Breast Cancer Liquid Biopsy Testing Devices Market Size 2024-2028
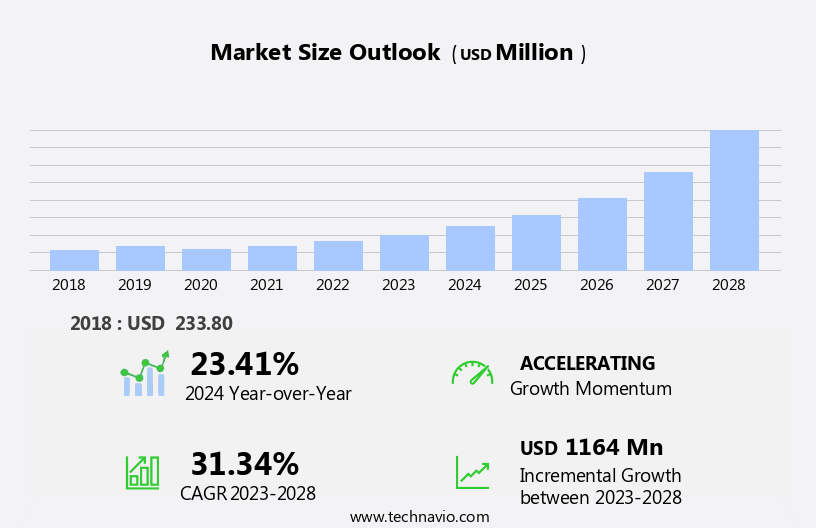
The breast cancer liquid biopsy testing devices market size is forecast to increase by USD 1.16 billion, at a CAGR of 31.34% between 2023 and 2028.
- The market is driven by the increasing prevalence of breast cancer and technological advances in diagnostics. The early detection and precise identification of breast cancer through liquid biopsy testing offer significant benefits, including minimally invasive procedures and real-time monitoring of tumor progression. This market's growth is fueled by the rising awareness of breast cancer and the need for more accurate and efficient diagnostic tools. However, high costs associated with liquid biopsy testing pose a considerable challenge for market expansion. The complex nature of these tests and the requirement for specialized equipment and expertise contribute to the high costs, which may limit their accessibility to a broader patient population.
- Companies in this market must focus on cost reduction strategies, such as developing more affordable testing solutions or partnering with healthcare providers to offer bundled services, to overcome this challenge and expand their reach. By addressing the cost issue and continuing to innovate, market players can capitalize on the growing demand for advanced breast cancer diagnostic tools and improve patient outcomes.
What will be the Size of the Breast Cancer Liquid Biopsy Testing Devices Market during the forecast period?
Explore in-depth regional segment analysis with market size data - historical 2018-2022 and forecasts 2024-2028 - in the full report.
Request Free Sample
The breast cancer liquid biopsy testing market continues to evolve, driven by advancements in technologies and applications across various sectors. Minimal residual disease (MRD) monitoring, Circulating Tumor DNA (ctDNA), and clinical utility are key areas of focus. Next-generation sequencing (NGS) and DNA methylation analysis play pivotal roles in MRD assessment, enabling the detection of ctDNA with high sensitivity and specificity. Epigenetic biomarkers, such as miRNAs, offer additional value in cancer diagnosis and prognosis. Non-invasive sampling through plasma preparation is a significant trend, facilitating early cancer detection and cancer recurrence monitoring. Sensitivity and specificity are crucial factors, with ongoing efforts to improve analytical sensitivity and assay validation.
Tumor heterogeneity and tumor mutational burden are also important considerations, leading to the development of multiplexed assays and cell sorting techniques. Molecular diagnostics, mutation profiling, and biomarker discovery are essential applications of liquid biopsy technology. Digital PCR and gene expression profiling are advanced techniques used to enhance the accuracy and reliability of these applications. Plasma preparation methods continue to advance, enabling the detection of protein biomarkers and improving detection limits. Personalized medicine and cancer therapy selection are major areas of growth, as liquid biopsy technology provides valuable insights for targeted therapy and precision oncology. The continuous unfolding of market activities and evolving patterns in the breast cancer liquid biopsy testing market underscore its importance in the field of cancer research and diagnosis.
How is this Breast Cancer Liquid Biopsy Testing Devices Industry segmented?
The breast cancer liquid biopsy testing devices industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2024-2028, as well as historical data from 2018-2022 for the following segments.
- Type
- CTCs and circulating nucleic acids
- Extracellular vesicles
- Geography
- North America
- US
- Canada
- Europe
- France
- Germany
- UK
- Rest of World (ROW)
- North America
By Type Insights
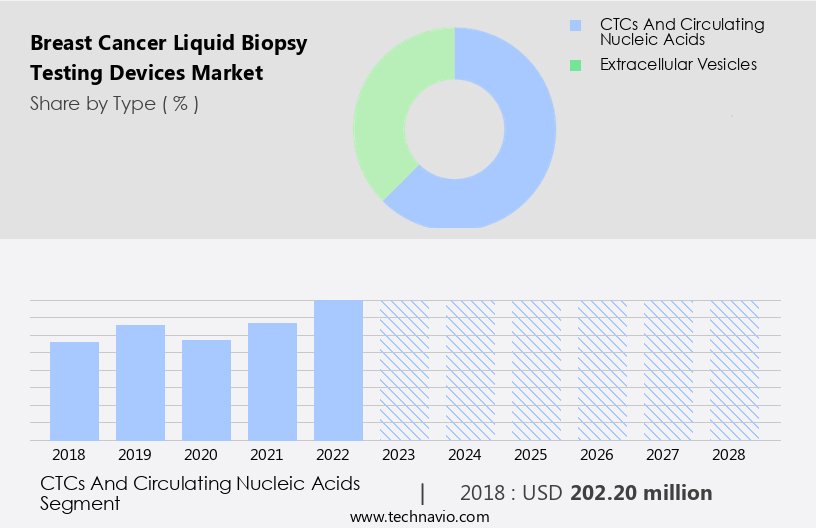
The ctcs and circulating nucleic acids segment is estimated to witness significant growth during the forecast period.
In the realm of cancer diagnostics, the identification of circulating tumor cells (CTCs) and circulating nucleic acids has emerged as a significant prognostic and predictive tool for various cancer types, including breast, lung, prostate, and colorectal. CTCs, which originate from primary and secondary tumors and enter the circulatory system, serve as representatives of the tumor and its metastases. In the context of breast cancer, CTCs are primarily utilized for biomarker analysis, enabling the detection of new genetic mutations. Circulating nucleic acids, such as cell-free DNA, epigenetic biomarkers, and microRNAs, are also extensively studied in this field.
Next-generation sequencing (NGS) technologies, including DNA methylation analysis and mutation profiling, facilitate the discovery of new biomarkers and enhance our understanding of tumor heterogeneity. Liquid biopsy technology, employing non-invasive sampling methods, offers increased sensitivity and specificity compared to traditional methods. Single-cell sequencing and digital PCR further improve analytical sensitivity, while assay validation and gene expression profiling ensure assay reliability. The integration of these techniques into clinical practice holds the potential for personalized medicine, cancer therapy selection, and early cancer detection. Plasma sample preparation methods, including cell sorting and protein biomarker detection, are essential components of this approach.
The tumor mutational burden and circulating tumor DNA (ctDNA) detection provide valuable insights into cancer recurrence monitoring and multiplexed assays. Overall, the advancements in liquid biopsy technology and the exploration of various biomarkers continue to shape the evolving landscape of cancer diagnostics.
The CTCs and circulating nucleic acids segment was valued at USD 202.20 billion in 2018 and showed a gradual increase during the forecast period.
Regional Analysis
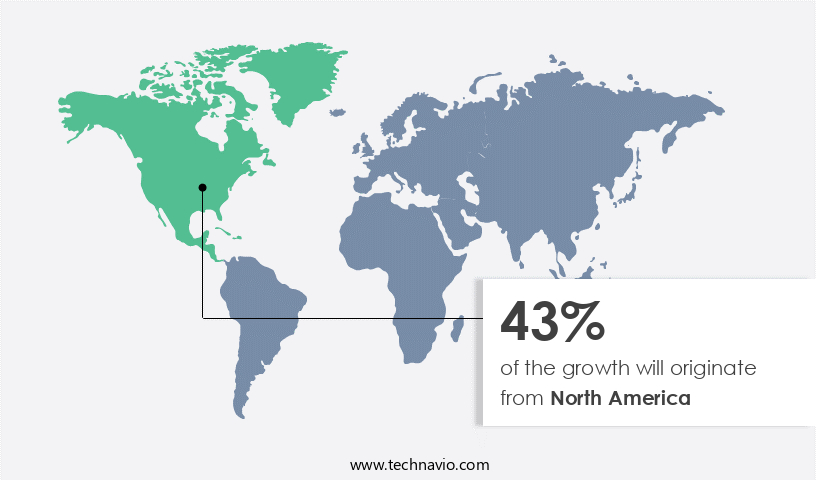
North America is estimated to contribute 43% to the growth of the global market during the forecast period. Technavio's analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
The market in North America is experiencing significant growth, fueled by several key factors. Advanced technologies, such as next-generation sequencing (NGS), DNA methylation analysis, and digital PCR, are increasingly being adopted for the detection and analysis of circulating tumor DNA (ctDNA), epigenetic biomarkers, and cell-free DNA. These technologies enable early cancer detection, cancer recurrence monitoring, and personalized medicine, including cancer therapy selection and tumor mutational burden assessment. The rise in healthcare expenditure, growing awareness about breast cancer screening and diagnostic procedures, and the increasing prevalence of breast cancer are significant market drivers. Furthermore, non-invasive sampling methods, such as plasma preparation and single-cell sequencing, offer advantages over traditional invasive methods.
The market is also witnessing advancements in sample enrichment methods, such as cell sorting and multiplexed assays, which improve analytical sensitivity and assay validation. These advancements contribute to the market's growth, as they enable the detection of minimal residual disease, tumor heterogeneity, and the identification of various biomarkers, including miRNAs, proteins, and gene expression profiles. The presence of numerous companies manufacturing breast cancer liquid biopsy testing devices and the continuous technological advances in liquid biopsy technology further propel market growth. Overall, the market in North America is poised for continued expansion, driven by the adoption of advanced technologies, increasing healthcare expenditure, and the growing prevalence of breast cancer.
Market Dynamics
Our researchers analyzed the data with 2023 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
What are the key market drivers leading to the rise in the adoption of Breast Cancer Liquid Biopsy Testing Devices Industry?
- The rising incidence of breast cancer serves as the primary catalyst for market growth in this sector.
- In the realm of cancer diagnostics, breast cancer continues to pose a significant health challenge, particularly among the aging population. The increasing prevalence of breast cancer is largely attributed to mutations in the BRCA1 and BRCA2 genes, which act as tumor suppressors. However, when mutated, these genes can lead to the development of breast cancer. To address the challenges of cancer recurrence monitoring, advanced technologies such as liquid biopsy have emerged. Liquid biopsy technology utilizes circulating tumor cells (CTCs) and other tumor-derived components in the bloodstream for analysis. A multiplexed assay, which can analyze multiple biomarkers simultaneously, is a valuable tool in this context.
- Analytical sensitivity is a crucial factor in the success of these assays. Assay validation and gene expression profiling are essential steps in ensuring the accuracy and reliability of the results. Sample enrichment methods, such as magnetic beads or immunocapture, are employed to isolate CTCs from the bloodstream for further analysis. The importance of these advancements lies in their potential to provide real-time information on the progression of the disease and the response to treatment. This information can lead to more effective treatment plans and improved patient outcomes.
What are the market trends shaping the Breast Cancer Liquid Biopsy Testing Devices Industry?
- The current market trend is shaped by significant technological advances. It is essential to stay informed about these innovations to maintain a competitive edge in your industry.
- Breast cancer, a significant health concern worldwide, is often linked to genetic mutations in specific genes. Molecular diagnostic tools, particularly those utilizing next-generation sequencing (NGS) in liquid biopsy procedures, are revolutionizing breast cancer diagnosis and treatment. These advanced techniques offer numerous benefits, including non-invasive sampling, increased accuracy, and cost reduction. Circulating tumor DNA (ctDNA) and epigenetic biomarkers, such as DNA methylation analysis, extracted from cell-free DNA (cfDNA), are crucial components of these tests. These biomarkers provide valuable insights into minimal residual disease and the presence of cancer cells, enabling clinicians to make informed decisions regarding treatment plans. NGS technology, with its high sensitivity and specificity, is a game-changer in breast cancer diagnosis.
- This cutting-edge approach allows for the simultaneous detection and analysis of multiple genetic mutations, providing a more comprehensive understanding of the disease. Furthermore, its ability to monitor disease progression and response to treatment makes it an essential tool in personalized medicine. In conclusion, the integration of NGS and other molecular techniques in breast cancer liquid biopsy testing devices is a significant stride in the field of oncology. These advancements offer numerous clinical utility benefits, including improved accuracy, cost reduction, and non-invasive sampling, making them an invaluable asset in the diagnosis and treatment of breast cancer.
What challenges does the Breast Cancer Liquid Biopsy Testing Devices Industry face during its growth?
- The high costs linked to liquid biopsy testing represent a significant challenge impeding the growth of the industry.
- Liquid biopsy testing is a significant advancement in breast cancer diagnosis, providing healthcare professionals with valuable insights into tumor heterogeneity. This non-invasive procedure involves analyzing plasma samples to detect and profile mutations, enabling more accurate diagnosis and personalized treatment plans. Molecular diagnostics, including microRNA detection and mutation profiling, play a crucial role in identifying breast cancer biomarkers. Single-cell sequencing and sensitive detection techniques enable the identification of rare cancer cells, further enhancing the diagnostic accuracy. The adoption of liquid biopsy testing is increasing due to its ability to provide real-time, comprehensive information about the tumor, making it a preferred alternative to traditional biopsy procedures.
- The potential for biomarker discovery and personalized medicine offers significant benefits to patients, making this an essential tool in the fight against breast cancer. With detection limits continuing to improve, liquid biopsy testing is poised to revolutionize the field of molecular diagnostics.
Exclusive Customer Landscape
The breast cancer liquid biopsy testing devices market forecasting report includes the adoption lifecycle of the market, covering from the innovator's stage to the laggard's stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the breast cancer liquid biopsy testing devices market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape
Key Companies & Market Insights
Companies are implementing various strategies, such as strategic alliances, breast cancer liquid biopsy testing devices market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
A. Menarini Industrie Farmaceutiche Riunite Srl - The CELLSEARCH CTC Test is a groundbreaking solution for detecting and quantifying circulating tumor cells in the blood of patients with metastatic breast, prostate, and colorectal cancers.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- A. Menarini Industrie Farmaceutiche Riunite Srl
- Bio Rad Laboratories Inc.
- Cell Microsystems Inc.
- Exact Sciences Corp.
- F. Hoffmann La Roche Ltd.
- Guardant Health Inc.
- Illumina Inc.
- Isogen Life Science BV
- Mesa Laboratories Inc.
- Myriad Genetics Inc.
- Natera Inc.
- NeoGenomics Laboratories Inc.
- Novogene Co. Ltd.
- OncoDNA
- Pfizer Inc.
- QIAGEN NV
- SAGA Diagnostics AB
- Sysmex Corp.
- Thermo Fisher Scientific Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Breast Cancer Liquid Biopsy Testing Devices Market
- In January 2024, F.DA. Approved the use of Guardant Health's liquid biopsy test, GuardantOMNIS, for breast cancer diagnosis and monitoring, marking a significant regulatory milestone in the market (FDA press release).
- In March 2024, Illumina and Grail, two major players, announced a strategic collaboration to develop and commercialize comprehensive tumor profiling tests using liquid biopsy technology (Illumina press release).
- In April 2025, Qiagen raised USD 300 million in a funding round to expand its liquid biopsy portfolio and strengthen its position in the market (Qiagen press release).
- In May 2025, Thermo Fisher Scientific entered into a partnership with the University of California, San Francisco, to establish a Center of Excellence for Liquid Biopsy, focusing on research and development in this field (Thermo Fisher Scientific press release). These developments underscore the growing importance of liquid biopsy testing devices in breast cancer diagnostics and the significant investments being made by key players to drive innovation and market expansion.
Research Analyst Overview
- The breast cancer liquid biopsy testing market is experiencing significant advancements, driven by the integration of various technologies to improve disease monitoring and treatment response prediction. DNA sequencing platforms and high-throughput screening are key components of this market, enabling the detection of predictive and prognostic biomarkers. Point-of-care testing and automated platforms are gaining traction, enabling real-time analysis and enhancing assay performance. Microfluidic devices and mass spectrometry are essential for CTC enumeration and drug resistance detection, respectively. Data analysis pipelines and bioinformatics tools facilitate patient stratification and regulatory approval.
- Sample processing techniques and multi-omics profiling are essential for enhancing diagnostic accuracy and ensuring clinical validation studies. The market is focused on miniaturizing devices and validating biomarkers to improve accessibility and affordability. Regulatory approval remains a critical challenge, as these tests must meet stringent standards for diagnostic and treatment response prediction.
Dive into Technavio's robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Breast Cancer Liquid Biopsy Testing Devices Market insights. See full methodology.
|
Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
145 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 31.34% |
|
Market growth 2024-2028 |
USD 1164 million |
|
Market structure |
Fragmented |
|
YoY growth 2023-2024(%) |
23.41 |
|
Key countries |
US, Germany, Canada, France, and UK |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
What are the Key Data Covered in this Breast Cancer Liquid Biopsy Testing Devices Market Research and Growth Report?
- CAGR of the Breast Cancer Liquid Biopsy Testing Devices industry during the forecast period
- Detailed information on factors that will drive the growth and forecasting between 2024 and 2028
- Precise estimation of the size of the market and its contribution of the industry in focus to the parent market
- Accurate predictions about upcoming growth and trends and changes in consumer behaviour
- Growth of the market across North America, Europe, Asia, and Rest of World (ROW)
- Thorough analysis of the market's competitive landscape and detailed information about companies
- Comprehensive analysis of factors that will challenge the breast cancer liquid biopsy testing devices market growth of industry companies
We can help! Our analysts can customize this breast cancer liquid biopsy testing devices market research report to meet your requirements.

 RIA -
RIA - 
