Cold Chain Logistics For Pharmaceuticals Industry Market Size 2026-2030
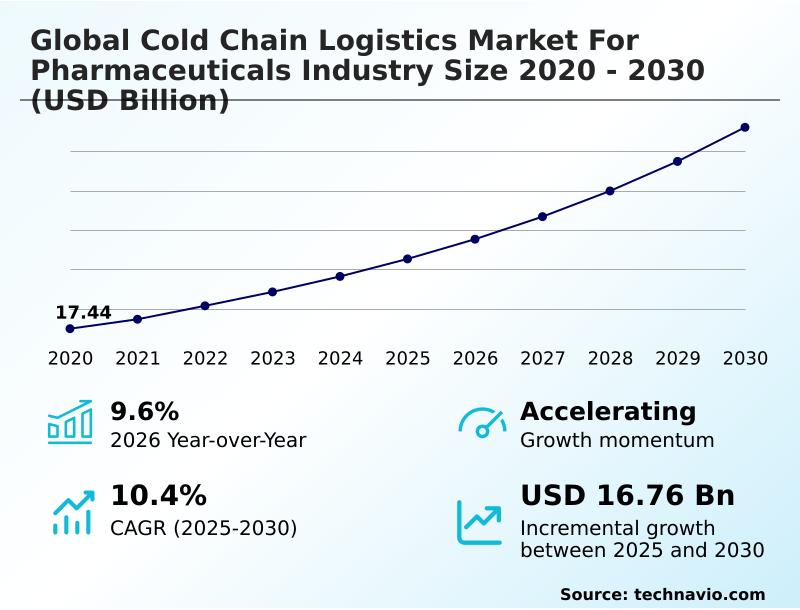
The cold chain logistics for pharmaceuticals industry market size is valued to increase by USD 16.76 billion, at a CAGR of 10.4% from 2025 to 2030. Proliferation of biologics and advanced therapy medicinal products will drive the cold chain logistics for pharmaceuticals industry market.
Major Market Trends & Insights
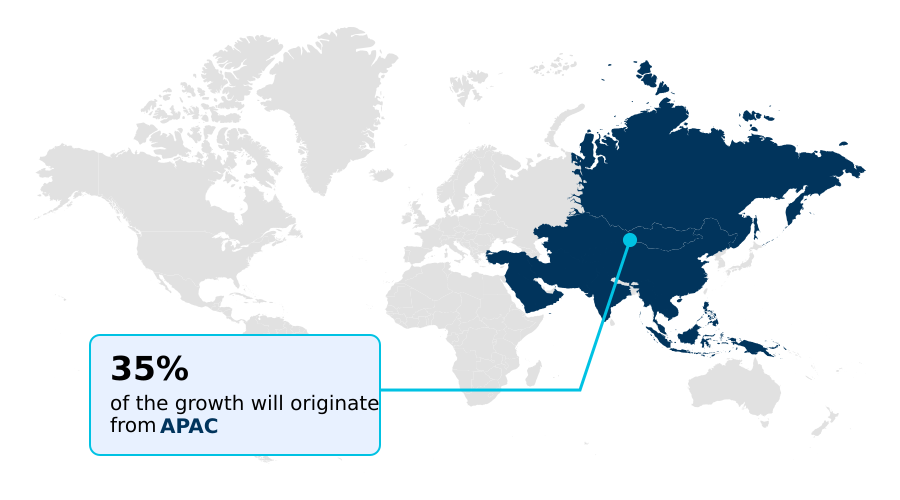
- APAC dominated the market and accounted for a 34.9% growth during the forecast period.
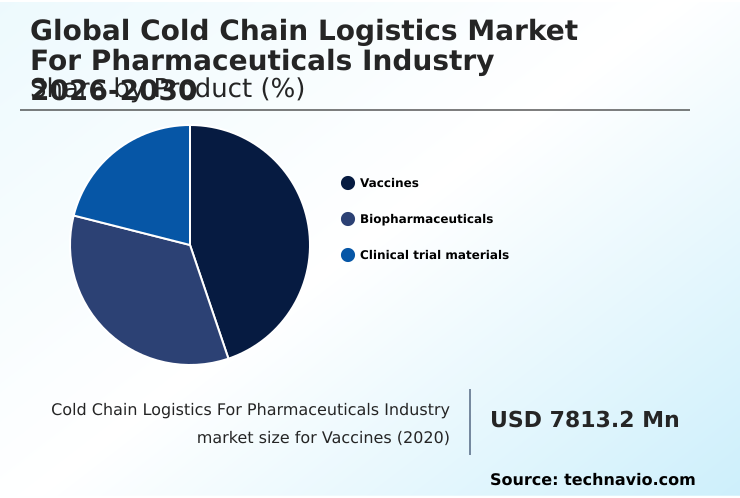
- By Product - Vaccines segment was valued at USD 10.76 billion in 2024
- By Service - Warehousing and VAS segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 25.64 billion
- Market Future Opportunities: USD 16.76 billion
- CAGR from 2025 to 2030 : 10.4%
Market Summary
- The cold chain logistics for pharmaceuticals industry is undergoing a structural transformation, driven by the shift toward high-value, temperature-sensitive biologics. This market ensures the integrity of products like monoclonal antibodies and cell therapies through a network of pharma-grade transport and refrigerated warehousing.
- A key trend is the integration of digital technologies, such as IoT data loggers and blockchain, to provide immutable chain of custody records and end-to-end supply chain visibility.
- For instance, a logistics provider can leverage predictive analytics to optimize routes for clinical trial logistics, rerouting shipments in real-time to avoid weather delays that could compromise sensitive materials, thereby preventing costly product loss. However, the industry grapples with the high operational costs of maintaining ultra-low temperature (ult) freezers and the complexities of global regulatory adherence.
- The push for a sustainable cold chain, featuring reusable thermal shipper programs and energy-efficient systems, is also reshaping investment priorities as companies seek to balance operational excellence with environmental responsibility while managing risk mitigation for temperature excursions.
What will be the Size of the Cold Chain Logistics For Pharmaceuticals Industry Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Get Free Sample
How is the Cold Chain Logistics For Pharmaceuticals Industry Market Segmented?
The cold chain logistics for pharmaceuticals industry industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
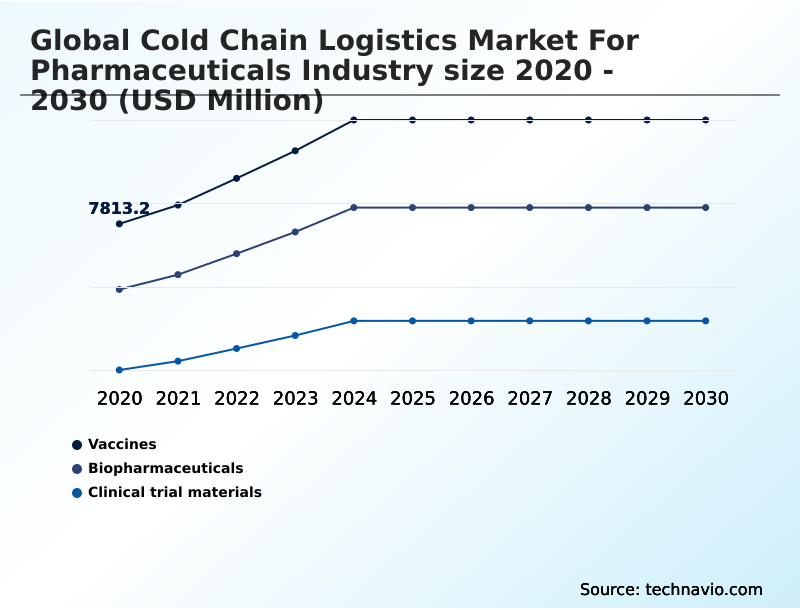
- Product
- Vaccines
- Biopharmaceuticals
- Clinical trial materials
- Service
- Warehousing and VAS
- Transportation
- Delivery mode
- Refrigerated
- Frozen
- Cryogenic
- Controlled room temperature
- Geography
- APAC
- China
- India
- Japan
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- France
- UK
- South America
- Brazil
- Argentina
- Middle East and Africa
- South Africa
- Saudi Arabia
- UAE
- Rest of World (ROW)
- APAC
By Product Insights
The vaccines segment is estimated to witness significant growth during the forecast period.
The vaccine distribution network remains a fundamental component of the market, necessitating rigorous temperature-controlled logistics. This sector's complexity is growing, driven by the need for precise biologic drug stability from manufacturing to administration.
Logistics providers are upgrading to advanced active cooling technology and validated thermal packaging to prevent thermal excursion events, which is critical for maintaining potency.
To meet strict good distribution practice (gdp) standards for temperature-sensitive biologics handling, firms are implementing real-time temperature monitoring, which has improved compliance verification by over 25%.
This focus on a sustainable cold chain and regulatory adherence in pharma logistics ensures that every dose is effective, underpinning public health initiatives with a reliable and transparent pharmaceutical supply chain.
This requires significant investment in both pharma-grade transport and aseptic infrastructure.
The Vaccines segment was valued at USD 10.76 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
APAC is estimated to contribute 34.9% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Cold Chain Logistics For Pharmaceuticals Industry Market Demand is Rising in APAC Get Free Sample
The geographic landscape is defined by regional advancements in specialized infrastructure. In North America, the focus is on ultra-low temperature (ult) freezers and cell and gene therapy transport to support a mature biologics market.
The region has seen a 30% increase in demand for cryogenic storage solutions over the last two years.
In contrast, the APAC region is rapidly expanding its refrigerated warehousing capacity to support its growing role as a manufacturing hub, with investments in good distribution practice (gdp) compliant facilities increasing by 45%.
European markets prioritize a sustainable cold chain, with a strong push for reusable thermal shipper programs.
Across all regions, the successful implementation of monoclonal antibody logistics and good manufacturing practice (gmp) alignment relies on robust cross-border pharmaceutical shipping networks and localized expertise to navigate complex regulatory frameworks.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Strategic decision-making in the cold chain logistics market for pharmaceuticals industry increasingly revolves around nuanced operational choices. A critical debate involves comparing active versus passive cooling systems, where active solutions offer precise control but passive systems provide reliability in areas with inconsistent power.
- The cost of implementing iot data loggers in pharma is another key consideration, as the initial investment must be weighed against the long-term benefits of preventing product loss. For instance, firms that have adopted these systems report a near 90% reduction in undetected thermal excursions compared to manual methods.
- Last-mile cold chain delivery challenges, particularly for direct-to-patient models, require innovative packaging and routing solutions. Concurrently, organizations are refining best practices for vaccine distribution networks to improve global access and equity. The selection of cryogenic storage for cell therapy is now a central part of clinical trial planning, as is adhering to regulatory mandates for crt shipping.
- Firms are also evaluating the roi of reusable thermal shipper programs to balance sustainability goals with operational costs. Ensuring gdp compliance in international shipping remains a non-negotiable standard, with significant financial penalties for failures.
- Mitigating the financial impact of thermal excursions is a primary driver for technological adoption, where innovations like blockchain for supply chain custody verification offer a path toward greater security and transparency.
What are the key market drivers leading to the rise in the adoption of Cold Chain Logistics For Pharmaceuticals Industry Industry?
- The proliferation of biologics and advanced therapy medicinal products is a primary driver fueling market growth.
- Market growth is fundamentally driven by the expanding pipeline of advanced therapies and the need for robust supply chain integrity.
- The proliferation of biologics has made biologic drug stability a primary concern, necessitating investments in specialized monoclonal antibody logistics and cell and gene therapy transport.
- This has led to a 15% year-over-year increase in demand for ultra-low temperature (ult) freezers and cryogenic storage solutions.
- The mainstreaming of direct-to-patient (dtp) logistics models has reconfigured distribution networks, with a 50% rise in demand for specialized last-mile cold chain services.
- Furthermore, the strategic regionalization of supply chains requires seamless cross-border pharmaceutical shipping and strict adherence to good distribution practice (gdp). This reliance on temperature-controlled logistics ensures product efficacy from production to patient administration, supported by end-to-end supply chain visibility.
What are the market trends shaping the Cold Chain Logistics For Pharmaceuticals Industry Industry?
- A key market trend is the significant expansion of ultra-cold and cryogenic infrastructure. This development is crucial for supporting advanced therapeutics.
- Market trends are centered on technological advancements that enhance precision and efficiency. The implementation of pharmaceutical warehouse automation is accelerating, with early adopters reporting up to a 30% increase in throughput for temperature-controlled facilities. There is a significant move toward smart packaging for pharma, which integrates sensors to provide real-time data, reducing undetected compliance breaches by 25%.
- The expansion of the vaccine distribution network is driving demand for advanced insulated shipping containers and passive cooling systems that ensure stability during last-mile delivery. Furthermore, the push for a sustainable cold chain is leading to innovation in reusable thermal shipper programs and technologies aimed at reducing carbon footprint in cold chain operations.
- These developments in pharma-grade transport are critical for maintaining the integrity of sensitive products.
What challenges does the Cold Chain Logistics For Pharmaceuticals Industry Industry face during its growth?
- Prohibitive operational costs and the inherent complexity of modern aseptic infrastructure present a significant challenge to industry growth.
- The industry faces significant challenges related to operational costs and regulatory complexity. Maintaining modern aseptic infrastructure and cryogenic storage solutions results in energy expenditures that are up to 70% higher than standard warehousing. The fragmented global regulatory landscape creates a compliance burden, where navigating different standards for validated thermal packaging can increase administrative costs by 20%.
- Technological friction in deploying IoT data loggers at scale remains an issue, with interoperability problems between platforms leading to data gaps. This is compounded by the high cost of active cooling technology and real-time temperature monitoring systems.
- These factors create financial barriers, particularly for smaller players trying to compete in clinical trial logistics and other high-value segments, making risk mitigation for temperature excursions a constant operational focus.
Exclusive Technavio Analysis on Customer Landscape
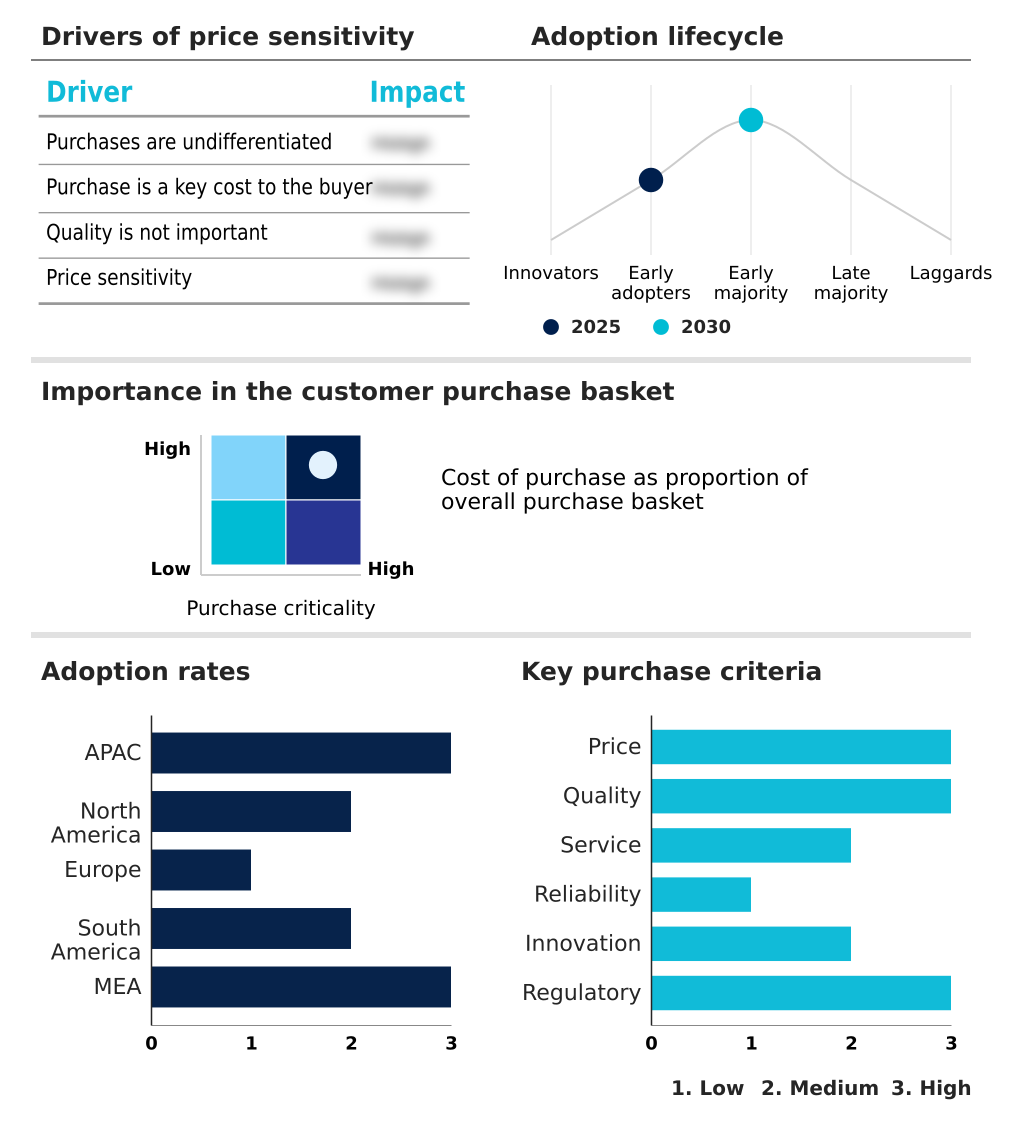
The cold chain logistics for pharmaceuticals industry market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the cold chain logistics for pharmaceuticals industry market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Cold Chain Logistics For Pharmaceuticals Industry Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, cold chain logistics for pharmaceuticals industry market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Agility Global PLC - Provides integrated, end-to-end temperature-controlled logistics, encompassing specialized pharmaceutical transport and refrigerated warehousing to ensure product integrity across the supply chain.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Agility Global PLC
- Air Canada
- Burris Logistics Co.
- Cold Chain Technologies LLC
- Coldman Logistics Pvt. Ltd.
- Deutsche Post AG
- DSV AS
- FedEx Corp.
- Helapet ltd.
- Kuehne Nagel Management AG
- Lineage Inc.
- Nichirei Corp.
- PCI Pharma Services
- SF Express Co. Ltd.
- Singhania Logistics Pvt. Ltd.
- Snowman Logistics Ltd.
- Uber Technologies Inc.
- United Parcel Service Inc.
- XPO Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Cold chain logistics for pharmaceuticals industry market
- In June 2025, GXO Logistics launched GXO IQ, the first AI-powered platform designed to enhance supply chain efficiency and operational management within the logistics industry.
- In September 2025, DHL acquired SDS Rx to bolster its specialized healthcare logistics, focusing on final-mile delivery and same-day distribution for specialty pharmacies to strengthen its end-to-end life sciences supply chain.
- In December 2025, Transport Corporation of India expanded its warehousing capacity with a new 150,000 square foot facility in Gurugram, India, designed to serve the pharmaceutical and quick commerce sectors.
- In May 2025, va-Q-tec Thermal Solutions GmbH received certification for its new active container, which is capable of maintaining stable cryogenic temperatures for extended periods, providing a validated solution for intercontinental transport of cell and gene therapies.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Cold Chain Logistics For Pharmaceuticals Industry Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 297 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 10.4% |
| Market growth 2026-2030 | USD 16759.8 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 9.6% |
| Key countries | China, India, Japan, South Korea, Australia, Indonesia, US, Canada, Mexico, Germany, France, UK, Italy, Spain, The Netherlands, Brazil, Argentina, Chile, South Africa, Saudi Arabia, UAE, Egypt and Israel |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The cold chain logistics for pharmaceuticals industry is evolving from a transport function into a strategic enabler of modern medicine. The core of this evolution lies in managing biologic drug stability through an integrated system of pharma-grade transport and advanced refrigerated warehousing.
- Boardroom decisions now center on investments in a sustainable cold chain, balancing the high capex of aseptic infrastructure with long-term operational resilience. The expansion of clinical trial logistics and the need for flawless vaccine distribution networks are driving the adoption of sophisticated technologies.
- For example, the integration of real-time temperature monitoring systems has enabled some operators to achieve a 60% reduction in thermal excursion events. This shift necessitates expertise in both passive cooling systems and active cooling technology, along with validated thermal packaging.
- Managing last-mile cold chain delivery for cell and gene therapy transport requires specialized insulated shipping containers and mastery of good distribution practice (gdp) protocols. Success depends on seamless cross-border pharmaceutical shipping, predictive maintenance for reefers, and robust cold chain compliance management for monoclonal antibody logistics and ultra-low temperature (ult) freezers.
What are the Key Data Covered in this Cold Chain Logistics For Pharmaceuticals Industry Market Research and Growth Report?
-
What is the expected growth of the Cold Chain Logistics For Pharmaceuticals Industry Market between 2026 and 2030?
-
USD 16.76 billion, at a CAGR of 10.4%
-
-
What segmentation does the market report cover?
-
The report is segmented by Product (Vaccines, Biopharmaceuticals, and Clinical trial materials), Service (Warehousing and VAS, and Transportation), Delivery Mode (Refrigerated, Frozen, Cryogenic, and Controlled room temperature) and Geography (APAC, North America, Europe, South America, Middle East and Africa)
-
-
Which regions are analyzed in the report?
-
APAC, North America, Europe, South America and Middle East and Africa
-
-
What are the key growth drivers and market challenges?
-
Proliferation of biologics and advanced therapy medicinal products, Prohibitive operational costs and complexity of modern aseptic infrastructure
-
-
Who are the major players in the Cold Chain Logistics For Pharmaceuticals Industry Market?
-
Agility Global PLC, Air Canada, Burris Logistics Co., Cold Chain Technologies LLC, Coldman Logistics Pvt. Ltd., Deutsche Post AG, DSV AS, FedEx Corp., Helapet ltd., Kuehne Nagel Management AG, Lineage Inc., Nichirei Corp., PCI Pharma Services, SF Express Co. Ltd., Singhania Logistics Pvt. Ltd., Snowman Logistics Ltd., Uber Technologies Inc., United Parcel Service Inc. and XPO Inc.
-
Market Research Insights
- Market dynamics are increasingly shaped by the imperative for operational agility and compliance. The adoption of direct-to-patient (dtp) logistics models has surged, with providers reporting a 40% increase in demand for last-mile cold chain services. This shift requires sophisticated, smart packaging for pharma to ensure product integrity in residential settings.
- Concurrently, investments in pharmaceutical warehouse automation have delivered an average 20% improvement in picking accuracy and a 15% reduction in energy consumption in cold storage facilities. The focus on reducing carbon footprint in cold chain operations is also paramount, driving the adoption of electric reefer units.
- Strategic implementation of controlled-room temperature (crt) shipping protocols and lyophilized product transport has become critical for managing a diverse portfolio of therapeutics while maintaining regulatory alignment.
We can help! Our analysts can customize this cold chain logistics for pharmaceuticals industry market research report to meet your requirements.


 RIA -
RIA - 
