Pressure Infusion Bags Market Size 2026-2030
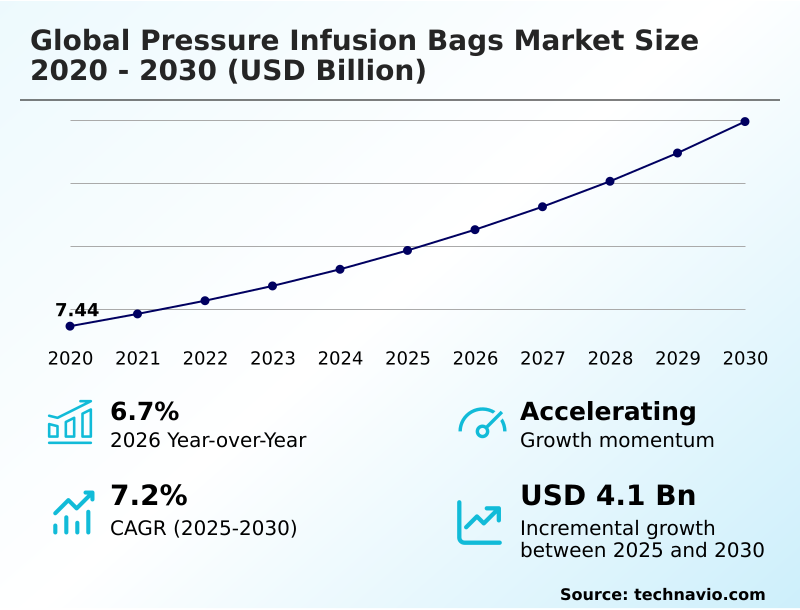
The pressure infusion bags market size is valued to increase by USD 4.10 billion, at a CAGR of 7.2% from 2025 to 2030. Escalation of surgical volumes and emergency trauma interventions will drive the pressure infusion bags market.
Major Market Trends & Insights
- Asia dominated the market and accounted for a 39% growth during the forecast period.
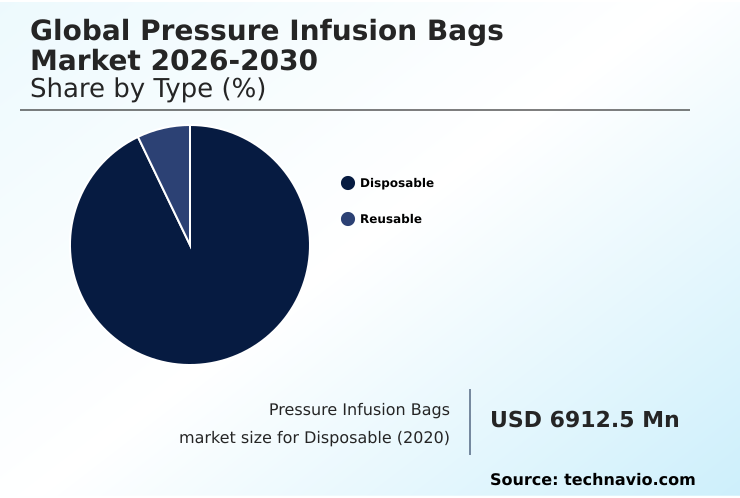
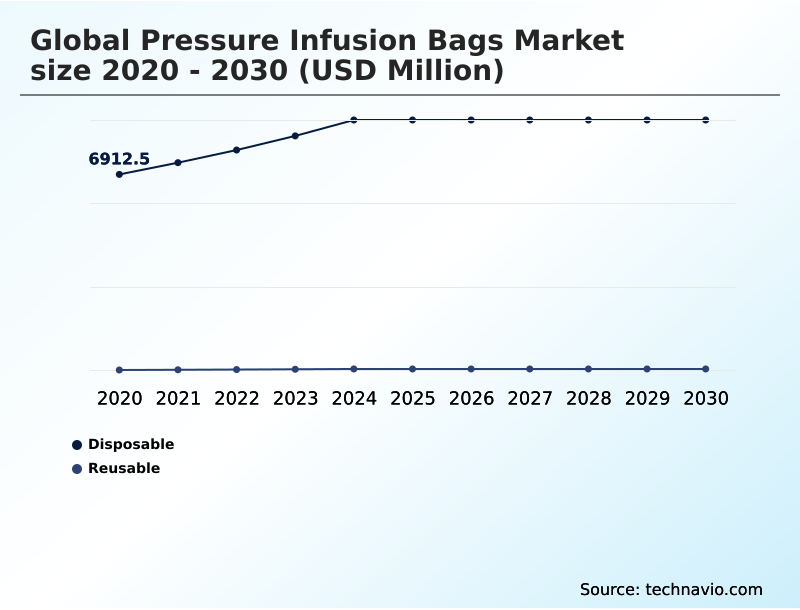
- By Type - Disposable segment was valued at USD 8.69 billion in 2024
- By Material - Nylon segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 6.51 billion
- Market Future Opportunities: USD 4.10 billion
- CAGR from 2025 to 2030 : 7.2%
Market Summary
- The pressure infusion bags market is defined by its critical role in emergency and perioperative care, where devices enabling rapid fluid resuscitation are essential. Growth is propelled by increasing surgical volumes and the need for hemodynamic stability in high-acuity settings.
- A significant trend is the institutionalization of single-use medical devices and smart infusion systems to enhance infection control protocols and provide automated pressure monitoring. For instance, a hospital system implementing IoT-enabled sensors and rfid-enabled tracking can achieve better digital traceability and inventory management, optimizing the supply of disposable pressure infusion bags and cuffs for trauma resuscitation.
- However, the industry faces challenges from supply chain fragility for biocompatible polymers like medical-grade nylon and polyurethane resins, alongside the complexities of regulatory compliance management. The evolution toward connected infusion systems and data-driven fluid delivery is reshaping the landscape, pushing for greater technical interoperability and integration with electronic health records.
What will be the Size of the Pressure Infusion Bags Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Get Free Sample
How is the Pressure Infusion Bags Market Segmented?
The pressure infusion bags industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Type
- Disposable
- Reusable
- Material
- Nylon
- Polyurethane
- Latex
- Others
- End-user
- Hospitals
- Clinics
- Others
- Geography
- North America
- US
- Canada
- Mexico
- Asia
- Europe
- Germany
- UK
- France
- Rest of World (ROW)
- North America
By Type Insights
The disposable segment is estimated to witness significant growth during the forecast period.
The disposable segment is crucial for rapid fluid administration, shaped by a focus on mitigating cross-contamination and strict infection control protocols. These single-use medical devices, made from materials like medical-grade nylon, are discarded after one use to prevent hospital-acquired infections.
In high-turnover settings like ambulatory surgical centers, disposable systems support immediate deployment without sterilization overhead, a shift underscored by a thirty-seven percent funding increase for trauma care modernization in some regions.
This transition favors ready-to-use, sterile solutions featuring pvc-free polymers that streamline clinical workflow optimization.
The institutionalization of these systems, including pressure infusion cuffs and bags, reflects a move toward enhanced safety and efficiency in both emergency medicine applications and routine intravenous fluid delivery.
The Disposable segment was valued at USD 8.69 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
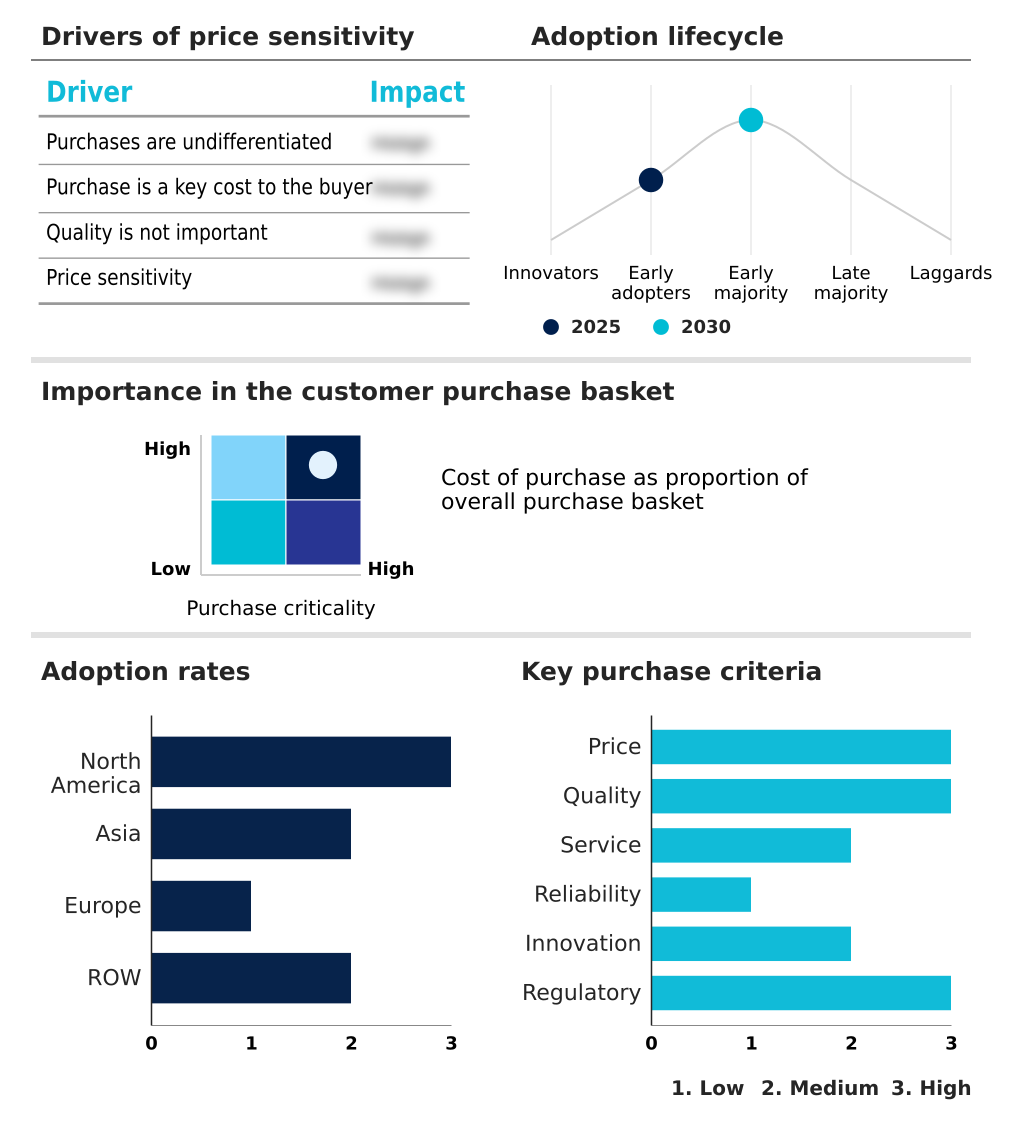
Asia is estimated to contribute 39% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Pressure Infusion Bags Market Demand is Rising in Asia Get Free Sample
The global landscape is diverse, with North America focusing on high-tech, smart infusion systems and data-driven fluid delivery.
The region's growth is steady, while Asia is set to contribute 39% of the market's incremental growth, driven by infrastructure development and rising healthcare standards that demand better emergency medicine applications.
Europe, with a more mature market, shows moderate growth but leads in adopting sustainable, pvc-free polymers and circular economy principles. This geographic variance creates a complex environment where a one-size-fits-all strategy is ineffective.
Providers must navigate different regulatory frameworks and demands, from North America's focus on technical interoperability to Asia’s volume-driven needs for both disposable pressure infusion bags and reusable systems.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the market is increasingly complex, influenced by competing priorities of cost, safety, and technological advancement. Understanding disposable pressure infusion bag benefits is crucial for hospitals aiming to improve infection control, while others analyze reusable pressure infusion cuff sterilization for long-term cost savings.
- The debate over nylon vs polyurethane infusion bags continues, with material science dictating performance in trauma care. Smart pressure infusion system advantages, particularly the integration of IoT sensors in infusion therapy, are becoming a key differentiator. These systems are vital for preventing air embolism with pressure bags.
- The focus on infection control with single-use infusers is a major driver, especially for pressure bags for rapid blood transfusion. Clinics weigh the cost-effectiveness of disposable infusion systems against capital investments. Automating fluid delivery in ICUs is a primary goal, pushing manufacturers to navigate stringent regulatory standards for infusion devices.
- Concurrently, supply chain challenges for medical polymers and the need for integrating infusion data with EHRs are significant hurdles. As the market expands to include pressure infusion bags for home care and biodegradable materials for medical bags, choosing pressure bags for ambulatory centers requires careful evaluation of advances in real-time infusion analytics and best practices for pressure bag usage.
- The growth contribution from developing regions is over twice that of established markets, highlighting a significant shift in resource allocation.
What are the key market drivers leading to the rise in the adoption of Pressure Infusion Bags Industry?
- The escalation of surgical volumes and a rising number of emergency trauma interventions are key drivers for the market.
- The market is propelled by the need for rapid fluid resuscitation and enhanced hemodynamic stability in critical care. Escalating surgical volumes and trauma interventions necessitate effective blood product administration and intravenous fluid delivery.
- This demand is particularly acute in emergency medicine applications and perioperative care, where controlled flow rate is vital. The strategic transition to single-use medical devices, driven by stringent infection control protocols, further fuels growth.
- A significant funding increase of 37% for trauma services in some emerging economies highlights the global push to modernize care.
- Moreover, with nearly one-fourth of the population in some developed nations projected to be over 60, the demand for procedures requiring such devices is set to rise.
What are the market trends shaping the Pressure Infusion Bags Industry?
- The inclusion of IoT-enabled smart monitoring is an emerging market trend, coupled with the deployment of real-time infusion analytics for enhanced clinical oversight.
- A primary trend is the move toward connected infusion systems and telemetry-capable equipment, enabling real-time infusion analytics. This data-driven fluid delivery approach enhances patient safety and supports clinical workflow optimization in settings like smart ward technologies. The adoption of high-performance polyolefin blends and other pvc-free iv bags aligns with sustainability goals and stringent regulations.
- This shift has improved biocompatibility in infusion solutions. As a result of these technological and material advancements, some facilities report enhanced tracking capabilities and a notable reduction in medication errors, contributing to better overall patient outcomes and operational efficiencies in surgical theater devices and intensive care unit equipment.
What challenges does the Pressure Infusion Bags Industry face during its growth?
- The intensification of regulatory compliance requirements and the stringent implementation of ISO standards present a key challenge to industry growth.
- The market faces significant hurdles from supply chain fragility and regulatory compliance management. The volatility in procuring specialized polyurethane resins and reinforced nylon construction materials has led to lead times increasing by over 20%, disrupting production. This instability directly impacts the availability of both disposable and reusable pressure infusion systems.
- Furthermore, achieving technical interoperability and digital traceability for smart infusion systems presents a major challenge, hindering seamless electronic health record integration. The economic landscape, marked by growth contracting to 2.6% in 2024, tightens healthcare budgets, intensifying price pressures and affecting investment in new technologies like automated fluid management and advanced home healthcare infusion solutions.
Exclusive Technavio Analysis on Customer Landscape
The pressure infusion bags market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the pressure infusion bags market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Pressure Infusion Bags Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, pressure infusion bags market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
ASP Global LLC - Provides disposable and reusable pressure infusion systems, enabling rapid fluid administration for critical care applications and enhancing patient safety through controlled delivery.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- ASP Global LLC
- B.Braun SE
- Baxter International Inc.
- Bosch Sohn GmbH u. Co. KG
- Cardinal Health Inc.
- Demax Medical Technology Co.
- DeRoyal Industries Inc.
- Friedrich Bosch GmbH and Co. KG
- ICU Medical Inc.
- Jorgensen Laboratories Inc.
- Medline Inc.
- Mercury Medical
- Metko Medical
- NovaMed USA
- Poly Medicure Ltd.
- Premier Inc.
- Romsons Group
- Rudolf Riester GmbH
- SunMed LLC
- Teleflex Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Pressure infusion bags market
- In February 2025, OSAA Innovation highlighted that its latest mechanical infusion pump innovations are strictly composed of non-latex components to support mobile, pole-free care without compromising safety for allergic individuals.
- In March 2025, CareFusion Technologies launched its next-generation smart pressure infusion system, which utilizes IoT-enabled sensors and artificial intelligence-based algorithms to provide clinicians with automated alerts regarding pressure dynamics.
- In May 2025, Intuitive Surgical received updated regulatory clearance for its advanced surgical systems in the United States, a development that signifies the broader regional trend toward minimally invasive procedures which often require precise fluid management.
- In February 2026, the United States Food and Drug Administration finalized the transition of its Quality System Regulation to the Quality Management System Regulation (QMSR), aligning domestic requirements with the international ISO 13485:2016 standard.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Pressure Infusion Bags Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 285 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 7.2% |
| Market growth 2026-2030 | USD 4103.6 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 6.7% |
| Key countries | US, Canada, Mexico, China, India, Japan, South Korea, Thailand, Indonesia, Germany, UK, France, Italy, Spain, The Netherlands, Brazil, Saudi Arabia, UAE, Turkey, South Africa, Israel, Argentina and Colombia |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The pressure infusion bags market is advancing beyond simple mechanical devices, driven by demands for enhanced safety and efficiency in critical care. The core of this evolution lies in the material science of biocompatible polymers and high-tensile materials, such as medical-grade nylon and polyurethane resins, which are essential for creating durable, puncture-resistant films.
- A key focus is on integrated pressure gauges and over-inflation relief valves to ensure patient safety during rapid fluid resuscitation and blood product administration. The shift toward single-use medical devices is accelerating to meet stringent infection control protocols and mitigate cross-contamination, especially in ambulatory surgical centers.
- Simultaneously, the development of smart infusion systems with IoT-enabled sensors and automated pressure monitoring is transforming perioperative care. These systems, featuring rfid-enabled tracking, are crucial for achieving digital traceability and electronic health record integration.
- However, this progress is constrained by supply chain issues, with lead times for key components increasing by over 20%, impacting the availability of both disposable pressure infusion bags and reusable pressure infusion systems. The market is also adapting to home infusion therapies and the need for invasive pressure monitoring solutions, reflecting a broader trend toward decentralized care.
What are the Key Data Covered in this Pressure Infusion Bags Market Research and Growth Report?
-
What is the expected growth of the Pressure Infusion Bags Market between 2026 and 2030?
-
USD 4.10 billion, at a CAGR of 7.2%
-
-
What segmentation does the market report cover?
-
The report is segmented by Type (Disposable, and Reusable), Material (Nylon, Polyurethane, Latex, and Others), End-user (Hospitals, Clinics, and Others) and Geography (North America, Asia, Europe, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Asia, Europe and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Escalation of surgical volumes and emergency trauma interventions, Intensification of regulatory compliance and stringent ISO standards implementation
-
-
Who are the major players in the Pressure Infusion Bags Market?
-
ASP Global LLC, B.Braun SE, Baxter International Inc., Bosch Sohn GmbH u. Co. KG, Cardinal Health Inc., Demax Medical Technology Co., DeRoyal Industries Inc., Friedrich Bosch GmbH and Co. KG, ICU Medical Inc., Jorgensen Laboratories Inc., Medline Inc., Mercury Medical, Metko Medical, NovaMed USA, Poly Medicure Ltd., Premier Inc., Romsons Group, Rudolf Riester GmbH, SunMed LLC and Teleflex Inc.
-
Market Research Insights
- The market is influenced by a dynamic interplay of efficiency and safety imperatives. Healthcare providers prioritize clinical workflow optimization, leading to higher adoption of systems that improve outcomes. For example, the move toward disposable systems aims to reduce hospital-acquired infection rates, while the integration of smart technologies is improving precision.
- This drive for advancement is tempered by economic realities; supply chain fragility for key components has increased lead times by over 20%, impacting operational planning.
- Concurrently, government initiatives, such as a 37% increase in funding for trauma services in some developing nations, create new avenues for growth and modernization, accelerating the adoption of advanced solutions for data-driven fluid delivery and real-time infusion analytics.
We can help! Our analysts can customize this pressure infusion bags market research report to meet your requirements.


 RIA -
RIA - 
