Human Papillomavirus (Hpv) Therapeutics Market Size 2024-2028
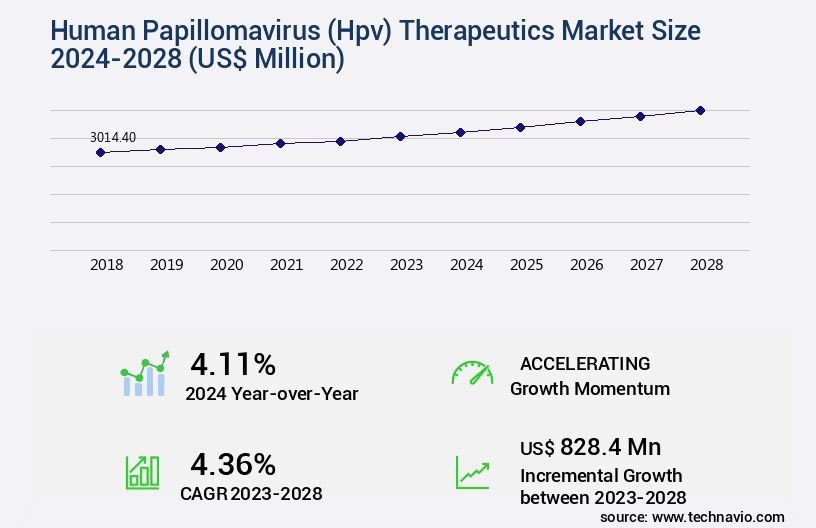
The human papillomavirus (hpv) therapeutics market size is valued to increase USD 828.4 million, at a CAGR of 4.36% from 2023 to 2028. Advent of vaccines will drive the human papillomavirus (hpv) therapeutics market.
Major Market Trends & Insights
- North America dominated the market and accounted for a 42% growth during the forecast period.
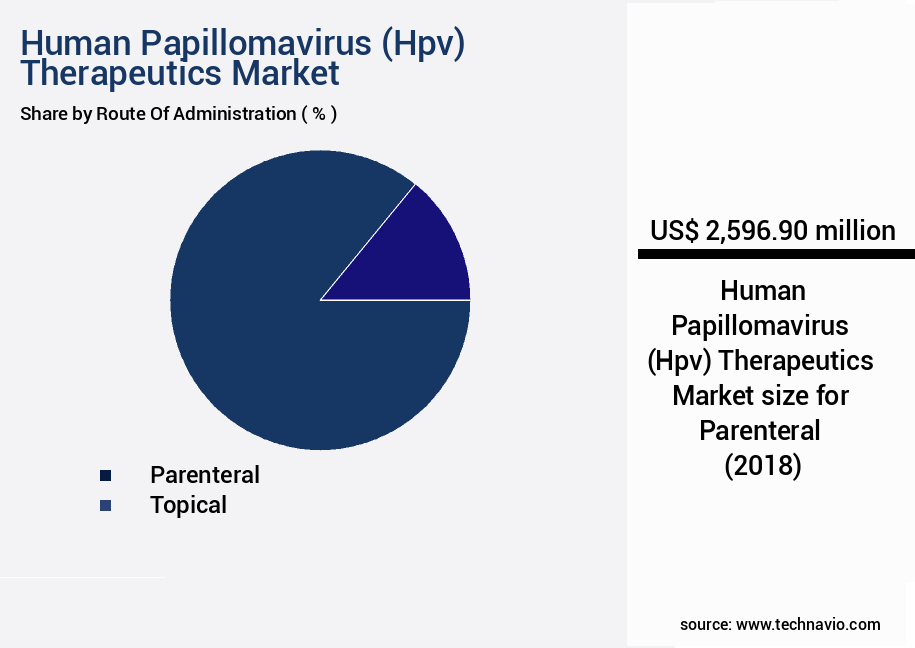
- By Route Of Administration - Parenteral segment was valued at USD 2596.90 million in 2022
- By Indication - Cervical cancer segment accounted for the largest market revenue share in 2022
Market Size & Forecast
- Market Opportunities: USD 42.92 million
- Market Future Opportunities: USD 828.40 million
- CAGR : 4.36%
- North America: Largest market in 2022
Market Summary
- The market encompasses a continually evolving landscape of core medical technologies and applications, service types, and regulatory frameworks. With the advent of vaccines and the relentless pursuit of new drugs for HPV treatment, this market is poised for significant advancements. For instance, HPV vaccines accounted for over 50% of the market share in 2020, underscoring their growing importance. However, challenges such as the anti-vaxxer movement pose a threat to market growth. Regions like North America and Europe are leading the charge in HPV therapeutics, driven by robust healthcare infrastructure and increased awareness.
- The market's dynamic nature is further fueled by ongoing research and development efforts in areas like immunotherapy and gene therapy. These factors collectively contribute to the evolving patterns and opportunities within the Human Papillomavirus Therapeutics Market.
What will be the Size of the Human Papillomavirus (Hpv) Therapeutics Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Human Papillomavirus (Hpv) Therapeutics Market Segmented and what are the key trends of market segmentation?
The human papillomavirus (hpv) therapeutics industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2024-2028, as well as historical data from 2018-2022 for the following segments.
- Route Of Administration
- Parenteral
- Topical
- Indication
- Cervical cancer
- Anal cancer
- Vulvar and vaginal cancer
- Others
- Geography
- North America
- US
- Canada
- Europe
- Germany
- APAC
- China
- South America
- Brazil
- Rest of World (ROW)
- North America
By Route Of Administration Insights
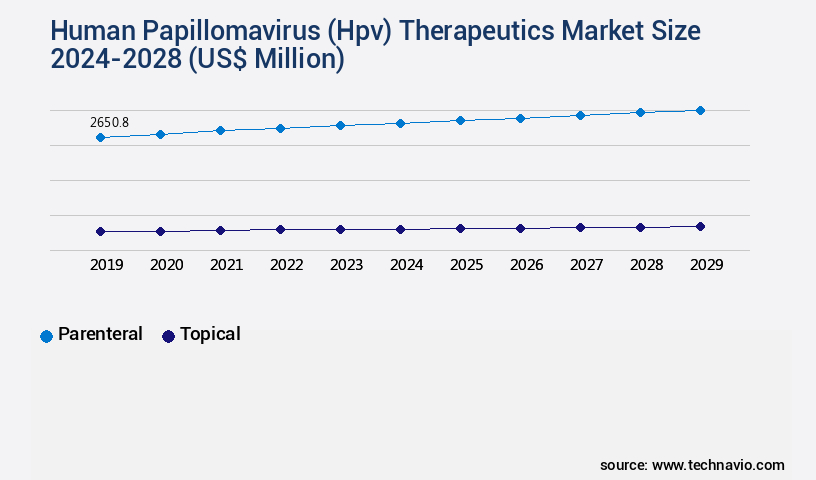
The parenteral segment is estimated to witness significant growth during the forecast period.
The Parenteral segment was valued at USD 2596.90 million in 2018 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 42% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Human Papillomavirus (Hpv) Therapeutics Market Demand is Rising in North America Request Free Sample
In the global HPV therapeutics market, North America holds a significant share, driven by the high prevalence of HPV infections and the availability of approved treatments in the region. HPV is the most common sexually transmitted infection in the US, affecting approximately one-fourth of the adult population, according to the Centers for Disease Control and Prevention (CDC). Among adults aged 18 to 59 years, the prevalence of HPV-related indications was 42.5%. HPV is linked to various health issues, including cancer, genital warts, and non-life-threatening infections.
The US alone has an estimated population of approximately 14 million people affected by HPV annually. This high prevalence and the associated health concerns underscore the importance of ongoing research and development in the HPV therapeutics market.
Market Dynamics
Our researchers analyzed the data with 2023 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
The global HPV therapeutics market is witnessing significant growth due to the increasing prevalence of high-risk HPV infections and the subsequent risk of developing cervical cancer. According to the World Health Organization, approximately 14 million new cases of HPV are diagnosed each year, with cervical cancer being the most common cancer in women under 35. Several therapeutic approaches are under development to address HPV-associated diseases, including therapeutic antibodies, oncolytic viruses, and immunotherapies. Therapeutic antibodies target specific HPV proteins to neutralize their effects, while oncolytic viruses selectively target and destroy cancer cells. Immunotherapies, such as checkpoint inhibitors, enhance the body's immune response against HPV-infected cells.
Despite the promising potential of these therapies, challenges remain. Immunotherapy side effects and viral vector safety profiles are critical concerns, as are drug delivery efficiency and clinical trial outcome measures. Patient selection criteria and biomarker discovery strategies are also essential for optimizing treatment efficacy. Vaccine development costs and antiviral drug development challenges further complicate the landscape. According to disease modeling software, the cost of developing a new HPV vaccine can exceed USD1 billion. However, the potential market size for HPV therapeutics is substantial, with significant growth potential. Adoption rates for HPV therapeutics in developed regions are significantly higher than in developing regions due to better cervical cancer screening guidelines and resources.
For instance, more than 70% of new product developments in the HPV therapeutics market focus on North America and Europe. In conclusion, the HPV therapeutics market is a dynamic and complex landscape, driven by the need to address the global burden of HPV infections and cervical cancer. Therapeutic approaches, such as therapeutic antibodies, oncolytic viruses, and immunotherapies, hold promise but face challenges related to safety, efficacy, and cost. Continued research and innovation are essential to overcome these challenges and bring effective HPV therapeutics to market.
What are the key market drivers leading to the rise in the adoption of Human Papillomavirus (Hpv) Therapeutics Industry?
- The arrival of vaccines serves as the primary catalyst for market growth.
- The HPV therapeutics market has experienced significant progress in the realm of HPV disease prevention. This advancement can be attributed to the increasing research and development of advanced vaccines with extended half-life. These vaccines have demonstrated high efficacy against various HPV-causing viruses and have proven effective in preventing multiple HPV-related indications. Consequently, the global utilization of these vaccines has surged, aiming to address the challenges posed by HPV.
- Notably, HPV affects approximately 42 million individuals in the US, making it the most common sexually transmitted infection. Despite the absence of specific growth rate percentages or monetary values, the market's trajectory is evident in the increasing adoption and application of these vaccines across various sectors.
What are the market trends shaping the Human Papillomavirus (Hpv) Therapeutics Industry?
- The research and development of new drugs is currently a significant market trend for the treatment of Orthostatic Hypotension.
- Orthostatic hypotension (OH), characterized by a significant drop in blood pressure upon standing, is a prevalent condition associated with increased risks of fatalities, syncope, falls, and cognitive decline. Medications, particularly cardiovascular and psychotropic drugs, are among the leading causes of OH. These medications can impair the blood pressure response to standing, leading to drug-induced OH. Comorbidities, advanced age, neurogenic autonomic dysfunction, and other OH risk factors often coincide with hypotensive medications, amplifying the risk of symptoms and side effects.
- Individuals with postural orthostatic tachycardia syndrome (POTS) frequently experience both OH and abnormal heart rate increases upon standing. This complex interplay between medications and underlying conditions highlights the importance of monitoring and managing OH to mitigate potential complications.
What challenges does the Human Papillomavirus (Hpv) Therapeutics Industry face during its growth?
- The anti-vaxxer movement poses a significant challenge to the growth of the vaccines industry by advocating against the use of vaccines, potentially undermining public trust and confidence in vaccination programs.
- The HPV therapeutics market faces a significant challenge with the increasing vaccine skepticism among the global population. Despite the US Food and Drug Administration's (FDA) endorsement of HPV vaccines for preventing and treating various indications, there is a growing reluctance towards vaccinations. This trend is primarily driven by concerns that vaccines may lead to autism, a belief that has gained popularity despite scientific evidence to the contrary. The notion of vaccine overload, where individuals receive multiple vaccines, is a common concern, with some believing that the immune system becomes weakened, potentially leading to autism.
- This hesitation towards vaccines poses a significant challenge to the HPV therapeutics market's growth trajectory. The market's dynamics are continuously evolving, with ongoing research and development efforts aimed at addressing vaccine hesitancy and improving vaccine efficacy.
Exclusive Technavio Analysis on Customer Landscape
The human papillomavirus (hpv) therapeutics market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the human papillomavirus (hpv) therapeutics market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Human Papillomavirus (Hpv) Therapeutics Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, human papillomavirus (hpv) therapeutics market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
2A Pharma ApS - This company specializes in the development and provision of human papillomavirus (HPV) therapeutics, featuring the L2-based HPV vaccine as a key offering. The L2 vaccine represents a significant advancement in HPV treatment, offering potential protection against various HPV strains associated with cancer and other diseases. The company's commitment to innovative research and development in HPV therapeutics positions it as a leader in the field.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- 2A Pharma ApS
- AbbVie Inc.
- Bausch Health Companies Inc.
- Bavarian Nordic AS
- BioVaxys Technology Corp.
- F. Hoffmann La Roche Ltd.
- GlaxoSmithKline Plc
- Inovio Pharmaceuticals Inc.
- ISA Pharmaceuticals BV
- Merck and Co. Inc.
- Serum Institute of India Pvt. Ltd.
- Walvax Biotechnology Co. Ltd.
- Xiamen Innovax Biotech Co. Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Human Papillomavirus (Hpv) Therapeutics Market
- In January 2024, Merck & Co. Announced the FDA approval of their HPV therapeutic, Gardasil 9, for the prevention of recurrent disease in adults. This expansion of the Gardasil franchise marked a significant strategic move to address the unmet need for effective HPV treatment and prevention (Merck press release, 2024).
- In March 2024, GSK and Vir Biotechnology entered a collaboration to develop and commercialize novel HPV therapeutics. This partnership aimed to leverage GSK's expertise in vaccine development and Vir Biotechnology's proprietary technology platform (GSK press release, 2024).
- In May 2024, Moderna Therapeutics initiated a Phase 1 clinical trial for its mRNA-based HPV vaccine, mRNA-1647. This trial marked a significant technological advancement in the HPV therapeutics market, as Moderna's mRNA technology has shown promise in other vaccine applications (Moderna press release, 2024).
- In April 2025, the European Commission approved the use of Janssen Pharmaceutica's HPV vaccine, Elilis, for the prevention of cervical, vulvar, vaginal, and anal cancers in females aged 12 to 26. This approval marked a key regulatory milestone for Janssen, expanding their presence in the competitive HPV therapeutics market (Janssen press release, 2025).
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Human Papillomavirus (Hpv) Therapeutics Market insights. See full methodology.
|
Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
168 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 4.36% |
|
Market growth 2024-2028 |
USD 828.4 million |
|
Market structure |
Concentrated |
|
YoY growth 2023-2024(%) |
4.11 |
|
Key countries |
US, Germany, Canada, China, and Brazil |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The market is a dynamic and evolving landscape, driven by ongoing research and advancements in HPV diagnostics, vaccines, and therapeutic approaches. HPV persistence studies continue to provide valuable insights into the natural history of HPV infections and their progression to cancer. Oncogenic HPV types, particularly high-risk subtypes, are the primary focus of therapeutic development. Immunological assessments, such as immune response assays, play a crucial role in evaluating clinical trial endpoints and disease progression modeling. Advanced viral vector designs are being explored to enhance the efficacy of HPV vaccines, while immunogenic cell death and HPV DNA detection are key targets for immunotherapy approaches.
- Patient stratification based on infection stages and risk subtypes is essential for optimizing therapeutic interventions. Molecular diagnostics are increasingly utilized for accurate identification and monitoring of HPV infections, enabling personalized treatment plans. Preclinical HPV models and viral load quantification are essential tools for assessing the efficacy of therapeutic vaccines, drug delivery systems, and targeted therapies. The role of cellular and humoral immunity in HPV clearance rates and lesion regression rates is a major area of investigation. The tumor microenvironment and cancer cell apoptosis are also critical factors influencing treatment response biomarkers and therapeutic antibodies. Low-risk HPV subtypes, which often cause genital warts, are also under investigation for potential therapeutic interventions.
- In the realm of HPV therapeutics, ongoing research focuses on the e6/e7 oncoproteins and viral integration analysis to better understand the mechanisms of HPV-associated diseases and develop more effective treatments. The development of novel therapeutic strategies, such as oncolytic virus therapy, holds great promise for improving patient outcomes and reducing the global burden of HPV-related cancers.
What are the Key Data Covered in this Human Papillomavirus (Hpv) Therapeutics Market Research and Growth Report?
-
What is the expected growth of the Human Papillomavirus (Hpv) Therapeutics Market between 2024 and 2028?
-
USD 828.4 million, at a CAGR of 4.36%
-
-
What segmentation does the market report cover?
-
The report segmented by Route Of Administration (Parenteral and Topical), Indication (Cervical cancer, Anal cancer, Vulvar and vaginal cancer, and Others), and Geography (North America, Europe, Asia, and Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia, and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Advent of vaccines, Anti-vaxxer movement
-
-
Who are the major players in the Human Papillomavirus (Hpv) Therapeutics Market?
-
Key Companies 2A Pharma ApS, AbbVie Inc., Bausch Health Companies Inc., Bavarian Nordic AS, BioVaxys Technology Corp., F. Hoffmann La Roche Ltd., GlaxoSmithKline Plc, Inovio Pharmaceuticals Inc., ISA Pharmaceuticals BV, Merck and Co. Inc., Serum Institute of India Pvt. Ltd., Walvax Biotechnology Co. Ltd., and Xiamen Innovax Biotech Co. Ltd.
-
Market Research Insights
- The market encompasses a diverse range of approaches to combat HPV-related diseases, including antiviral drugs, vaccines, and combination therapies. According to recent estimates, the global market for HPV therapeutics is projected to reach USD12 billion by 2025, growing at a steady annual rate. This growth is driven by the increasing prevalence of HPV infections and the development of new therapeutic strategies. One notable area of innovation is in the development of vaccines, with over 200 HPV vaccine candidates in various stages of preclinical and clinical development. For instance, vaccine development pipelines focus on improving prophylactic vaccine efficacy through advanced HPV serotyping and epitope mapping techniques.
- Meanwhile, antiviral drugs target viral replication inhibition and cell cycle regulation, with some drugs demonstrating therapeutic efficacy metrics such as apoptosis induction and safety and tolerability in in vitro and in vivo studies. However, HPV-related cancers, such as cervical, head and neck, and anal cancers, pose significant challenges due to their complex molecular mechanisms, including viral oncogenesis and viral pathogenesis. To address these challenges, researchers are exploring combination therapies, which leverage the synergistic effects of multiple therapeutic agents. For example, combination therapies that incorporate immune checkpoint inhibitors and DNA repair pathway modulators show promise in overcoming drug resistance mechanisms and improving therapeutic efficacy.
- Despite these advancements, the HPV therapeutics market faces ongoing challenges, including the need for personalized medicine and the development of biomarkers for HPV-related cancers. Additionally, understanding the role of cancer stem cells and HPV genotyping in disease progression and treatment response remains a critical area of research. Ultimately, the continued evolution of HPV therapeutics will require a multidisciplinary approach, integrating advances in drug metabolism, clinical trial design, and molecular mechanisms to develop effective and safe treatments for HPV-related diseases.
We can help! Our analysts can customize this human papillomavirus (hpv) therapeutics market research report to meet your requirements.


 RIA -
RIA - 
