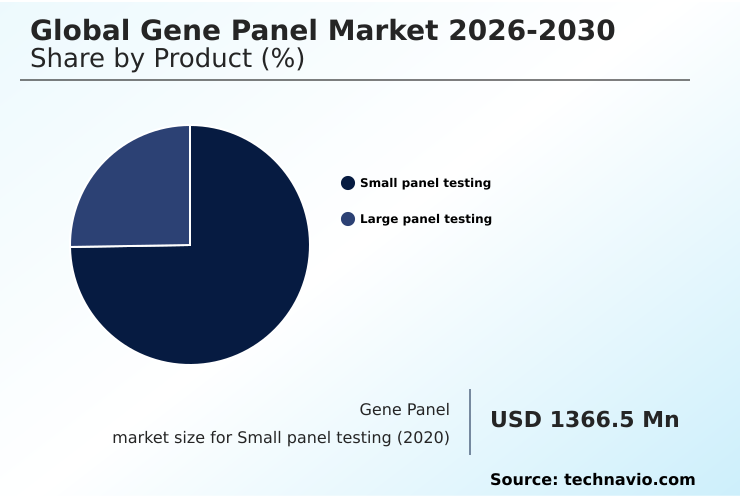
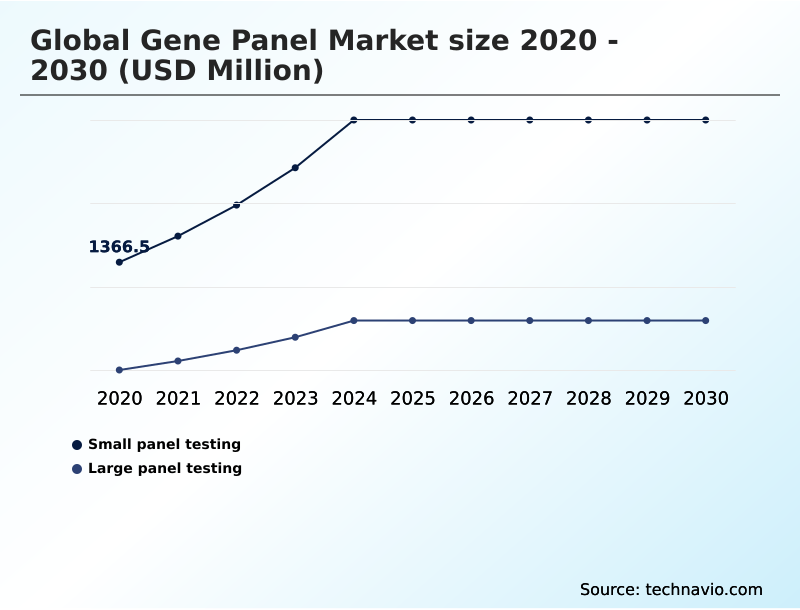
- By Product - Small panel testing segment was valued at USD 2.56 billion in 2024
- By Technique - Amplicon segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 10.13 billion
- Market Future Opportunities: USD 7.81 billion
- CAGR from 2025 to 2030 : 23.6%
Market Summary
- The gene panel market is characterized by rapid technological evolution, fundamentally altering clinical diagnostics and personalized medicine. Growth is fueled by the increasing adoption of next-generation sequencing, which has drastically lowered sequencing costs and improved the diagnostic yield from multi-gene panel testing. This shift enables broader applications, from precision oncology biomarker identification to hereditary disease diagnostics.
- A primary trend is the move toward comprehensive genomic profiling and liquid biopsy assays, which provide a more holistic view of a patient's molecular landscape. For instance, a hospital network can implement a standardized sample-to-report workflow for in-house gene panel testing, reducing reliance on external labs and cutting turnaround time by up to 40%, thereby accelerating treatment decisions.
- However, the industry faces challenges related to the complexity of data interpretation, particularly with variants of uncertain significance, and navigating the fragmented regulatory approval pathway and reimbursement landscape, which can hinder the adoption of novel tests.
What will be the Size of the Gene Panel Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Gene Panel Market Segmented?
The gene panel industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Product
- Small panel testing
- Large panel testing
- Technique
- Amplicon
- Hybridization
- Application
- Oncology
- Genetic disorders
- Pharmacogenomics
- Others
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Product Insights
The small panel testing segment is estimated to witness significant growth during the forecast period.
The small panel testing segment is foundational to the gene panel market, offering a focused, hypothesis-driven approach.
These panels, designed for rapid and cost-effective analysis, are crucial for routine diagnostics where a defined set of genes informs a specific medical question, such as targeted therapy selection.
This targeted methodology simplifies the bioinformatics pipeline and reduces turnaround time, a key factor in settings like decentralized hospital laboratory operations.
By concentrating on fewer than one hundred genes, these tests improve diagnostic yield for specific conditions and minimize variants of uncertain significance.
This streamlined approach, leveraging techniques like polymerase chain reaction, has been shown to improve the efficiency of patient stratification by over 25%.
This focus on efficiency and clear, actionable results supports broader adoption in clinical workflows and population health screening initiatives.
The Small panel testing segment was valued at USD 2.56 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
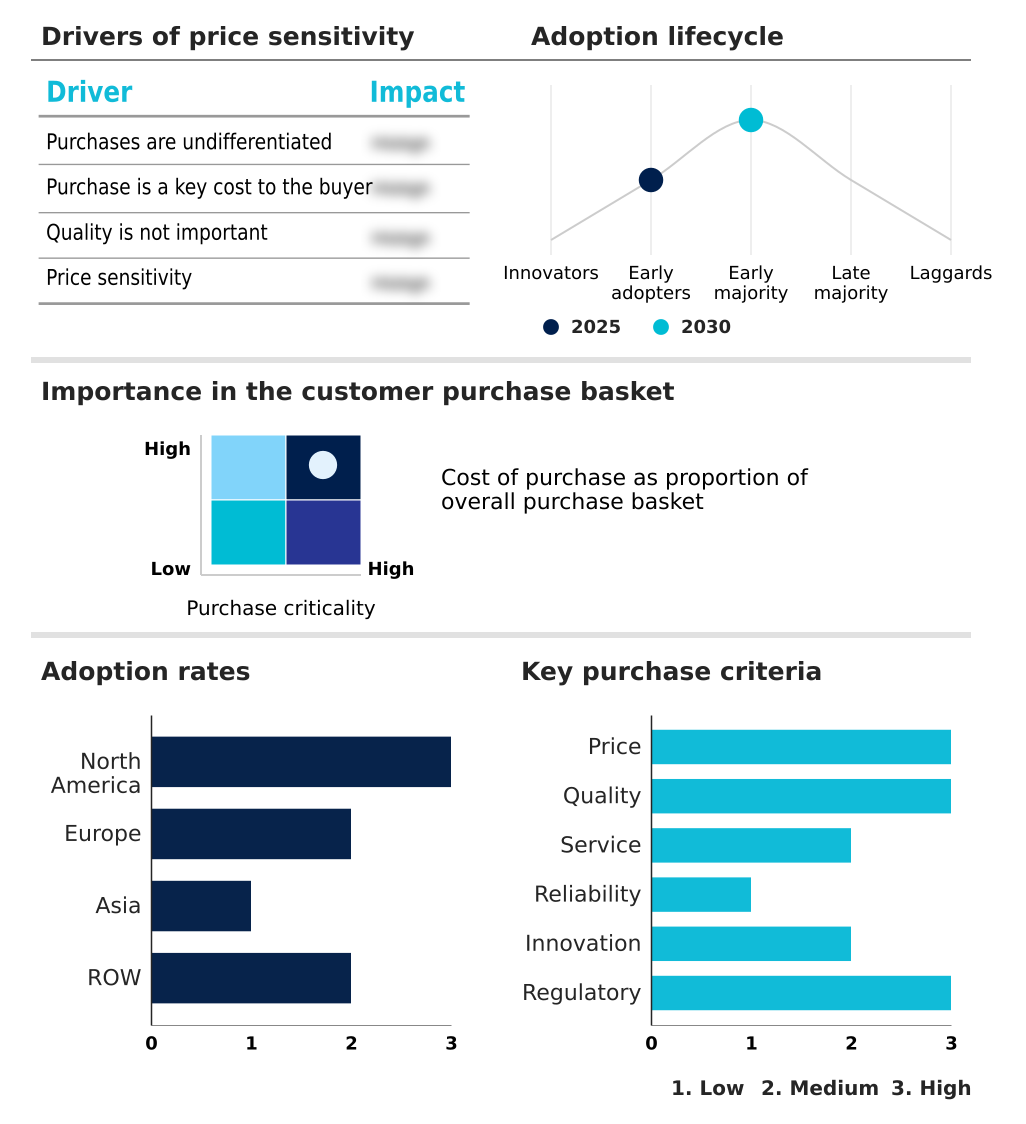
North America is estimated to contribute 42.6% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Gene Panel Market Demand is Rising in North America Request Free Sample
The geographic landscape of the gene panel market is led by North America, where a sophisticated R&D ecosystem and high adoption rates of next-generation sequencing sustain market leadership.
Europe is a mature market, with national initiatives promoting the integration of genomic medicine services and evidence-based medicine.
The Asia-Pacific region is experiencing the fastest growth, driven by government investment in precision medicine and a rising middle class demanding advanced healthcare, which has led to a 20% year-over-year increase in a decentralized hospital laboratory adopting in-house gene panel testing.
Key applications like hereditary cancer screening and Mendelian disorder diagnosis are gaining traction globally.
However, adoption in emerging markets is often challenged by the need for robust test validation guidelines and more favorable reimbursement landscapes to justify the cost-effectiveness analysis required by local health authorities.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the gene panel market requires a deep understanding of competing technologies and evolving clinical needs. A central consideration is the debate between whole exome sequencing vs gene panel testing, where factors like diagnostic yield and cost-effectiveness must be balanced.
- For many routine applications, small gene panels in routine diagnostics remain highly effective, while large gene panels for genomic profiling are essential for complex cases. The rise of ai-powered custom gene panel design is enabling unprecedented specificity, particularly for custom gene panel design for oncology and panels for hereditary cardiac conditions.
- This trend complements the increasing use of liquid biopsy for early cancer detection and multi-omics analysis in cancer profiling. The clinical utility of multi-gene panels is now well-established, with a critical role of panels in drug development and companion diagnostic development for therapies.
- However, significant operational challenges persist, including the challenges in vus interpretation and the need for standardizing gene panel test reporting. The industry must also navigate logistical hurdles like improving the turnaround time for gene panel results while ensuring rigorous analytical validation of ngs panels.
- A major pharmaceutical firm reported that optimizing its companion diagnostic workflow reduced patient screening times by over 35%, directly impacting clinical trial enrollment speed.
- Addressing reimbursement policies for genomic testing and overcoming regulatory hurdles for gene panel tests are paramount, as is demonstrating the cost-effectiveness of pharmacogenomic panels to secure broader market access, especially for applications like gene panels for neurological disorders. The choice between pan-cancer panels vs organ-specific panels further complicates portfolio strategy, demanding a nuanced approach to market positioning.
What are the key market drivers leading to the rise in the adoption of Gene Panel Industry?
- The market's growth is primarily driven by the ascendancy of precision oncology and the corresponding proliferation of targeted therapies that require genomic profiling.
- Market growth is fundamentally driven by the expanding clinical utility of genomic information in guiding treatment decisions.
- The proliferation of targeted therapies has made companion diagnostics an essential component of the drug development process, with precision oncology biomarkers now guiding therapy selection in over 60% of new cancer drug approvals.
- This has solidified the role of multi-gene panel testing as a standard of care. The ability to perform somatic mutation detection and identify actionable biomarkers from a single test significantly improves efficiency over sequential testing.
- Furthermore, applications in non-oncology areas such as rare disease diagnostics are accelerating, driven by the goal of diagnostic odyssey reduction.
- The adoption of these panels in clinical trial objectives for patient stratification is now standard practice, improving trial outcomes and accelerating therapeutic approvals.
What are the market trends shaping the Gene Panel Industry?
- A key market trend involves the convergence of pan-cancer profiling with multi-omics analysis. This approach integrates transcriptomics and proteomics to provide a more holistic view of tumor biology.
- Key market trends are reshaping the application of genomic profiling, moving toward more comprehensive and integrated approaches. The convergence of pan-cancer genomic profiling with RNA expression analysis is a significant development, enabling a more holistic view of tumor biology and improving gene fusion detection.
- This trend is amplified by the rise of liquid biopsy assays, which analyze circulating tumor dna for non-invasive treatment monitoring and early cancer detection. The use of advanced in-silico design and AI-powered platforms for creating custom panels has reduced development cycles by up to 50%.
- This allows for rapid deployment in niche areas like neurological disorder panels and proactive health screening, driving the personalization of medicine beyond oncology. These advancements are pushing the boundaries of what is possible in diagnostics.
What challenges does the Gene Panel Industry face during its growth?
- A significant challenge affecting industry growth is the complex and fragmented regulatory and reimbursement landscape, which creates barriers to market entry and adoption.
- The market faces significant hurdles related to data complexity and economic validation, which can slow adoption despite technological advancements. A primary issue is data interpretation complexity, particularly the high rate of variants of uncertain significance (VUS) returned by large panels, which complicates clinical decision-making.
- The reporting of a VUS occurs in up to 40% of comprehensive tests, creating challenges for genetic counseling. Another major restraint is the intricate regulatory approval pathway, which demands extensive evidence of clinical utility and can be a lengthy process.
- Demonstrating value through rigorous health technology assessment and securing favorable reimbursement policies remain critical challenges, as payers increasingly demand proof that these tests lead to better patient outcomes and provide system-wide value, often requiring a 15-20% improvement in outcomes to justify coverage.
Exclusive Technavio Analysis on Customer Landscape
The gene panel market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the gene panel market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Gene Panel Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, gene panel market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Agilent Technologies Inc. - Delivers high-sensitivity custom NGS panels for precise and accurate genomic analysis, supporting targeted research and diagnostics with reliable results.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Agilent Technologies Inc.
- Azenta Inc.
- BGI Group
- Centogene N.V.
- Color Health Inc.
- Danaher Corp.
- Eurofins Scientific SE
- F. Hoffmann La Roche Ltd.
- Fagron NV
- GenXPro GmbH
- Illumina Inc.
- MedGenome Labs Ltd.
- MGZ Medical Genetics Center
- Novogene Co. Ltd.
- OPKO Health Inc.
- Oxford Nanopore Technologies
- QIAGEN N.V.
- Quest Diagnostics Inc.
- Thermo Fisher Scientific Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Gene panel market
- In August 2025, The U.S. Food and Drug Administration approved a pan-cancer gene panel as a universal companion diagnostic, streamlining patient access to targeted therapies across all solid tumor types.
- In April 2025, The United States Food and Drug Administration granted breakthrough designation to a liquid biopsy assay that uses multi-omic analysis for early cancer detection in high-risk individuals.
- In March 2025, Agilent Technologies launched its ureSelect AI design wizard, an AI-powered cloud platform that enables researchers to rapidly design and order optimized custom gene panels.
- In May 2025, The European Union's updated In Vitro Diagnostic Regulation (IVDR) fully took effect, increasing clinical evidence requirements and regulatory scrutiny for genetic test manufacturers.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Gene Panel Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 293 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 23.6% |
| Market growth 2026-2030 | USD 7814.1 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 21.5% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, India, Japan, South Korea, Indonesia, Thailand, Brazil, Saudi Arabia, UAE, Turkey, Argentina, Colombia, South Africa and Israel |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The gene panel market's trajectory is defined by continuous innovation in genomic technologies and their expanding clinical applications. The foundation of this market rests on next-generation sequencing, which has driven significant sequencing cost reduction and enabled sophisticated multi-gene panel testing.
- Core technologies like polymerase chain reaction and advanced target enrichment chemistry, including both amplicon-based chemistry and hybrid capture technology, are central to the development of these panels. Progress in the field is increasingly dependent on the efficiency of the entire sample-to-report workflow, from handling formalin-fixed paraffin-embedded tissue to analysis via robust bioinformatics pipelines.
- For boardroom strategy, a key focus is navigating the complex reimbursement landscape. The decision to invest in developing a new companion diagnostic, for example, is now directly tied to the perceived clinical utility and the likelihood of securing a favorable regulatory approval pathway, a process where a 10% improvement in analytical sensitivity can be the deciding factor for approval.
- This strategic calculus underscores the market's evolution from a research-centric field to a pivotal component of mainstream healthcare delivery.
What are the Key Data Covered in this Gene Panel Market Research and Growth Report?
-
What is the expected growth of the Gene Panel Market between 2026 and 2030?
-
USD 7.81 billion, at a CAGR of 23.6%
-
-
What segmentation does the market report cover?
-
The report is segmented by Product (Small panel testing, and Large panel testing), Technique (Amplicon, and Hybridization), Application (Oncology, Genetic disorders, Pharmacogenomics, and Others) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Ascendancy of precision oncology and proliferation of targeted therapies, Complex and fragmented regulatory and reimbursement landscape
-
-
Who are the major players in the Gene Panel Market?
-
Agilent Technologies Inc., Azenta Inc., BGI Group, Centogene N.V., Color Health Inc., Danaher Corp., Eurofins Scientific SE, F. Hoffmann La Roche Ltd., Fagron NV, GenXPro GmbH, Illumina Inc., MedGenome Labs Ltd., MGZ Medical Genetics Center, Novogene Co. Ltd., OPKO Health Inc., Oxford Nanopore Technologies, QIAGEN N.V., Quest Diagnostics Inc. and Thermo Fisher Scientific Inc.
-
Market Research Insights
- The market's momentum is sustained by the expanding utility of genomic profiling in personalized medicine. The integration of pharmacogenomic testing into routine care has demonstrated a 30% reduction in adverse drug reaction incidents in certain patient cohorts. Furthermore, the application of multi-modal analysis in clinical trials enables more precise patient stratification, improving study success rates by over 15%.
- This focus on evidence-based medicine is driving demand for advanced diagnostic tools. Health technology assessment bodies now increasingly favor tests that demonstrate clear cost-effectiveness analysis, supporting their inclusion in genomic medicine service frameworks.
- The ongoing efforts to establish clear test validation guidelines are also crucial for ensuring consistent quality and overcoming issues like algorithmic bias in genomics, thereby fostering greater clinical trust and adoption.
We can help! Our analysts can customize this gene panel market research report to meet your requirements.


 RIA -
RIA - 
